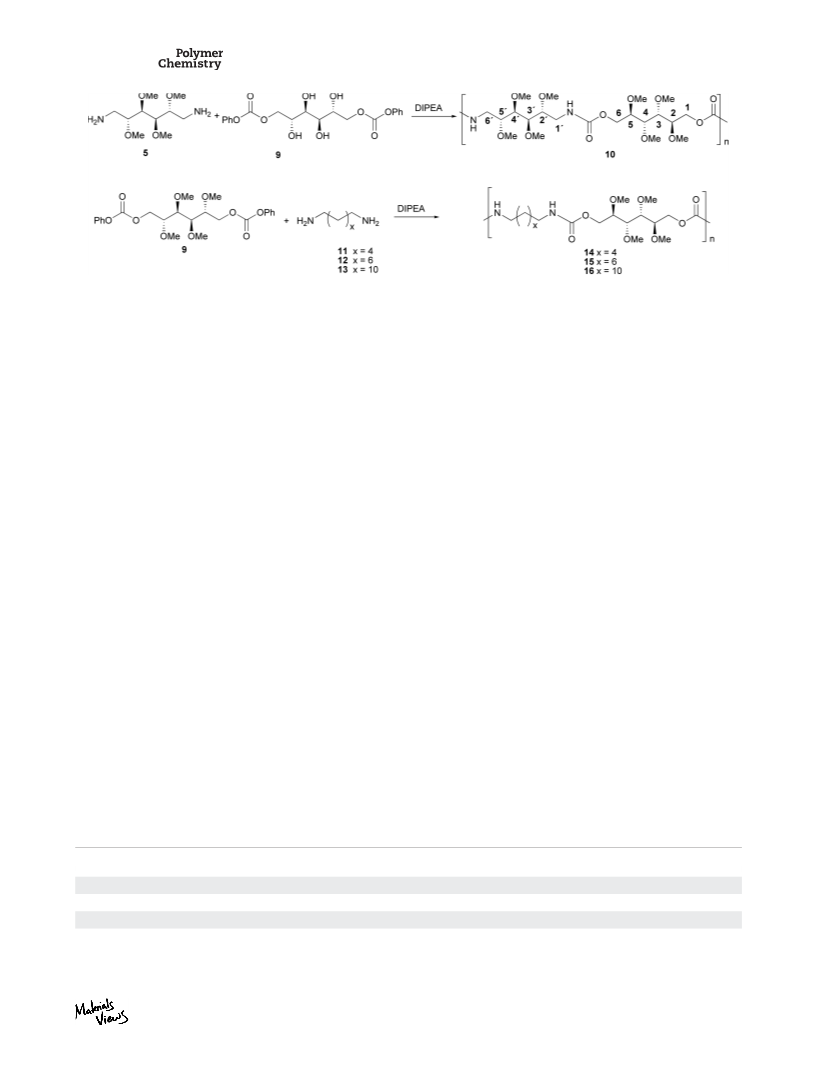
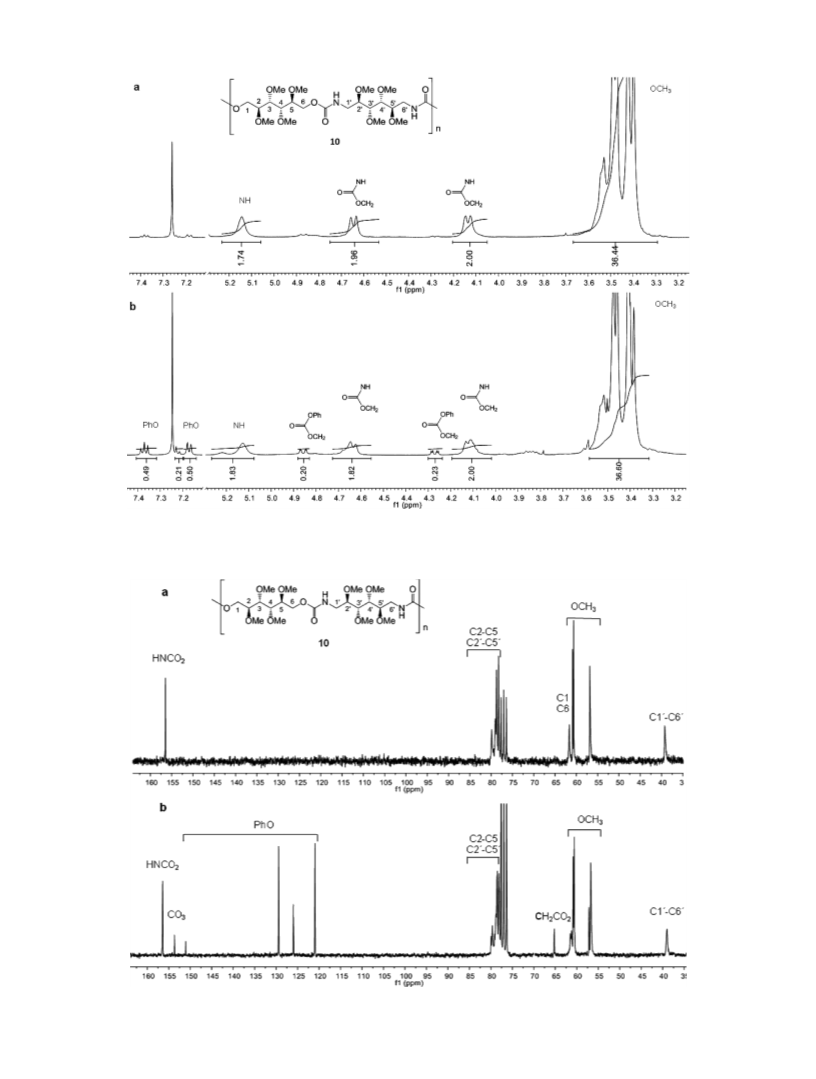
JOURNAL OF
POLYMER SCIENCE
ARTICLE
WWW.POLYMERCHEMISTRY.ORG
TABLE 1 Polymerization of Comonomers 5 and 9 and Properties of Polyurethane 10
a
a
b
Entry
Solvent
Monomer (M)
DIPEA (M)
Time (d)
Yield (%)
Mw
Mn
Mw/Mn
Mn
1
2
3
4
5
a
DMF
DMF
THF
THF
THF
0.1
0.5
0.1
0.5
0.5
0.4
3.0
0.4
3.0
1.5
2
4
4
7
4
58
57
72
61
70
4,850
4,160
4,390
3,750
1.10
1.11
1.21
1.11
1.40
4,710
4,100
5,170
4,270
5,230
14,170
21,020
12,780
14,960
10,180
11,550
b
Determined by SEC.
Determined by end-group analysis based on 1H NMR spectroscopy.
HRMS (ESI, Q-Tof, m/z): [M þ Na]þ, calcd for [C10H22O6Na]þ:
156.5 (CO2NH). Anal. calcd for [C22H42N2O12.(H2O)0.5]n: C
49.34, H 8.09, N 5.23. Found: C 49.13, H 7.96, N 5.37.
261.1309, found: 261.1318.
Polyurethanes 14 and 15 were prepared as described for
10. In the case of 16, DIPEA was used as solvent to give a
0.5 M initial concentration of comonomers 9 and 13. The po-
1,6-Di-O-phenyloxycarbonyl-2,3,4,5-tetra-O-methyl-D-
mannitol (9)
To a solution of Compound 8 (0.2 g, 0.8 mmol) in dry pyri-
dine (2.5 mL), phenylchloroformate (0.28 mL, 1.81 mmol)
was added. The mixture was stirred at rt for 2 h. After sub-
sequent addition of MeOH and toluene, the mixture was con-
centrated and purified by column chromatography (hexane/
EtOAc 4:1) to yield 9 as a white crystalline solid (0.33 g,
ꢀ
lymerization reaction was conducted at 85 C for 48 h.
