
Macromolecules p. 6402 - 6410 (2005)
Update date:2022-08-17
Topics:
 Demirel, A. Levent
Demirel, A. Levent
 Yurteri, Seda
Yurteri, Seda
 Cianga, Ioan
Cianga, Ioan
 Yagci, Yusuf
Yagci, Yusuf
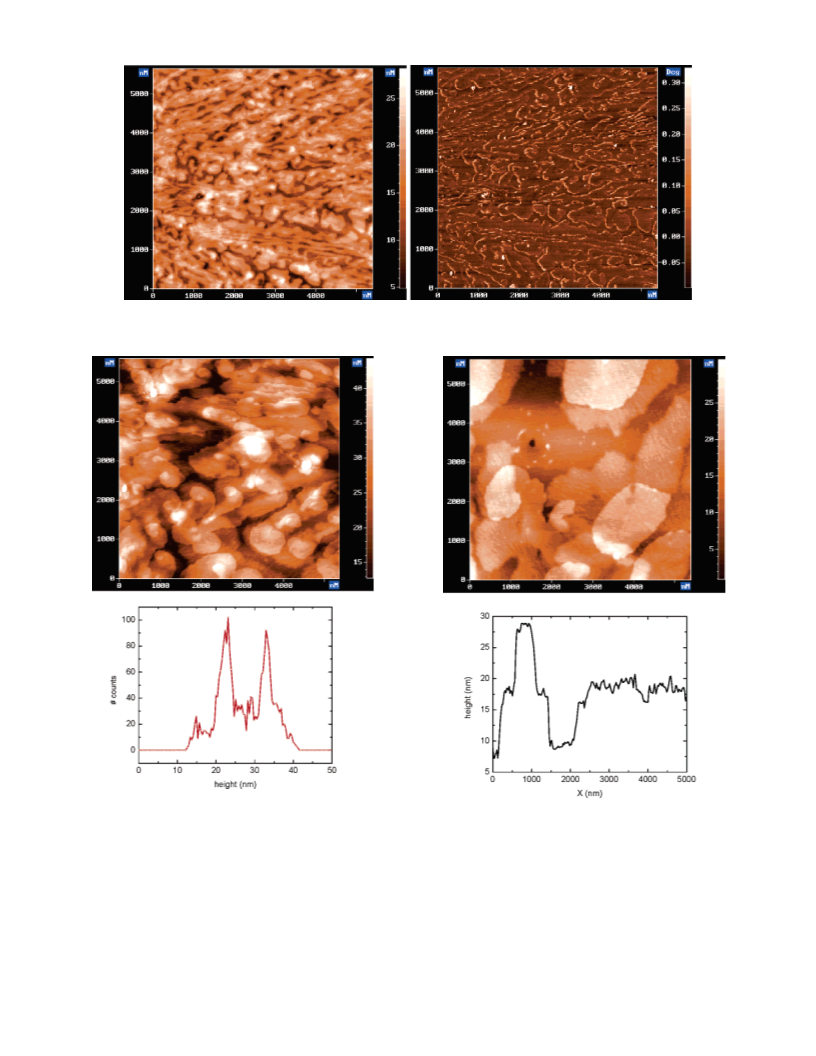
Poly(ε-caprolactone) (PCL)-based macromonomers and the corresponding substituted polyphenylene polymers have been synthesized in various chemical structures. The effect of chemical structure and the crystallization of PCL on the resulting morphology in thin films have been investigated. PCL macromonomers containing the central 2,5-dibromo-1,4-phenylene moiety were synthesized by ring-opening polymerization (ROP). Poly(phenylene) polymers were then synthesized by cross-coupling of the PCL macromonomers in Yamamato polycondensation reactions. Thin films of macromonomers and polymers were prepared by spin-coating on silicon substrates, and the resulting morphology in thin films was characterized by atomic force microscopy (AFM). Substitution of semicrystalline PCL side chains to the rigid poly(phenylene) backbone induced layered morphology in thin films. Our results indicate that increasing backbone rigidity causes grafted PCL side blocks to crystallize in better-defined layered structures parallel to the underlying substrate. Such layering was not observed when polystyrene (PSt) or poly(2-methyloxazoline) (POx) polymers were grafted to the rigid poly(phenylene) backbone. Hindering PCL crystallization by attaching PSt or POx to the end of PCL also prevented the formation of layered structures.
