
Bulletin of the Chemical Society of Japan p. 3295 - 3300 (1988)
Update date:2022-08-11
Topics:
 Matsui, Hiroshi
Matsui, Hiroshi
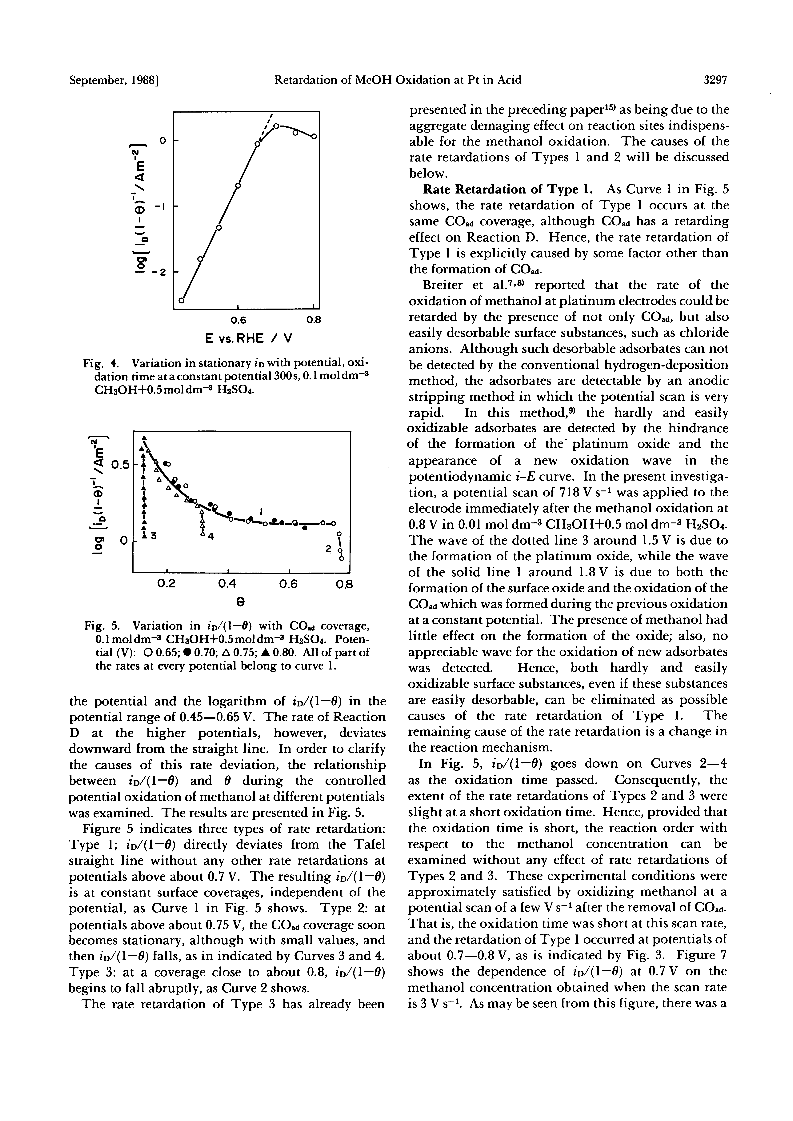
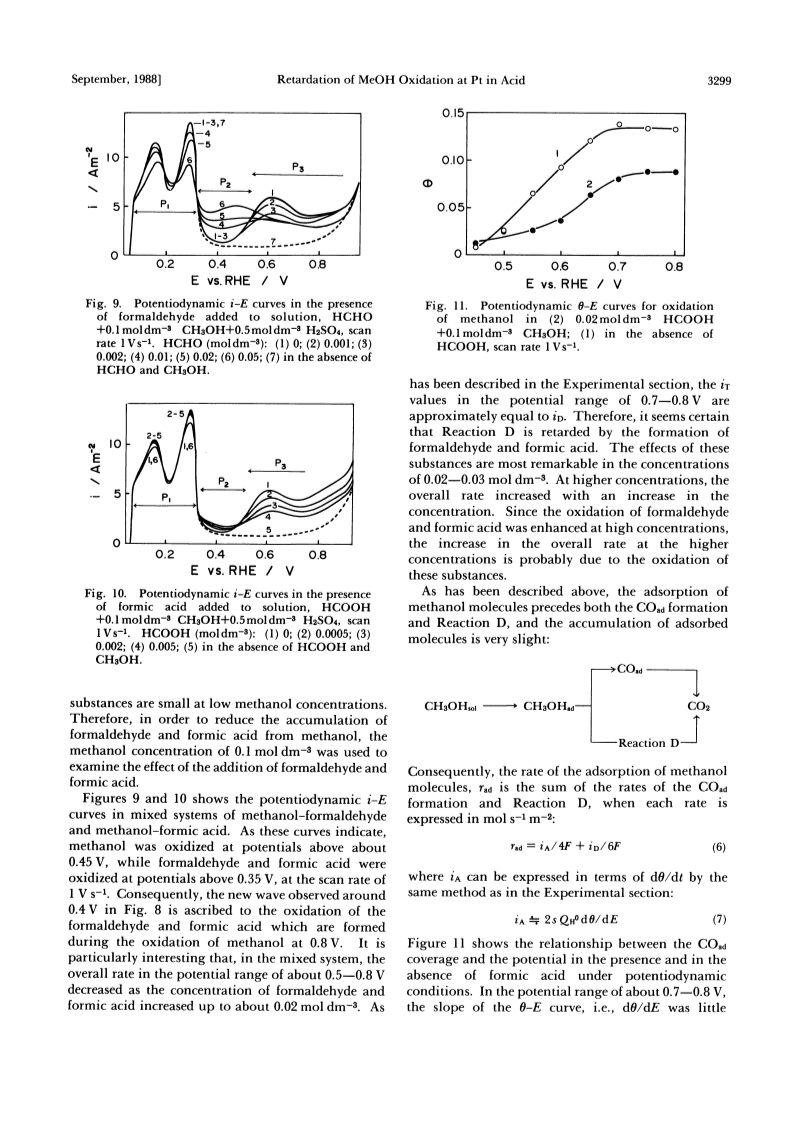
The rate retardation of the oxidation of methanol at the potential range of about 0.65-0.8 V vs. a reversible hydrogen electrode on a platinum electrode in 0.5 mol dm-3 H2SO4 was studied.The rate retardation of the overall oxidation was caused by that of the oxidation, Reaction D, not via COad.From the relationship among the rate of Reaction D, the COad coverage, and the potentials, three types of rate retardation were found out: Type 1-Reaction D is not accelerated by the potential, and the rate of the reaction is determined by the COad coverage and the methanol concentration.Type 2- the rate of Reaction D decreases at stationary COad coverages as the oxidation is prolonged.Type 3- the rate decreases at COad coverages close to the limiting value.It is proposed that Types 1 and 2 of the rate retardations take place when the adsorption of methanol molecules is rate-determining, and when the formaldehyde and formic