25
Polyurethane 14, [a]D ¼ þ11.0 (c ¼ 0.8 in CHCl3); IR
(KBr): m ¼ 3334 (w, NAH), 2937 (s, CAH), 1704 (s, C¼¼O),
1539 cmꢁ1 (s, NAH); 1H NMR (500 MHz, CDCl3, d): 1.33
(4H, s, CH2-3’, CH2-4’), 1.50 (4H, t, J2’,3’ (J4’,5’) 6.1 Hz, CH2-2’,
CH2-5’), 3.17 (4H, q, J1’,2’ ¼ J1’,NH (J5’,6’¼ J6’,NH) 6.1 Hz, CH2-1’,
CH2-6’), 3.40, 3.45 (6H each, 2s, CH3O), 3.47 (2H, d, H-2, H-
5), 3.54 (2H, d, J2,3 (J4,5) 8.1 Hz, H-3, H-4), 4.08 (2H, d, J1a,1b
(J6a,6b) 11.7 Hz, H-1a, H-6a), 4.65 (2H, d, J1a,1b (J6a,6b) 11.7
Hz, H-1b, H-6b), 4.90 (2H, br s, NH). 13C NMR (125.7 MHz,
CDCl3, d): 26.3 (CH2-3’, CH2-4’), 29.8 (CH2-2’, CH2-5’), 40.9
(CH2-1’, CH2-6’), 56.7, 60.6 (CH3O), 61.0 (CH2O), 78.2, 78.5
(CHOCH3), 156.4 (CO2NH). Anal. calcd for [C20H40N2O8]n: C
53.19, H 8.43, N 6.89. Found: C 53.21, H 8.20, N 6.90.
25
85% from 5); mp 97–99 ꢀC; [a]D ¼ þ42.0 (c ¼ 1.0 in
CHCl3); IR (KBr): m ¼ 1764 cmꢁ1 (s, C¼¼O); 1H NMR (500
MHz, CDCl3, d): 3.50 (6H, s, CH3O), 3.53 (6H, s, CH3O), 3.59
(2H, ddd, J1a,2 (J5,6a) 2.3, J1b,2 (J5,6b) 3.4, J2,3 (J4,5) 8.0 Hz, H-
2/H-5), 3.66 (2H, d, J2,3 (J4,5) 8.0 Hz, H-3/H-4), 4.33 (2H, dd,
J1a,1b (J6a,6b) 12.0, J1b,2 (J5,6b) 3.4 Hz, H-1b/H-6b), 4.92 (2H,
dd, J1a,1b (J6a,6b) 12.0, J1a,2 (J5,6a) 2.1 Hz, H-1a/H-6a), 7.43-
7.22 (10H, m, H-aromatic); 13C NMR (125.7 MHz, CDCl3, d):
57.2, 60.8 (CH3O), 65.2 (C-1/C-6), 78.2 (C-3/C-4), 78.7 (C-2/
C-5), 121.0, 126.0, 129.4, 151.1 (C-aromatic), 153.7 (CO).
Anal. calcd for C24H30O10: C 60.24, H 6.32. Found: C 60.50, H
25
Polyurethane 15, [a]D ¼ þ9.4 (c ¼ 0.8 in CHCl3); IR (KBr):
6.68. HR-MS (ESI, Q-Tof, m/z): [M
þ
Na]þ, calcd for
m ¼ 3338 (w, NAH), 2932 (s, CAH), 1704 (s, C¼¼O), 1535
cmꢁ1 (s, NAH); 1H NMR (500 MHz, CDCl3, d): 1.27 (8H, s,
CH2-3’ꢁCH2-6’), 1.46 (4H, s, CH2-2’, CH2-7’), 3.14 (4H, d,
J1’,NH (J8’,NH) 5.4 Hz, CH2-1’, CH2-8’), 3.38, 3.43 (6H each, 2s,
[C24H30O10Na]þ: 501.1731, found: 501.1732.
General Procedure for the Polymerization
To an equimolar mixture of a,x-diamine (5, 11–13) and 9
(0.2–0.3 mmol) in THF or DMF, DIPEA was added. The mix-
ture was stirred under argon atmosphere under the condi-
tions indicated in Table 1. The reaction temperature was 85
ꢀC, except for Entry 2 that was 100 ꢀC. Then, the solvent
was evaporated, and the residue was purified by dissolution
in CH2Cl2 and precipitation with hexane. This procedure was
repeated three times.
