1
374
H. Xu et al. / Tetrahedron: Asymmetry 25 (2014) 1372–1375
of Japonilure in 90% yield with 93% ee.20 Similarly, the synthesis of
the Osaka Beetle pheromone (S)-1 with 93% ee was accomplished
via asymmetric alkynylation of 4 with (S,S)-ProPhenol. The racemic
and n-BuLi (288 mL, 2.5 M in hexanes, 720 mmol) was added slowly.
The mixture was stirred for 3 h at À30 °C, and then 1-bromooctane
(31.2 mL, 180 mmol) was added dropwise via syringe. The resulting
mixture was stirred for another 30 min and warmed to room tem-
perature. After stirring for 24 h at room temperature, the reaction
mixture was quenched with water (50 mL) at 0 °C. The aqueous
phase was extracted with ether, and the combined organic phases
were washed with saturated brine solution, dried over anhydrous
1
was obtained by repeating the similar approach from (±)-5,
which was prepared from the direct alkynylation of propargylic
aldehyde 4 without any chiral ligand.1
7a
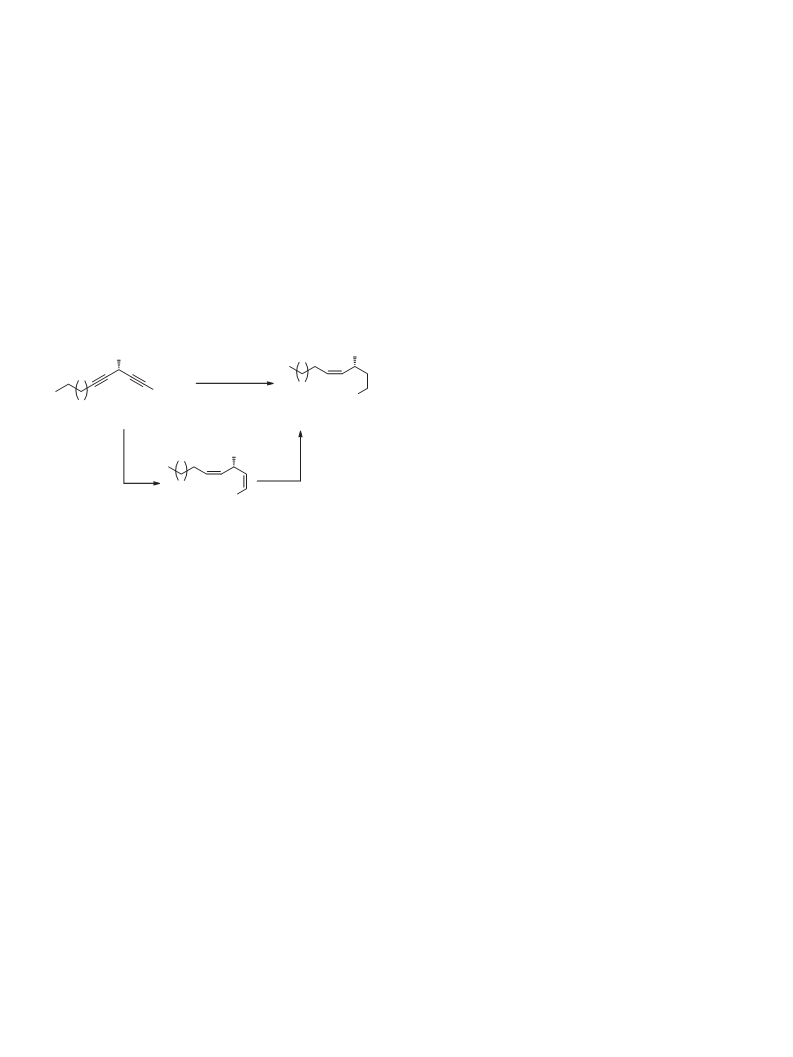
The selective reduction of two alkyne bonds of (S)-5 (Scheme 2)
is particularly noteworthy. Initially, we intended to convert (S)-5
into (R)-6 in two steps; the partial reduction of the carbon–carbon
2 4
Na SO , and concentrated under reduced pressure. The residue
25
triple bonds to two cis-double bonds followed by selective reduc-
tion of the conjugate double bond.26 However, treatment of diynol
ester (S)-5 with hydrogen gas in the presence of Lindlar’s catalyst
did not give the desired dienol ester (S)-7.25b Fortunately, semi-
hydrogenating (S)-5, catalyzed by Brown’s P2-Ni catalyst, directly
afforded enol ester (R)-6 in 83% yield.2 To the best of our knowl-
edge, this is the first synthesis of enol ester via selective and partial
reduction from diynol ester.
