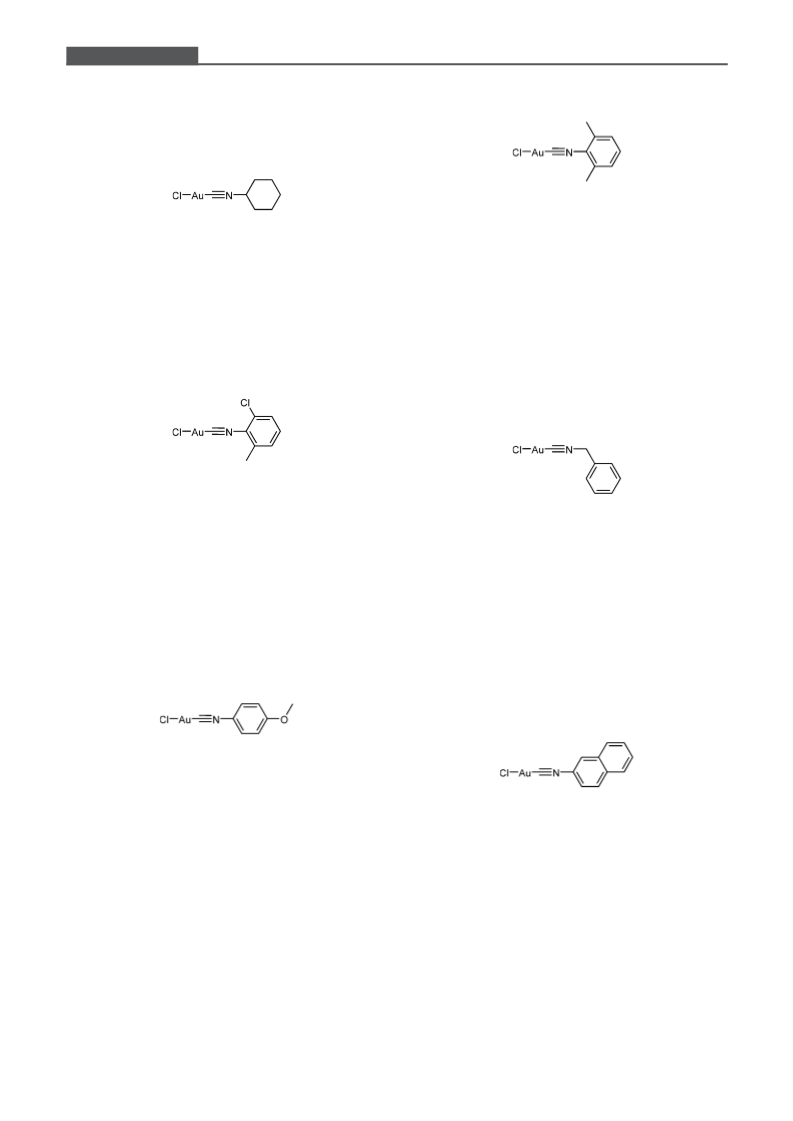
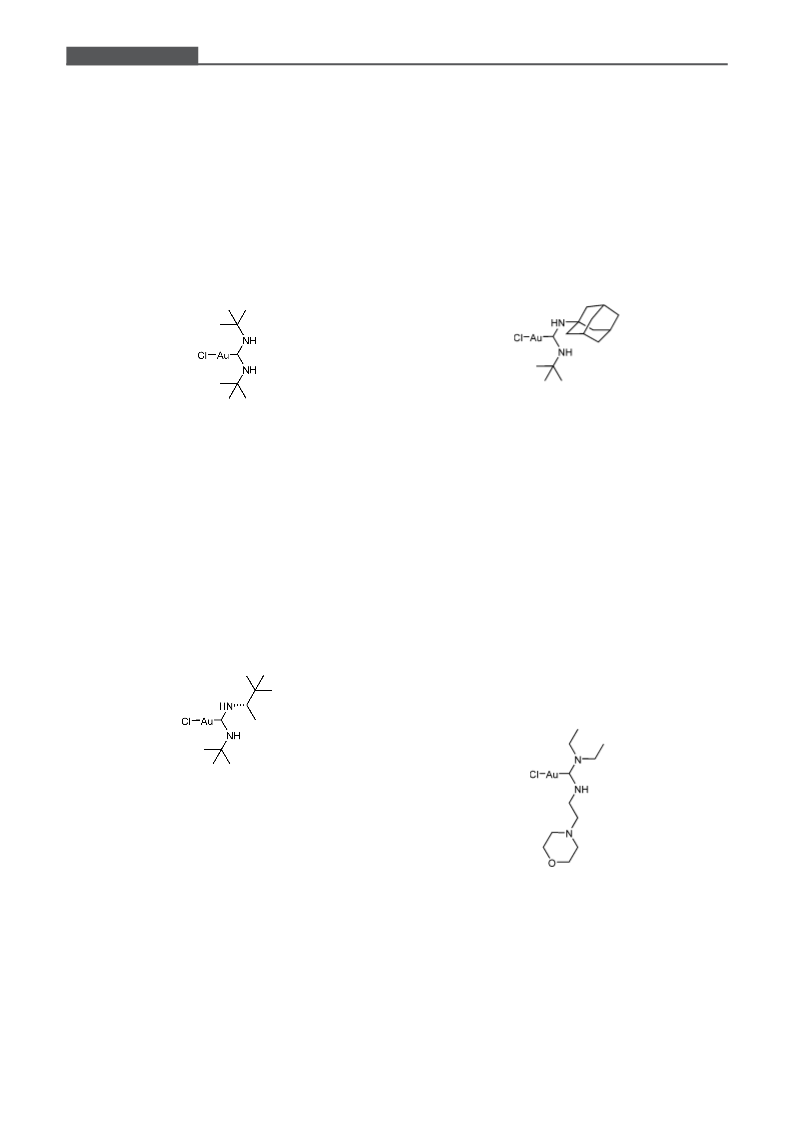
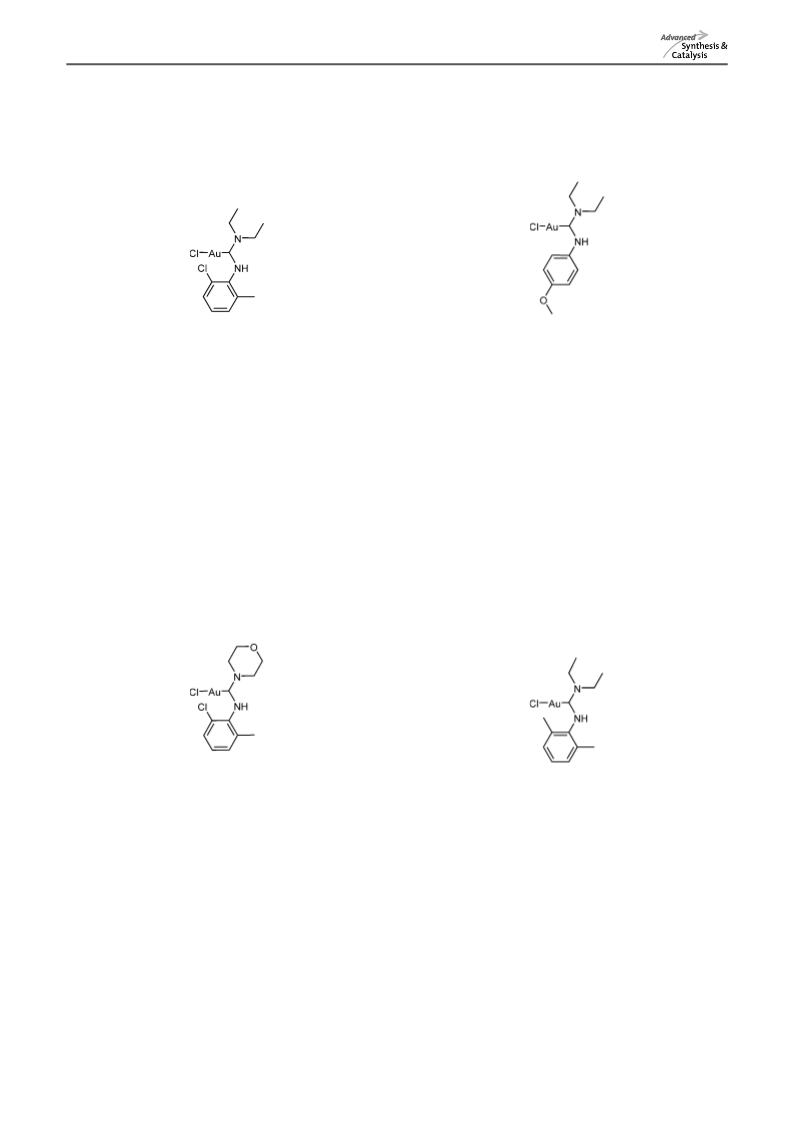
New and Easily Accessible Nitrogen Acyclic Gold(I) Carbenes
˜
938C. IR (KBr): n=3283, 2963, 2949, 2851, 2811, 1566, 1448,
1431, 1380, 1358, 1361, 1070, 1007, 867 cmÀ1 1H NMR
;
(CDCl3, 300 MHz): d=1.22 (t, J=7.3 Hz, 3H), 1.27 (t, J=
7.1 Hz, 3H), 2.51–2.57 (m, 4H), 2.64 (t, J=5.7 Hz, 2H), 3.31
(q, J=7.3 Hz, 2H), 3.71 (t, J=7.3 Hz, 4H), 3.85 (dt, J=
6.0 Hz, J=5.2 Hz, 2H), 3.90 (q, J=7.1 Hz, 2H), 6.62 (bs,
1H); 13C NMR (CDCl3, 75 MHz): d=11.9 (q), 14.7 (q), 41.4
(t), 45.2 (t), 53.1 (t, 2C), 53.6 (t), 57.3 (t), 66.8 (t, 2C), 189.9
(s); MS [FAB (+)]: m/z=446 [M+H]+ (56), 408 (88), 391
(20), 329 (24), 308 (46), 307 (100); HR-MS [FAB (+)]
C11H23AuClN3O (445.74): m/z=446.1200, calcd. for
[C11H24Au35ClN3O]+: 446.1274.
1
1630, 1543, 1451, 1379, 1354, 1092 cmÀ1; H NMR (CDCl3,
300 MHz): d=1.18 (t, J=7.3 Hz, 3H), 1.26 (t, J=7.1 Hz,
3H), 1.68–1.70 (m, 6H), 2.12–2.17 (m, 3H), 2.26–2.29 (m,
6H), 3.28 (q, J=7.3 Hz, 2H), 3.95 (q, J=7.1 Hz, 2H), 5.62
(bs, 1H); 13C NMR (CDCl3, 75 MHz): d=11.7 (q), 14.6 (q),
29.6 (d, 3C), 35.9 (t, 3C), 40.3 (t), 44.8 (t, 3C), 54.4 (s), 55.2
(t), 187.9 (s); MS [FAB (+)]: m/z=467 [M+H]+ (5), 432
(31), 431 (100), 430 (28), 429 (41); HR-MS [FAB (+)]