CH3O), 3.38-3.52 (4H, m, H-2ꢁH-5), 4.06 (2H, d, J1a,1b (J6a,6b
)
12.2 Hz, H-1a, H-6a), 4.63 (2H, d, J1a,1b (J6a,6b) 12.2 Hz, H-1b,
H-6b), 4.85 (2H, br s, NH). 13C NMR (125.7 MHz, CDCl3, d):
26.6 (CH2-4’, CH2-5’), 29.1 (CH2-3’, CH2-6’), 29.8 (CH2-2’, CH2-
7’), 41.0 (CH2-1’, CH2-8’), 56.7, 60.6 (CH3O), 60.9 (CH2O),
78.1, 78.4 (CHOCH3), 156.3 (CO2NH). Anal. calcd for
[C20H38N2O8.(H2O)0.5]n: C 54.16, H 8.86, N 6.32. Found: C
53.96, H 8.84, N 6.16.
25
Preparative Scale Synthesis of Polyurethanes 10, 14–16
For the synthesis of 10 in preparative scale, the dicarbonate
9 (0.223 g, 0.47 mmol) and the diamine 5 (0.111 g, 0.47
mmol) were dissolved in THF, and the polymerization was
conducted under the conditions of Table 1, Entry 5. Polyur-
Polyurethane 16, [a]D ¼ þ7.3 (c ¼ 1.2 in CHCl3); IR (KBr):
m ¼ 3337 (w, NAH), 2928 (s, CAH), 1704 (s, C¼¼O), 1535
cmꢁ1 (s, NAH); 1H NMR (500 MHz, CDCl3, d): 1.23, 1.26
(16H, 2 s, H-3’ꢁH-10’), 1.47 (4H, m, H-2’, H-11’), 3.16 (4H, d,
J1’,NH (J12’,NH) 6.2 Hz, H-1’, H-12’), 3.40, 3.44 (6H each, 2s,
25
ethane 10 was isolated as a white foam; [a]D ¼ þ11.4 (c
CH3O), 3.40–3.53 (4H, m, H-2ꢁH-5), 4.07 (2H, dd, J1a,2 (J5,6a,
)
¼ 0.7 in MeOH); IR (KBr): m ¼ 3338 (w, NAH), 1718 (s,
C¼¼O), 1524 cmꢁ1 (s, NAH); 1H NMR (500 MHz, CDCl3, d)
3.42, 3.43, 3.48, 3.53 (6H each, 4s, CH3O), 3.41–3.54 (12H,
m, CHOCH3, CH2N), 4.14 (2H, d, J1a,1b (J6a,6b) 10.9 Hz, CH2O),
4.66 (2H, d, J1a,1b (J6a,6b) 10.9 Hz, CH2O), 5.15 (2H, br s, NH).
13C NMR (125.7 MHz, CDCl3, d) 39.3 (CH2N), 56.9, 60.7, 60.9
(4 CH3O), 61.7 (CH2O), 78.3, 78.7, 79.1, 79.9 (CHOCH3),
2.8, J1a,ab (J6a,6b) 12.2 Hz, H-1a, H-6a), 4.63 (2H, d, J1a,ab
(J6a,6b) 12.2 Hz, H-1b, H-6b), 4.81 (2H, br s, NH). 13C NMR
(125.7 MHz, CDCl3, d): 26.7, 29.2, 29.5 (CH2-3’ꢁCH2-10’),
29.9 (CH2-2’, CH2-11’), 41.1 (CH2-1’, CH2-12’), 56.7, 60.6
(CH3O), 61.0 (CH2O), 78.2, 78.4 (C-2ꢁC-5), 156.3 (CO2NH).
Anal. calcd for C24H46N2O8: C 58.75, H 9.45, N 5.71. Found:
C 58.51, H 9.64, N 5.60.
WWW.MATERIALSVIEWS.COM
JOURNAL OF POLYMER SCIENCE PART A: POLYMER CHEMISTRY 2013, 51, 463–470
465

 Fidalgo, Daniela M.
Fidalgo, Daniela M.
 Kolender, Adriana A.
Kolender, Adriana A.
 Varela, Oscar
Varela, Oscar