View More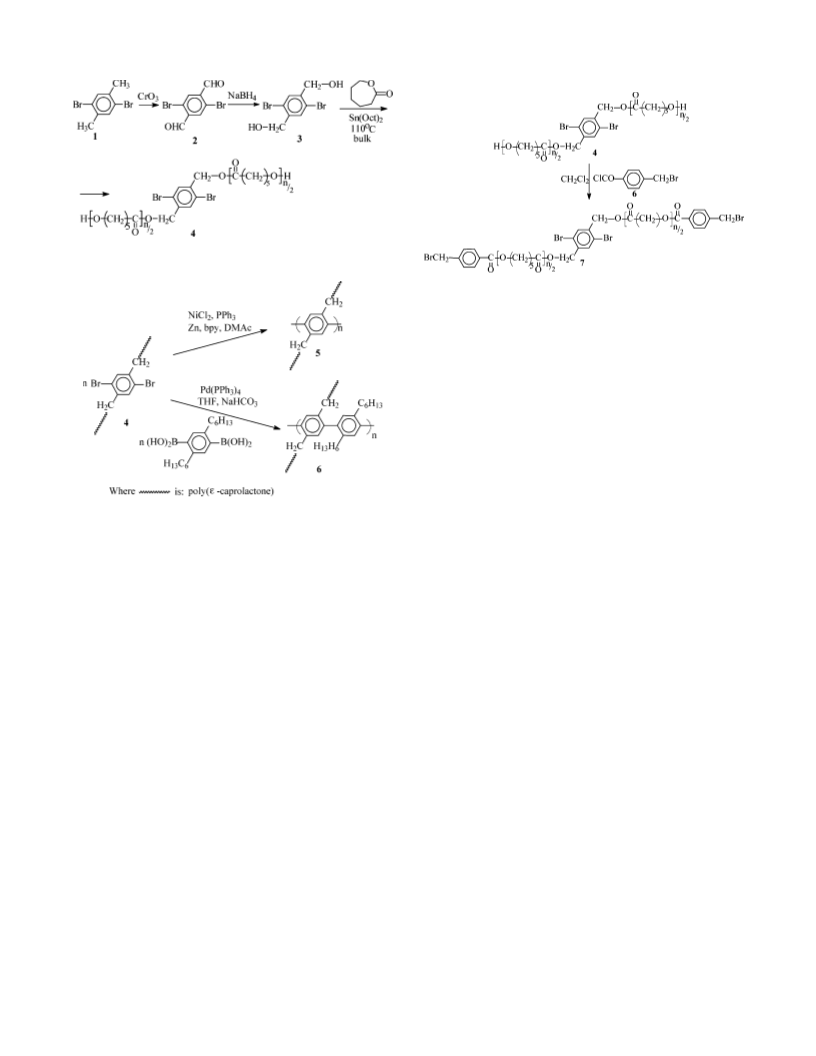





Jiaxing Trustworthy Import And Export Co.,Ltd
Contact:+86-573-82030555
Address:Room 1202, Unit B, Charming plaza,No.1558 East Zhongshan Road , Jiaxing City, Zhejiang Province, China.
YingYing Pharmaceutical Co.,Ltd
Contact:86-18854126208
Address:55#, yingxiongshan road
Contact:0833-5590788/5590338/5590055
Address:Victory in the town of Red Star Village,Mount Emei City,industrial concentration area storage processing logistics parkpark
Xian Changyue Biological Technology Co., Ltd.
website:https://www.xachangyue.com/
Contact:+86-029-62886900
Address:Keji Road NO.70
Dalian Synco Chemical Co., Ltd.
Contact:+86-411-83635150
Address:Rm 1004, 24, Tangshan Street, Dalian
Doi:10.1016/S0960-894X(01)00680-1
(2002)Doi:10.1039/b302552g
(2003)Doi:10.1021/jo01146a023
(1951)Doi:10.1021/ja305552u
(2012)Doi:10.1039/b605362a
(2006)Doi:10.1016/j.carres.2017.07.007
(2017)