was purified by silica gel chromatography (n-hexanes/ethyl acetate
10:1) to give 3 (27.26 g, 90% yield) as a colorless oil. 1H NMR
3
(300 MHz, CDCl ) d: 0.88 (t, J = 6.9 Hz, 3H), 1.28–1.39 (m, 10H),
1.46–1.55 (m, 2H), 2.17–2.24 (m, 2H), 2.28 (t, J = 5.9 Hz, 1H), 4.23–
1
3
4.26 (m, 2H); C NMR (75 MHz, CDCl
3
) d: 13.95, 18.64, 22.55,
1
28.55, 28.80, 29.02, 29.09, 31.75, 51.12, 78.27, 86.35; HRMS (ESI-
+
TOF) calcd for C11H21O [M+H] 169.1592, found 169.1589.
4
.2.2. Synthesis of undec-2-ynal 4
A solution of 3 (24.352 g, 144.95 mmol) in DCM (200 mL) was
OH
OH
cooled to 0 °C, and Dess–Martin reagent (79.90 g, 188.44 mmol)
was added slowly at 0–5 °C. The reaction mixture was maintained
Ni(OAc)2.4H2O
NaBH4
6
for 3 h at 5 °C and quenched with Na
2 2 3
S O (950 mL, 5% in saturated
COOMe
ethylenediamine
MeOOC
6
EtOH, H2, 25 o
aqueous NaHCO , 191.9 mmol) at 0 °C. The resulting mixture was
C
3
(
S)-5
(R)-6, 83% yield
stirred for another 1 h, and filtered through a Celite pad. The aque-
ous phase was extracted with ether. The combined organic phases
were washed with saturated brine solution, dried over anhydrous
Lindlar cat.
NaBH4
H2, quinoline
OH
THF/MeOH
o
2 4
Na SO , and concentrated under reduced pressure. The residue
0
-18
C
EtOH, rt
was purified by silica gel chromatography (n-hexanes/ethyl acetate
6
X
30:1) to furnish 4 (22.89 g, 95% yield) as a colorless oil. 1H NMR
MeOOC
(
300 MHz, CDCl
1.55–1.65 (m, 2H), 2.39–2.44 (m, 2H), 9.18 (t, J = 0.9 Hz, 1H);
NMR (75 MHz, CDCl ) d: 14.04, 19.11, 22.60, 27.54, 28.82, 28.95,
3
) d: 0.89 (t, J = 7.0 Hz, 3H), 1.28–1.45 (m, 10H),
13
(
S)-7
C
3
Scheme 2. The reduction of (S)-5.
2
C
9.05, 31.76, 81.70, 99.38, 177.20; HRMS (ESI-TOF) calcd for
À
11
H17O [MÀH] 165.1279, found 165.1281.
3
. Conclusion
4
.2.3. Synthesis of (S)-methyl 4-hydroxytetradeca-2,5-diynoate
Herein, Japonilure and its enantiomer, Anomala osakana phero-
(
S)-5
To a stirred solution of methyl propionate (10.089 g, 120 mmol)
in toluene (60 mL), dimethylzinc (100 mL, 1.2 M in toluene,
20 mmol) was added slowly at 25 °C. After stirring for 1.5 h at
mone, were prepared in five steps with 93% ee. The synthetic pro-
cedure is mild, concise, and highly enantioselective, involving the
asymmetric addition of methyl propionate to undec-2-ynal with
Zn-ProPhenol catalyst. This is the first synthesis of an enol ester
via a semi-hydrogenating diynol ester.
