1
0118 Inorganic Chemistry, Vol. 49, No. 21, 2010
Apfel et al.
calculated that protonation of the Fe-Fe bond pair should
their olfactory properties, and the carbon/silicon switch
strategy has been successfully used for the development of
7
a
be favored. In contrast to that report, we investigated the
12
chemical and electrochemical properties of [Fe ( μ-SCH -
new silicon-based odorants. Recently, we have also shown
that the impact and friction sensitivity of explosives can be
2
2
SCH S)(CO) ] and observed interaction of pivalic acid with
2
6
9
13
at least one sulfur atom. This might be an alternative
pathway for the formation of hydrides via an initial proton-
ation of the thiolato sulfur atoms.
affected by carbon/silicon exchange.
The main focus of this work was the synthesis of iron
complexes containing silicon-based thiolato ligands derived
Inspired by these results and investigations of Glass et al.
from (mercaptomethyl)silanes of the formula type R Si-
n
10
on [Fe {μ-SCH Sn(CH ) CH S}(CO) ], which exhibits an
(CH SH)
(n = 0-2; R = organyl) and the determination
2
2
3 2
2
6
2
4-n
increased electron density at the thiolato sulfur atoms by
hyperconjugation of the σ(Sn-C) and 3p(S) orbitals, we have
prepared a series of model compounds for the [FeFe] hydro-
genase with silicon-containing thiolato ligands bridging the
diiron moiety. These complexes should offer the possibility to
investigate protonation processes at the coordinating thiolato
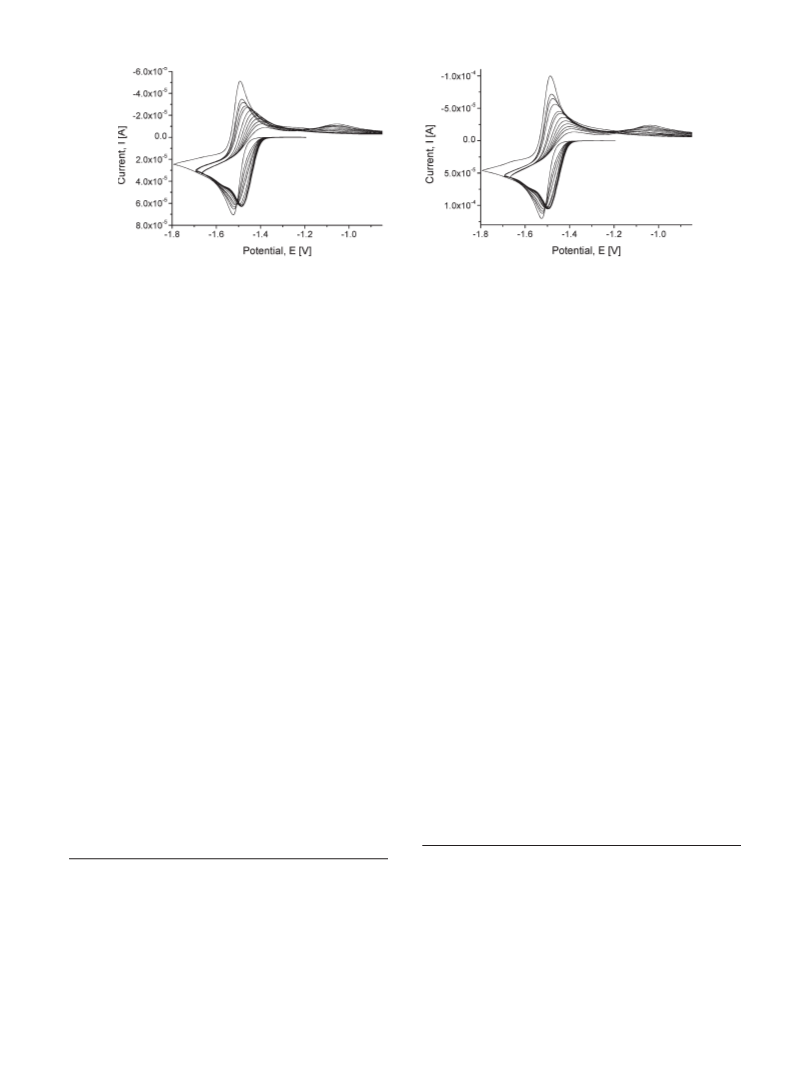
sulfur atoms by spectroscopic (IR, NMR) as well as electro-
chemical (cyclic voltammetry, difference pulse voltammetry)
techniques.
of the electrochemical properties of these complexes (in
comparison to the corresponding carbon analogues) to
investigate the influence of silicon on electrocatalysis and
the formation of dihydrogen. These studies were also per-
formed as part of our systematic investigations on function-
alized tetraorganylsilanes of the formula type R Si(CH X)
(n = 0-3; R = organyl; X = functional group).
n
2
4-n
13,14
Results and Discussion
It is well-known from silicon-containing drugs that the
presence of one or more silicon atoms in these molecules
influences their chemical, physical, and biological properties.