[(tert-Butylamino)(2-morpholinoethylamino)methyl-
idene]gold(I) Chloride (2h)
C15H26AuClN2
(466.80):
m/z=466.1448,
calcd.
for
[C15H26Au35ClN2]+: 466.1450.
[(1-Adamantylamino)(morpholino)methylidene]-
gold(I) Chloride (2j)
[(tert-Butylamino)(2-morpholinoethylamino)methylidene]-
gold(I) chloride (2h) was prepared according to GP 2 where
isocyanogold(I) chloride (1j) (100 mg, 268 mmol) and tert-bu-
tylamine (5 mL) as solvent were employed. Addition of sa-
turated NH4Cl solution, extraction with dichloromethane
and filtration (celite, silica gel and Na2SO4) furnished 2h as
a colourless solid; yield: 99.0 mg (222 mmol, 83%); mp 40–
[(1-Adamantylamino)(morpholino)methylidene]gold(I)
chloride (2j) was prepared according to GP 2 where isocya-
nogold(I) chloride (1d) (100 mg, 254 mmol) and morpholine
(44.3 mg, 508 mmol) were employed. Addition of saturated
NH4Cl solution, extraction with dichloromethane and filtra-
tion (celite, silica gel and Na2SO4) furnished 2j as a colour-
less solid; yield: 115 mg (239 mmol, 94%); mp 224–2268C
˜
428C. IR (KBr): n=3284, 3073, 2963, 2931, 2819, 1630, 1559,
1
1115 cmÀ1; H NMR (CDCl3, 500 MHz): rotamer A: d=1.61
(s, 9H), 2.48–2.67 (m, 6H), 3.13–3.16 (m, 1H), 3.69–3.83 (m,
5H), 6.95–7.03 (m, 2H), rotamer B: d=1.57 (s, 9H), 2.48–
2.67 (m, 6H), 3.13–3.16 (m, 1H), 3.69–3.83 (m, 5H), 6.31–
