2
68
steroids 7 1 ( 2 0 0 6 ) 266–272
ı = 0.80 (s, 3H, 18-H3), 0.82 (s, 3H, 19-H3), 1.98 (s, 3H, CH3CO),
.85 (dd, J = 12.8 Hz and 11.8 Hz, 7␣-H), 4.99 (m, 1H, 3␣-H); 13C
(CDCl3, 300 MHz) ı = 0.85 (s, 3H, 18-H3), 1.00 (s, 3H, 19-H3), 2.84
(dd, J = 12.3 Hz and 12.6 Hz, 1H, 7␣-H), 2.88 (d, J = 15.9, 1H, 4␣-
H); 13C NMR (CDCl3, 75 MHz) ı = 82.62 (C5), 210.15 and 210.55
2
NMR (CDCl3, 75 MHz) ı = 70.58 (C3), 80.06 (C5), 171.17 (CH3COO),
+
+
2
11.71 (C6), 220.19 (C17); MS [m/z (%)] 362 (30) M , 302 (81), 220
(C3 and C6), 219.82 (C17); MS [m/z (%)] 318 (39) M , 275 (82), 220
(
68), 205 (61), 110 (78), 95 (71), 81 (64), 43 (100).
(97), 101 (56), 85 (87), 67 (65), 55 (77), 41 (100).
2
.2.2. 5˛-Hydroxy-6-oxoandrostane-3ˇ-yl acetate (11)
2.2.8. 5˛-Hydroxyandrostane-6,17-dione (17)
Mp (diethyl ether) 236–238 C; lit. 236–238 C [51]. IR (cm )
3431.71, 1737.55, 1697.05, 1247.72; 1H NMR (CDCl3, 300 MHz)
ı = 0.78 (s, 3H, 18-H3), 0.82 (s, 3H, 19-H3), 2.84 (dd, J = 12.2 Hz
and 12.4 Hz, 1H, 7␣-H); 13C NMR (CDCl3, 75 MHz) ı = 79.03 (C5),
◦
◦
◦
◦
−1
Mp (diethyl ether–petroleum ether) 220–221 C; lit. 220–221 C
48] IR (cm 1) 3411.46, 1728.87, 1700.91, 1365.35, 1272.79,
−
[
1
1240.00; H NMR (CDCl3, 300 MHz) ı = 0.65 (s, 3H, 18-H3), 0.79 (s,
3H, 19-H3), 1.99 (s, 3H, CH3CO), 2.76 (dd, J = 12.5 Hz and 12.8 Hz,
7
␣-H), 5.01 (m, 1H, 3␣-H); 13C NMR (CDCl3, 75 MHz) ı = 70.62
+
212.29 (C6), 220.27 (C17); MS [m/z (%)] 304 (22) M , 273 (41), 233
(
(
(
C3), 80.19 (C5), 171.00 (CH3COO), 212.32 (C6); MS [m/z (%)] 348
24) M , 288 (92), 206 (74), 135 (89), 109 (95), 95 (100), 81 (77), 43
94).
(100), 220 (42), 205 (71), 67 (42), 57 (62), 41 (78).
+
5
3
.
Conversion of ꢀ -steroids to the
2
.2.3. 5˛-Hydroxy-6,20-dioxopregnane-3ˇ-yl acetate (12)
corresponding 5,6-epoxides using the
KMnO4/Fe(ClO4)3·nH2O system, in the presence
of NaH2PO4·3H2O
◦
◦
−1
Mp (diethyl ether) 220–223 C; lit. 222.5–224 C [47]. IR (cm )
3
3
394.10, 1728.87, 1708.62, 1362.46, 1240.97; 1H NMR (CDCl3,
00 MHz) ı = 0.57 (s, 3H, 18-H3), 0.79 (s, 3H, 19-H3), 2.00 (s, 3H,
CH3CO), 2.10 (s, 3H, 21-H3), 2.75 (dd, J = 12.5 Hz and 12.8 Hz, 7␣-
H), 5.01 (m, 1H, 3␣-H); 13C NMR (CDCl3, 75 MHz) ı = 70.51 (C3),
20-Oxo-pregn-5-ene-3-yl acetate 3 (358.51 mg; 1 mmol) was
dissolved in dichloromethane (9 ml) at room temperature,
in a reaction flask. A mixture of KMnO4 (2 g; 12.6 mmol),
Fe(ClO4)3·nH2O (0.5 g; 1.96 mmol) and NaH2PO4·3H2O (0.5 g,
3.20 mmol) was ground to a fine powder. Water (150 l) was
added and the final mixture was transferred to the reaction
flask, followed by the addition of t-butyl alcohol (0.39 g; 0.5 ml).