The pharmacodynamics and pharmacokinetics of drugs can
be affected significantly by replacement of a carbon by a
silicon atom (sila-substitution). This can be exploited in
medicinal chemistry for drug design; and indeed, the carbon/
silicon switch strategy has been successfully used for the
development of new silicon-based drugs. Likewise, sila-
substitution of odorants has also been demonstrated to affect
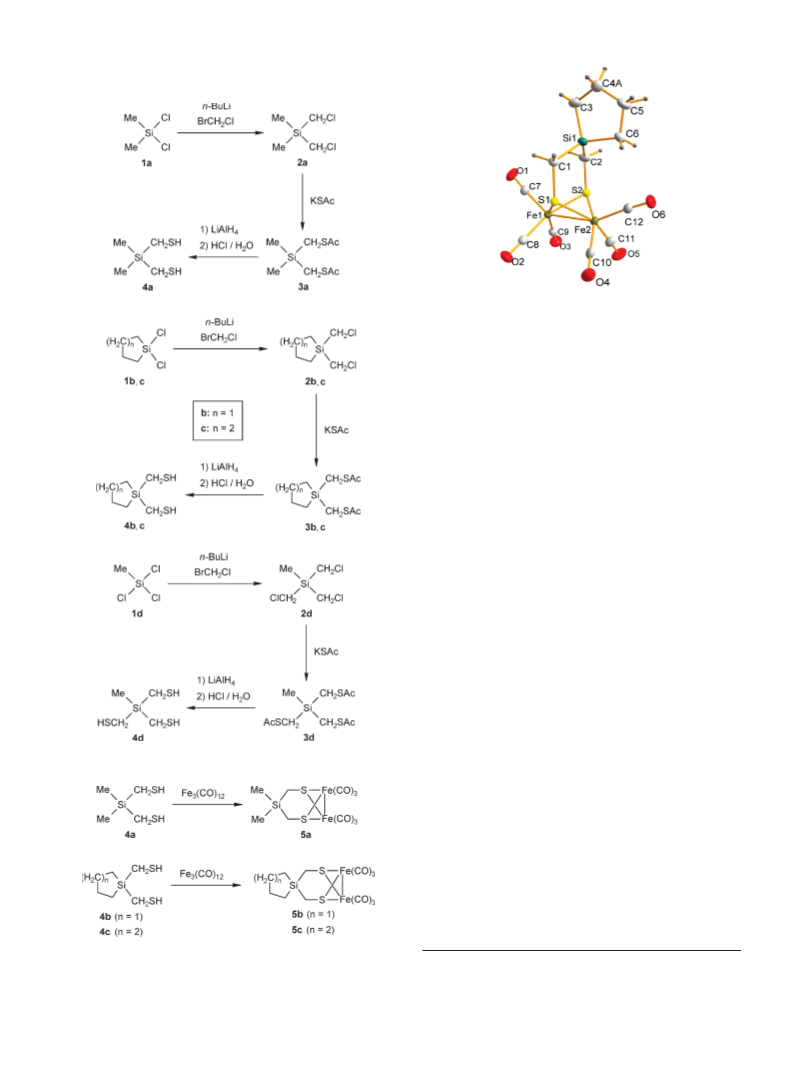
Synthesis and Characterization of the Silicon-Containing
Thiolato Ligands. The di- and trifunctional (mercapto-
methyl)silanes 4a-d were synthesized according to Scheme 1,
starting from the respective chlorosilanes 1a-d. Thus,
treatment of 1a-d with (chloromethyl)lithium, generated
in situ from bromochloromethane and n-butyllithium in
tetrahydrofuran (THF), afforded the (chloromethyl)-
silanes 2a-d (37-77% yield), which upon treatment with
potassium thioacetate in THF furnished the corresponding
11
15
(acetylthiomethyl)silanes 3a-d (77-97% yield). Reaction
of 3a-d with lithium aluminum hydride in diethyl ether,
followed by workup with hydrochloric acid, finally af-
forded the respective (mercaptomethyl)silanes 4a-d
(
5) (a) Borg, S. J.; Behrsing, T.; Best, S. P.; Razavet, M.; Liu, X.; Pickett,
C. J. J. Am. Chem. Soc. 2004, 126, 16988–16999. (b) Chong, D. S.; Georgakaki,
I. P.; Mejia-Rodriguez, R.; Sanabria- Chinchilla, J.; Soriaga, M. P.; Darensbourg,
M. Y. Dalton Trans. 2003, 4158–4163.
(
67-92% yield). Compounds 2a-d, 3a-d, and 4a-d were
(
6) (a) Capon, J.-F.; Ezzaher, S.; Gloaguen, F.; P ꢀe tillon, F. Y.; Schollhammer,
isolated as liquids. Their identities were established by
1
13
29
P.; Talarmin, J. Chem.;Eur. J. 2008, 14, 1954–1964. (b) Olsen, M. T.; Barton,
B. E.; Rauchfuss, T. B. Inorg. Chem. 2009, 48, 7507–7509.
(
Darensbourg, M. Y. J. Am. Chem. Soc. 2003, 125, 518–524. (b) Tye, J. W.;
Darensbourg, M. Y.; Hall, M. B. J. Mol. Struct.: THEOCHEM 2006, 771,
elemental analyses (C, H, S) and H, C, and Si NMR
spectroscopic studies (solvent, CDCl3).
7) (a) Zhao, X.; Chiang, C.-Y.; Miller, M. L.; Rampersad, M. V.;
Synthesis and Characterization of the Iron Complexes.