acid formed from methanol are accumulated in the vicinity of the electrode, respectively.Type 3 of the rate retardation has been explained in a preceding paper in terms of the aggregate damaging effect of COad.
View More



He Bei Shun Er Chemical Co., LTD.
Contact:86-0311-86996932/86860168
Address:No 18,North street
Alpha(shijiazhuang) Chemical Co., Ltd
Contact:+86-311-85652680
Address:Shijiazhuang City ,Hebei Province China
Chiral Quest (Suzhou) Company Ltd
website:http://www.chiralquest.com
Contact:+86-0512-62956066
Address:B1/9, 218 Xinghu Street, Suzhou Industrial Park
Shouguang Nuomeng Chemical Co., Ltd.
Contact:+86-536-5119508/18363669993
Address:Hou Zhen Industrial Park, Shouguang, Shandong, China
Suzhou BEC Fine Chemicals Co., Ltd.
website:http://www.bek.com.cn
Contact:0512-68095917 18913193865
Address:6, Jin Shan Road, Suzhou New District, 215011 China Suzhou Nations Pharmaceutical Innovation Center Inside
Doi:10.1016/0040-4039(95)00711-K
(1995)Doi:10.1038/s41557-019-0304-z
(2019)Doi:10.1039/P29760001342
(1976)Doi:10.3762/bjoc.13.98
(2017)Doi:10.1016/S0040-4039(00)70652-6
(1989)Doi:10.1021/jo00898a038
(1975)