6.36 (m, 2H), rotamer C: d=1.41 (s, 9H), 2.48–2.67 (m,
6H), 3.13–3.16 (m, 1H), 3.69–3.83 (m, 5H), 7.11–7.18 (m,
2H); 13C NMR (CDCl3, 125 MHz): rotamer A: d=31.5 (q,
3C), 45.8 (s), 53.5 (t, 2C), 54.1 (t), 56.7 (t), 66.4 (t, 2C),
188.5 (s), rotamer B: d=31.5 (q, 3C), 38.7 (t), 46.1 (s), 53.1
(t, 2C), 53.7 (t), 67.0 (t, 2C), 192.3 (s), rotamer C: d=29.1
(q, 3C), 53.2 (t, 2C), 53.8 (t), 57.8 (t), 66.3 (t, 2C), 188.3 (s),
one quarternary carbon atom not detected; MS [FAB (+)]:
m/z=446 [M+H]+ (76), 444 (24), 408 (100); HR-MS [FAB
(+)] C11H23AuClN3O (445.74): m/z=446.1212, calcd. for
[C11H24Au35ClN3O]+: 446.1273.
˜
(decomp.). IR (KBr): n=3340, 2906, 2848, 1547, 1421, 1367,
1305, 1233, 1118, 1027, 871 cmÀ1 1H NMR (CDCl3,
;
500 MHz): d=1.70–1.73 (m, 6H), 2.16–2.20 (m, 3H), 2.29–
2.30 (m, 6H), 3.31 (bs, 2H), 3.75 (bs, 4H), 4.28 (bs, 2H),
5.80 (bs, 1H); 13C NMR (CDCl3, 125 MHz): d=29.7 (t, 3C),
35.9 (d, 3C), 43.3 (t), 44.7 (t, 3C), 55.0 (s), 58.1 (t), 65.2 (t),
67.1 (t), 190.2 (s); MS [FAB (+)]: m/z=481 [M+H]+ (7),
445 (100), 444 (19), 443 (29); HR-MS [FAB (+)]
C15H24AuClN2O (480.78): m/z=445.1500, calcd. for
[C15H24AuN2O]+: 445.1554.
[(1-Adamantylamino)(diethylamino)methylidene]-
gold(I) Chloride (2i)
[(Diethylamino)(pentylamino)methylidene]gold(I)
Chloride (2k)