After 1 h at room temperature, TLC control showed that the
reaction was complete and the final product was separated
from the inorganic residue by addition of diethyl ether to the
reaction flask which was allowed to stay under magnetic stir-
ring for a few minutes. The mixture was then filtrated through
a celite pad and the solid residue thoroughly washed with
hot ether (total volume of 150 ml). The filtrate was washed
with water (30 ml) and dried over anhydrous sodium sulphate.
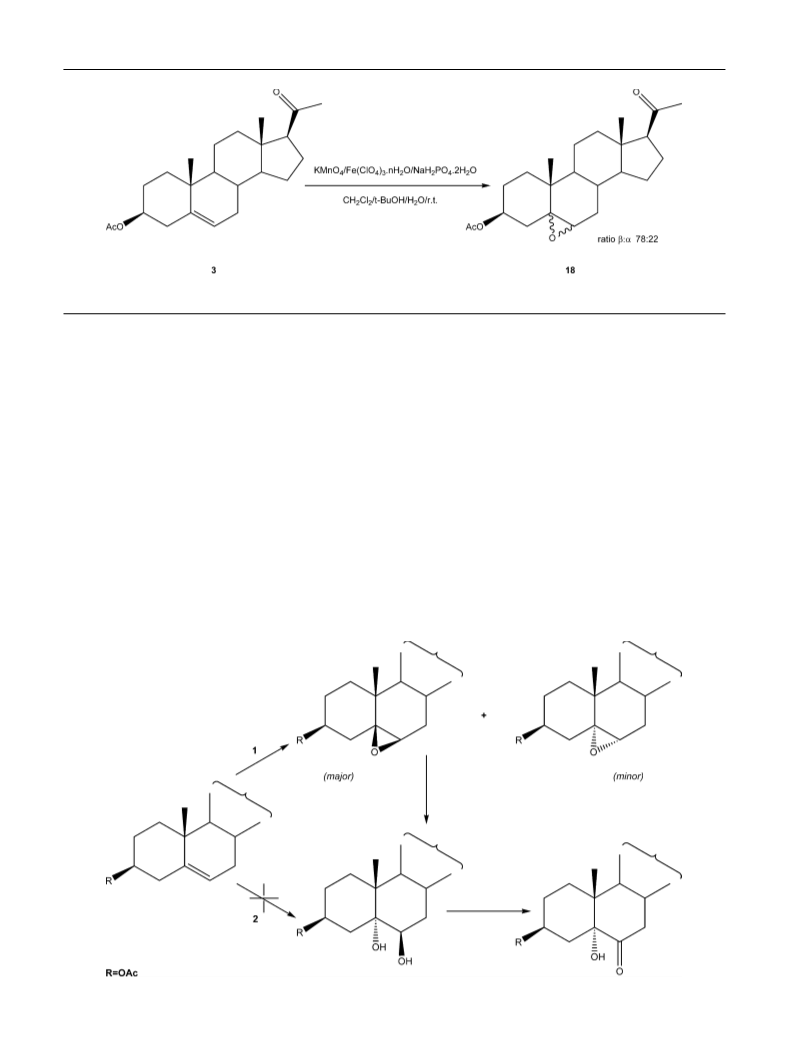
Evaporation of the solvent under vacuum afforded 18 (320 mg,
ratio /␣ 78:22 as calculated by integration of the 6-H signals
in crude samples).
8
0.16 (C5), 171.03 (CH3COO), 209.28 (C20), 211.76 (C6); MS [m/z
+
(
%)] 390 (6) M , 330 (47), 248 (24), 110 (27), 95 (23), 81 (22), 55 (18),
4
3 (100).
2
.2.4. 3ˇ-Chloro-5˛-hydroxycholestan-6-one (13)
◦
◦
Mp (acetone–petroleum ether) 181–183 C; lit. 180–186 C [11].
IR (cm 1) 3455.81, 1706.69, 1465.63, 1377.89; 1H NMR (CDCl3,
00 MHz) ı = 0.64 (s, 3H, 18-H3), 0.84 (s, 3H, 19-H3), 0.86 (d,
J = 6.5 Hz, 6H, 26-H3 and 27-H3), 0.91 (d, J = 6.5 Hz, 3H, 21-H3),
.69 (dd, J = 12.0 Hz and J = 13.0 Hz, 1H, 7␣-H), 4.21 (m, 1H, 3␣-
H); 13C NMR (CDCl3, 75 MHz) ı = 41.83 (C7), 69.80 (C3), 80.74 (C5),
11.48 (C6).