Treatment of the bis(mercaptomethyl)silanes 4a-c with
1
23–128.
(
8) (a) Windhager, J.; Seidel, R. A.; Apfel, U.-P.; G o€ rls, H.; Linti, G.;
Weigand, W. Chem. Biodiversity 2008, 5, 2023–2041. (b) Windhager, J.; Apfel,
U.-P.; Yoshino, T.; Nakata, N.; G €o rls, H.; Rudolph, M.; Ishii, A.; Weigand, W.
Chem. Asian J. 2010, 5, 1600-1610. (c) Liu, T.; Li, B.; Singleton, M. L.;
Hall, M. B.; Darensbourg, M. Y. J. Am. Chem. Soc. 2009, 131, 8296–8307. (d)
Li, B.; Liu, T.; Singleton, M. L.; Darensbourg, M. Y. Inorg. Chem. 2009, 48,
(12) Recent publications dealing with sila-substituted odorants:
(a) B u€ ttner, M. W.; Penka, M.; Doszczak, L.; Kraft, P. Organometallics
2007, 26, 1295–1298. (b) Doszczak, L.; Kraft, P.; Weber, H.-P.; Bertermann, R.;
Triller, A.; Hatt, H.; Tacke, R. Angew. Chem. 2007, 119, 3431–3436. Angew.
Chem., Int. Ed. 2007, 46, 3367-3371. (c) B €u ttner, M. W.; Metz, S.; Kraft, P.;
Tacke, R. Organometallics 2007, 26, 3925–3929. (d) B €u ttner, M. W.; Burschka,
C.; Junold, K.; Kraft, P.; Tacke, R. ChemBioChem 2007, 8, 1447–1454.
(e) B €u ttner, M. W.; N €a tscher, J. B.; Burschka, C.; Tacke, R. Organometallics
8
393–8403.
9) Windhager, J.; Rudolph, M.; Br €a utigam, S.; G o€ rls, H.; Weigand, W.
Eur. J. Inorg. Chem. 2007, 2748–2760.
10) Glass, R. S.; Gruhn, N. E.; Lorance, E.; Singh, M. S.; Stessman,
N. Y. T.; Zakai, U. I. Inorg. Chem. 2005, 44, 5728–5737.
11) Recent publications dealing with sila-substituted drugs: (a) Showell,
(
(
2
007, 26, 4835–4838. (f ) Tacke, R.; Metz, S. Chem. Biodiversity 2008, 5, 920–
9
41. (g) Metz, S.; N €a tscher, J. B.; Burschka, C.; G €o tz, K.; Kaupp, M.; Kraft, P.;
(
Tacke, R. Organometallics 2009, 28, 4700–4712. (h) N €a tscher, J. B.; Laskowski,
G. A.; Barnes, M. J.; Daiss, J. O.; Mills, J. S.; Montana, J. G.; Tacke, R.;
Warneck, J. B. H. Bioorg. Med. Chem. Lett. 2006, 16, 2555–2558. (b) Ilg, R.;
Burschka, C.; Schepmann, D.; W €u nsch, B.; Tacke, R. Organometallics 2006, 25,
N.; Kraft, P.; Tacke, R. ChemBioChem 2010, 11, 315–319.
(13) Recent publications dealing with sila-substituted explosives:
(
a) Klap o€ tke, T. M.; Krumm, B.; Ilg, R.; Troegel, D.; Tacke, R. J. Am.