[(1-Adamantylamino)(diethylamino)methylidene]gold(I)
chloride (2i) was prepared according to GP 2 where isocya-
nogold(I) chloride (1d) (100 mg, 254 mmol) and diethyl-
[(Diethylamino)(pentylamino)methylidene]gold(I) chloride
(2k) was prepared according to GP 2 where isocyanogold(I)
chloride (1b) (100 mg, 303 mmol) and diethylamine (44.4 mg,
607 mmol) were employed. Evaporation of the solvent af-
forded 2k as a colourless oil; yield: 122 mg (303 mmol,
ACHTUNGTRENNUNGamine (37.2 mg, 508 mmol) were employed. Addition of sa-
turated NH4Cl solution, extraction with dichloromethane
and filtration (celite, silica gel and Na2SO4) furnished 2i as a
colourless solid; yield: 115 mg (246 mmol, 97%); mp 175–
1778C (decomp.). Crystals were suitable for X-ray crystal
˜
>99%). IR (film): n=3302, 2957, 2930, 2870, 1561, 1429,
1381, 1349, 1299, 1201, 1077, 676 cmÀ1 1H NMR (CDCl3,
;
300 MHz): d=0.84 (t, J=6.9 Hz, 3H), 1.13–1.29 (m, 10H),
1.56–1.65 (m, 2H), 3.25 (q, J=7.3 Hz, 2H), 3.70 (q, J=
structure analysis.[20] IR (KBr): n=3386, 2973, 2910, 2848,
˜
Adv. Synth. Catal. 2010, 352, 1315 – 1337
ꢂ 2010 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
1331

 Hashmi, A. Stephen K.
Hashmi, A. Stephen K.
 Hengst, Tobias
Hengst, Tobias
 Lothschuetz, Christian
Lothschuetz, Christian
 Rominger, Frank
Rominger, Frank