−
3
2
2
2
.2.5. 5˛-Hydroxy-6-oxocholestane-3ˇ-yl benzoate (14)
◦
◦
−1
Mp (dibutyl ether) 228–230 C; lit. 230–231 C [49]. IR (cm )
3
3
1
1
414.35, 1711.51, 1275.68; 1H NMR (CDCl3, 300 MHz) ı = 0.65 (s,
H, 18-H3), 0.87 (d, J = 6.1 Hz, 6H, 26-H3 and 27-H3), 0.88 (s, 3H,
9-H3), 0.92 (d, J = 6.3 Hz, 3H, 21-H3), 2.77 (dd, J = 12.6 Hz and
3.1.
5ˇ,6ˇ-Epoxy-20-oxopregnane-3ˇ-yl acetate (18)
ꢀ
2.7 Hz, 1H, 7␣-H), 5.31 (m, 1H, 3␣-H), 7.42 (t, J = 7.3 Hz, 2H, 3 -H
ꢀ
ꢀ
ꢀ
◦
◦
and 5 -H), 7.55 (t, J = 7.2 Hz, 1H, 4 -H), 8.01 (d, J = 7.1 Hz, 2H, 2 -
H and 6 -H); 13C NMR (CDCl3, 75 MHz) ı = 41.76 (C7), 71.23 (C3),
Mp (methanol) 129–132 C; lit. 129–131 C [52]. Spectroscopic
data according to literature [40].
ꢀ
8
0.53 (C5), 128.30 (C3ꢀ and C5ꢀ ), 129.57 (C2ꢀ and C6ꢀ ), 130.47 (C1ꢀ ),
1
32.91 (C4ꢀ ), 166.31 (C6H5CO), 212.20 (C6); MS [m/z (%)] 522 (2)
M , 400 (72), 318 (16), 122 (28), 105 (100), 77 (41), 57 (32), 43 (37).
+
4.
Results and discussion
5
2
.2.6. 5˛-Hydroxy-6-oxocholestane-3ˇ-yl acetate (15)
By using the KMnO4/Fe(ClO4)3·nH2O system on our ꢀ -steroids
(Scheme 1), we were able to achieve direct synthesis of the
corresponding ␣-ketols in high yields (Table 1, entries 1–7).
The protecting groups for the 3-hydroxy group (acetate, ben-
zoate) were resistant to these reaction conditions. However,
the tetrahydropyranyl group was unstable and the 3-keto
derivative was obtained (Table 1, entry 7; Scheme 1). The 3-
chloro group also resisted to these reaction conditions.
◦
◦
−1
)
Mp (methanol) 222–225 C; lit. 226.5–228 C [47]. IR (cm
394.10, 1733.69, 1713.44, 1365.35, 1280.50, 1243.86; 1H NMR
3
(
(
CDCl3, 300 MHz) ı = 0.62 (s, 3H, 18-H3), 0.79 (s, 3H, 19-H3), 0.84
d, J = 6.2 Hz, 6H, 26-H3 and 27-H3), 0.88 (d, J = 6.5 Hz, 3H, 21-H3),
1
4
.99 (s, 3H, CH3CO), 2.71 (dd, J = 12.5 Hz and 12.6 Hz, 1H, 7␣-H),
.99 (m, 1H, 3␣-H); 13C NMR (CDCl3, 75 MHz) ı = 70.66 (C3), 80.25
+
(
(
C5), 171.02 (CH3CO), 212.40 (C6); MS [m/z (%)] 460 (6) M , 400
100), 318 (21), 110 (42), 93 (25), 81 (26), 55 (22), 43 (54).
On the reaction with substrate 6, a mixture of the 5,6-
epoxide (ratio /␣ 80:20), the corresponding trans-diol and
the ␣-ketol was identified by 1H NMR, after 2 h. When the
KMnO4/Fe(ClO4)3·nH2O system was applied to the steroidal
5,6-epoxide 8 (Table 1, entry 8; Scheme 2), the corresponding
2
.2.7. 5˛-Hydroxyandrostane-3,6,17-trione (16)
◦
◦
Mp (ethyl acetate–hexane) 253–255 C; lit. 254–256 C [50]. IR
(
cm 1) 3435.99, 1737.55, 1715.37, 1703.80, 1243.86; 1H NMR
−

 Salvador, Jorge A.R.
Salvador, Jorge A.R.
 Moreira, Vania M.
Moreira, Vania M.
 Hanson, James R.
Hanson, James R.
 Carvalho, Rui A.
Carvalho, Rui A.