1
room temperature, a solution of (R,R)-ProPhenol (5.344 g, 8 mmol)
in toluene (10 mL) was added slowly at 0–5 °C. The resulting mix-
ture was stirred at 4 °C for 1 h and aldehyde 4 (6.651 g, 40 mmol)
was added via syringe at a slow rate. The reaction solution was
stirred for another 24 h at 4 °C, and then quenched with water
(20 mL) at 0 °C. The mixture was stirred for 1 h and filtered
through a Celite pad. The aqueous phase was extracted with ether.
The combined organic phases were washed with a saturated brine
solution, dried over anhydrous Na SO , and concentrated under
reduced pressure. The residue was purified by silica gel chroma-
tography (n-hexanes/ethyl acetate 10:1) to give (S)-5 (8.01 g, 80%
yield, 95% ee) as a colorless oil. [
NMR (300 MHz, CDCl ) d: 0.88 (t, J = 6.9 Hz, 3H), 1.26–1.38 (m,
4
4
. Experimental
.1. General
All reactions were performed under an argon atmosphere. Sol-
vents were dried according to standard procedures and distilled
before use. All reagents were purchased commercially and used
2
4
without further purification, unless stated otherwise. 1H and 13
C
2
5
1
NMR spectra were recorded at 300 and 75 MHz, respectively.
High-resolution Mass spectra were recorded on an Agilent instru-
ment by the TOF MS technique. Enantiomeric excesses (ee) were
determined on an Agilent 1200 HPLC system using Chiralcel
OD-H column, and elution with n-hexanes and isopropanol; or
determined on an Agilent GC 7890B with FID using Astec Chiraldex
G-TA column. The optical rotations were mensured on a PERKIN
ELMER 341 Polarimeter.
a
]
D 3
= +4.5 (c 1.0, CHCl ); H
3
10H), 1.50–1.55 (m, 2H), 2.20–2.25 (m, 2H), 2.36 (d, J = 7.9 Hz,
1H), 3.80 (s, 3H), 5.20 (sextet, J = 7.9 Hz, 1H); C NMR (75 MHz,
CDCl ) d: 14.05, 18.65, 22.62, 28.13, 28.83, 29.01, 29.11, 31.79,
52.18, 52.88, 74.76, 75.29, 84.02, 87.67, 153.52; HRMS (ESI-TOF)
1
3
3
+
calcd for C15H23O3 [M+H] 251.1647, found 251.1642. Enantio-
meric excess was determined by HPLC with a Chiralcel OD-H col-
umn (95:5 n-hexanes: isopropanol, 1.0 mL/min, 220 nm); minor
(R)-enantiomer t = 9.64 min, major (S)-enantiomer t = 10.36 min.
4
.2. Synthesis of the Japonilure and its enantiomer (Anomala
r
r
osakana pheromone)
4
.2.4. Synthesis of (R)-methyl 4-hydroxytetradeca-2,5-diynoate
(R)-5
According to the similar procedure described above, aldehyde 4
(6.651 g, 40 mmol) was converted into (R)-5 (8.09 g, 81% yield, 93%
4
.2.1. Synthesis of undec-2-yn-1-ol 3
A solution of propargyl alcohol 2 (21.3 mL, 180 mmol), HMPA
(
187.9 mL, 180 mmol), and THF (200 mL) was cooled to À78 °C,

 Xu, Hao
Xu, Hao
 Li, Shuo-Ning
Li, Shuo-Ning
 Yang, Yan-Qing
Yang, Yan-Qing
 Zhou, Yun
Zhou, Yun
 Yang, Qian-Zhen
Yang, Qian-Zhen
 Bian, Qing-Hua
Bian, Qing-Hua
 Zhong, Jiang-Chun
Zhong, Jiang-Chun
 Wang, Min
Wang, Min