2898
K. R. Guertin et al. / Bioorg. Med. Chem. Lett. 13 (2003) 2895–2898
against PTP1B in vitro under the low DTT conditions
(Table 1). The compound has a favorable t1/2 and
excellent oral bioavailability. The large steady state
volume of distribution (Vss) of 13 is approximately 6
times the total body water volume of the animal sug-
gesting deeptissue and cell penetration. On the other
hand, the compound suffers from high systemic clear-
ance which exceeds hepatic blood flow of the mouse.
8. Leung, K. W. K.; Posner, B. I.; Just, G. Bioorg. Med.
Chem. Lett. 1999, 9, 353.
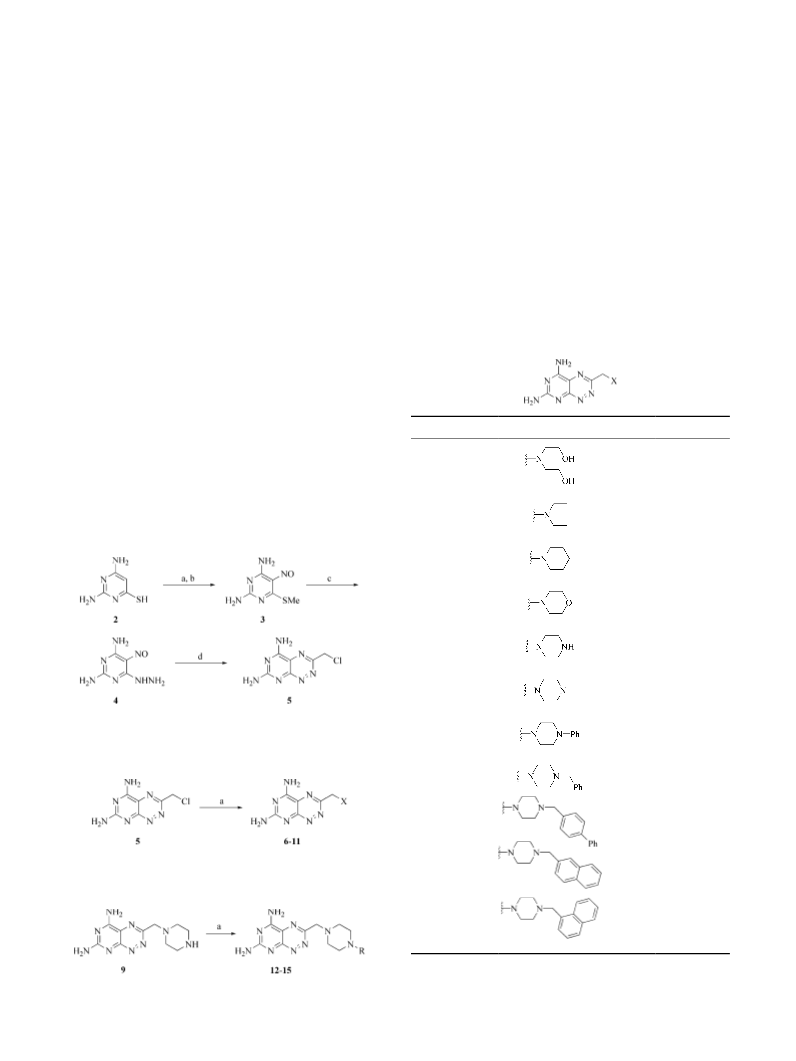
9. Satisfactory spectral data was obtained for all compounds.
Final products 6–15 were purified by C18 reversed-phase
HPLC, eluting with 0.1% TFA, H2O/CH3CN gradient, lyo-
philized and isolated as their trifluoroacetate salts.
10. Assay conditions: PTP1B (0.5–2 nM) was incubated with
compound for 15 min in buffer containing 37.5 mM MES pH
6.2, 140 mM NaCl, 2 mM EDTA, 0.05% BSA and either 300
nM or 2 mM DTT. The reaction was initiated by the addition
of 50 mM substrate [phosphorylated peptide 1146(TRDI-
(pY)E]. After 20 min at room temperature (22–25 ꢀC), the
reaction was stopped with KOH and the amount of free
phosphate measured using malachite green as previously
described (Harder, K. W.; Owen, P.; Wang, L. K.; Asbersold,
R.; Clark-Lewis, I.; Jirik, F. R. Biochem J. 1994, 298, 395). In
order to measure the PTPase activity across a panel of phos-
phatases, similar assay conditions were used as described
except 6,8-difluoro-4-methylumbelliferylphosphate (DiFMUP,
Molecular Probes) was used as substrate at the Km for each
enzyme and 37.5 mM diethylglutarate pH 6.2 was used in
place of MES. The reaction was stopped with KOH and the
fluorescence of the dephosphorylated substrate measured
(excitation: 360 nM/emission: 460 nM).
In conclusion,
a novel series of pyrimido[5,4-3]-
[1,2,4]triazine-5,7-diamine-based hypoglycemic agents
have been discovered. These compounds display non-
selective or ‘vanadate like’ PTPase inhibitory properties
in vitro and 12 and 13 were found to effectively lower
blood glucose levels in ob/ob mice upon oral adminis-
tration. From a toxicological standpoint, the lack of a
metallic component suggests that compounds of this
class may offer an advantage over the vanadium-based
anti-diabetic agents currently in clinical development.