5
396–5408. (c) B €u ttner, M. W.; Burschka, C.; Daiss, J. O.; Ivanova, D.; Rochel,
Chem. Soc. 2007, 129, 6908–6915. (b) Evangelisti, C.; Klap €o tke, T. M.; Krumm,
N.; Kammerer, S.; Peluso-Iltis, C.; Bindler, A.; Gaudon, C.; Germain, P.; Moras,
D.; Gronemeyer, H.; Tacke, R. ChemBioChem 2007, 8, 1688–1699. (d) Tacke,
R.; Popp, F.; M €u ller, B.; Theis, B.; Burschka, C.; Hamacher, A.; Kassack, M. U.;
Schepmann, D.; W €u nsch, B.; Jurva, U.; Wellner, E. ChemMedChem 2008, 3,
B.; Nieder, A.; Berger, R. J. F.; Hayes, S. A.; Mitzel, N. W.; Troegel, D.; Tacke, R.
Inorg. Chem. 2010, 49, 4865–4880.
(14) Recent publications dealing with functional tetraorganylsilanes of
n
2
4-n
the formula type R Si(CH X) (R = organyl; X = functional group; n =
1
52–164. (e) Warneck, J. B.; Cheng, F. H. M.; Barnes, M. J.; Mills, J. S.; Montana,
0-3): (a) Daiss, J. O.; Bart, K. A.; Burschka, C.; Hey, P.; Ilg, R.; Klemm, K.;
Richter, I.; Wagner, S. A.; Tacke, R. Organometallics 2004, 23, 5193–5197.
(b) Ilg, R.; Troegel, D.; Burschka, C.; Tacke, R. Organometallics 2006, 25, 548–
551. (c) Troegel, D.; Walter, T.; Burschka, C.; Tacke, R. Organometallics 2009,
28, 2756–2761. (d) Troegel, D.; M €o ller, F.; Burschka, C.; Tacke, R. Organome-
tallics 2009, 28, 5765–5770. (e) Weidner, T.; Ballav, N.; Siemeling, U.; Troegel,
D.; Walter, T.; Tacke, R.; Castner, D. G.; Zharnikov, M. J. Phys. Chem. C 2009,
113, 19609–19617. (f ) Troegel, D.; Lippert, W. P.; M €o ller, F.; Burschka, C.;
J. G.; Naylor, R. J.; Ngan, M.-P.; Wai, M.-K.; Daiss, J. O.; Tacke, R.; Rudd, J. A.
Toxicol. Appl. Pharmacol. 2008, 232, 369–375. (f ) Lippert, W. P.; Burschka, C.;
G €o tz, K.; Kaupp, M.; Ivanova, D.; Gaudon, C.; Sato, Y.; Antony, P.; Rochel, N.;
Moras, D.; Gronemeyer, H.; Tacke, R. ChemMedChem 2009, 4, 1143–1152.
(
g) Tacke, R.; M €u ller, V.; B €u ttner, M. W.; Lippert, W. P.; Bertermann, R.; Daiss,
J. O.; Khanwalkar, H.; Furst, A.; Gaudon, C.; Gronemeyer, H. ChemMedChem
009, 4, 1797–1802. (h) Johansson, T.; Weidolf, L.; Popp, F.; Tacke, R.; Jurva, U.
2
Drug Metab. Dispos. 2010, 38, 73–83. (i) Tacke, R.; Nguyen, B.; Burschka, C.;
Lippert, W. P.; Hamacher, A.; Urban, C.; Kassack, M. U. Organometallics 2010,
Tacke, R. J. Organomet. Chem. 2010, 695, 1700–1707.
(15) (a) Kobayashi, T.; Pannell, K. H. Organometallics 1990, 9, 2201–
2203. (b) Kobayashi, T.; Pannell, K. H. Organometallics 1991, 10, 1960–1964.
2
9, 1652–1660.

 Apfel, Ulf-Peter
Apfel, Ulf-Peter
 Troegel, Dennis
Troegel, Dennis
 Halpin, Yvonne
Halpin, Yvonne
 Tschierlei, Stefanie
Tschierlei, Stefanie
 Uhlemann, Ute
Uhlemann, Ute
 Goerls, Helmar
Goerls, Helmar
 Schmitt, Michael
Schmitt, Michael
 Popp, Juergen
Popp, Juergen
 Dunne, Peter
Dunne, Peter
 Venkatesan, Munuswamy
Venkatesan, Munuswamy
 Coey, Michael
Coey, Michael
 Rudolph, Manfred
Rudolph, Manfred
 Vos, Johannes G.
Vos, Johannes G.
 Tacke, Reinhold
Tacke, Reinhold
 Weigand, Wolfgang
Weigand, Wolfgang