Acknowledgements
11. (a) Shaver, A.; Ng, J. B.; Hall, D. A.; Soo Lum, B.; Posner,
B. I. Inorg. Chem. 1993, 32, 3109. (b) Huyer, G.; Liu, S.; Kelly,
J.; Moffat, J.; Payette, P.; Kennedy, B.; Tsaprailis, G.;
Gresser, M. J.; Ramachandran, C. J. Biol. Chem. 1997, 272,
843.
We thank Drs. Jeff Tilley and Nader Fotouhi for helpful
discussions and Dr. Hans Kuehnle for in vivo eval-
uation of compounds 12 and 13.
12. Upon reaction of 16 with DTT, the CH proton of the tri-
azine ring of product 17 is shifted upfield by approximately 3
ppm compared to 16, indicating a loss in aromaticity of this
ring. In DMSO-d6, two NH protons could be detected at 6.93
and 8.73 ppm. Mass spectrometry revealed a species whose
mass is 2 units higher than parent 16, corresponding to the
addition of hydrogen and consistent with the structure 17.
13. Mahadev, K.; Zilbering, A.; Zhu, L.; Goldstein, B. J . J .
Biol. Chem. 2001, 276, 21938.
14. Olivier, A. R. Unpublished results. A detailed account of
this work will be published separately.
15. (a) Fischer, E. H.; Charbonneau, H.; Tonks, N. K.
Science 1991, 253, 401. (b) Pot, D. A.; Dixon, J. E. Biochim.
Biophys. Acta 1992, 1136, 35.
References and Notes
1. Defronzo, R. A. Diabet. Rev. 1997, 5, 177.
2. Klein, R. Diabetes Care 1995, 18, 258.
3. Neel, B. G.; Tonks, N. K. Curr. Opin. Cell Biol. 1997, 9, 93.
4. (a) Goldstein, B. J. J. Cell. Biochem. 1992, 31, 33. (b)
Goldstein, B. J. Receptor 1993, 3, 1. (c) Ahmad, F.; Goldstein,
B. J. Biochim. Biophys. Acta 1995, 1248, 57.
5. Byon, J. C. H.; Kusari, A. B.; Kusari, J. Mol. Cell. Bio-
chem. 1998, 182, 101.
6. Elchebly, M.; Payette, P.; Michaliszyn, E.; Cromlish, W.;
Collins, S.; Loy, A. L.; Normandin, D.; Cheng, A.; Himms-
Hagen, J.; Chan, C. C.; Ramachandran, C.; Gresser, M. J.;
Tremblay, M.; Kennedy, B. P. Science 1999, 283, 1544.
7. (a) Huyer, G.; Liu, S.; Kelly, J. J. Biol. Chem. 1997, 272,
843. (b) Tsiani, E.; Fantus, G. Trends Endocrinol. Metab.
1997, 8, 51. (c) Posner, B. I.; Faure, R.; Burgess, J. W.; Bevan,
A. P.; Lachance, D.; Zhang-Sun, G.; Fantus, I. G.; Ng, J. B.;
Hall, D. A.; Lum, B. S. J. Biol. Chem. 1994, 269, 4596. (d)
Yale, J. F.; Lachance, D.; Bevan, A. P.; Vigeant, C.; Shaver,
A.; Posner, B. I. Diabetes 1995, 44, 1274. (e) Halberstan, M.;
Cohen, N.; Shlimovich, P.; Rosetti, L.; Shamoon, H. Diabetes
1996, 45, 659. (f) Fantus, I. G.; Tsiani, E. Mol. Cell. Biochem.
1998, 182, 109.
16. Coleman, D. L. Diabetologia 1978, 14, 141.
17. As determined by C, H, N, O, F combustion analysis, the
trifluoroacetate salt of compounds 12 and 13 contains 3 mol of
TFA per mol of parent 12 and 13. Thus a 50-mg/kg dose of 12
corresponds to 28 mg/kg of parent drug.
18. Preliminary toxicological data was obtained following the
5 day dosing study in ob/ob mice (Table 3). No significant
adverse treatment related effects were observed on organ
weights, hematological parameters or serum chemistries. A
histopathological evaluation of selected tissues was per-
formed and no treatment related microscopic changes were
observed.

 Guertin, Kevin R.
Guertin, Kevin R.
 Setti, Lina
Setti, Lina
 Qi, Lida
Qi, Lida
 Dunsdon, Rachel M.
Dunsdon, Rachel M.
 Dymock, Brian W.
Dymock, Brian W.
 Jones, Philip S.
Jones, Philip S.
 Overton, Hilary
Overton, Hilary
 Taylor, Mathew
Taylor, Mathew
 Williams, Glyn
Williams, Glyn
 Sergi, Joseph A.
Sergi, Joseph A.
 Wang, Karen
Wang, Karen
 Peng, Ying
Peng, Ying
 Renzetti, Marcia
Renzetti, Marcia
 Boyce, Rogely
Boyce, Rogely
 Falcioni, Fiorenza
Falcioni, Fiorenza
 Garippa, Ralph
Garippa, Ralph
 Olivier, Andree R.
Olivier, Andree R.