AMIDE AND LACTAM HYDROLYSIS OF N-(2-HYDROXYACETYL)-2-PYRROLIDONE
739
separated and washed with a saturated solution of
NaHCO3. The organic phase was then dried with
anhydrous Na2SO4. The solution was filtered and excess
toluene evaporated under reduced pressure at 608C. A
solid was obtained. mp: 94–968C; yield: 3.4 g (61%).
NMR(1H,CDCl3): d: 2.10 (m,2H); 2.50 (t,2H); 3.80
(t,2H); 4.60 (s,4H); 7.30 (m,5H). Mass spectrum: m/z: 233
(Mþ), 91 (M-142); 127 (M-106).
2 < pD < 6, KH2PO4; 6 < pD < 10, K2HPO4; and
10 < pD < 13, K3PO4. Buffer concentrations of 0.3,
0.5, 0.7, and 1.0 M and a [KCl] ¼ 1 M were used. pD
values were obtained directly from pH measurements by
using the relation10: pD ¼ pH þ 0.40. The following
values of pD were used to evaluate the equilibrium and
hydrolysis rate: 0.4, 1.7, 4.9, 6.9, 7.4, 8.3, 10.5, 11.2, 11.8,
12.4, and 13.0. Equilibrium constants were evaluated in
triplicate; a standard deviation <10% was obtained.
N-(2-Hydroxyacetyl)-2-pyrrolidone (3)
Rate constant measurements
0.23 g (0.98 mmol) of N-benzylacetil-2-pyrrolidone was
dissolved in 30 mL of ethyl acetate (HPLC grade). To this
solution 0.43 g of Pd on Alumina (5%) catalyst in 35 mL
of ethyl acetate was added. The mixture was reduced
under a hydrogen atmosphere at 35 psi during 3 h. After
the reduction, the mixture was filtered and the resultant
liquid concentrated under reduced pressure. Awhite solid
was obtained. mp: 62–638C (lit10: 64–658C); yield: 0.12 g
(80%). NMR (1H,CDCl3): d:2.10 (m,2H); 2.60 (t,2H);
3.30 (s,1H); 3.85 (t,2H); 4.70 (s,2H). NMR(1H,D2O): d:
2.14 (m,2H); 2.65 (t,2H); 3.82 (t,2H); 4.70 (s,2H). Mass
spectrum: m/z: 143 (Mþ); 86 (M-57); 113 (M-30). IR
Kinetics were measured by following the disappearance
of the exocyclic methylene NMR (1H) signal of
compound 3 at ca. 4.6 ppm. Buffer phosphate (0.7 mL)
in D2O was used to dissolve compound 3 to yield a final
concentration of 0.25 M in the NMR tube. Two kinds of
experiments were conducted. One, keeping the buffer
concentration constant at 0.5 M at pD: 7.3, 8.7, 10.8, 11.3,
12.7, and 13 and the second keeping pD constant at 8.7
and 12.7 and changing the buffer concentration to 0.3, 0.7,
and 1.0 M. In all cases, a 400 MHz NMR, JEOL Eclipse
Plus instrument, was used. Rate constants were obtained
from the slope of a plot of ln (ItꢀIinf) versus t, where I is
the NMR integral of the signal. Rate constants were
measured in triplicate. The errors between measurements
did not exceed 5%.
(KBr): 3528 cmꢀ1 (OH), 1732 cmꢀ1 (C O lactam),
—
—
1699 cmꢀ1 (C O amide). UV (H O): 226 nm.
—
—
2
Methyl ester of NBA
0.22 g (0.94 mol) of N-benzylacetyl-2-pirrolidone was
dissolved in 30 mL of methanol (HPLC grade). To this
solution 0.46 g of Pd-Alumina (5%) catalyst dissolved
in 30 mL of methanol was added. The mixture was
reduced under a H2 atmosphere at 30 psi. After 3 h,
the reaction was stopped and the mixture filtered. The
resultant liquid was concentrated under reduced
pressure at 408C. The CGMS spectrum of the liquid
showed three peaks corresponding to toluene, N-(2-
hydroxyacetyl)-2-pyrrolidone (3) and the methyl ester
of N-(a-hydroxyacetyl)-g-aminobutyric acid. Mass
spectrum: 175 (Mþ); 146 (M-29); 104 (M-71); 91
(M-84); 65 (M-110). NMR(1H,CDCl3): d: 2.00 (m,2H);
2.45 (t,2H); 3.25 (t,2H); 3.80 (s,3H); 4.16 (s,2H). This
compound was used to identify one of the hydrolysis
products of 3, the NBA (4).
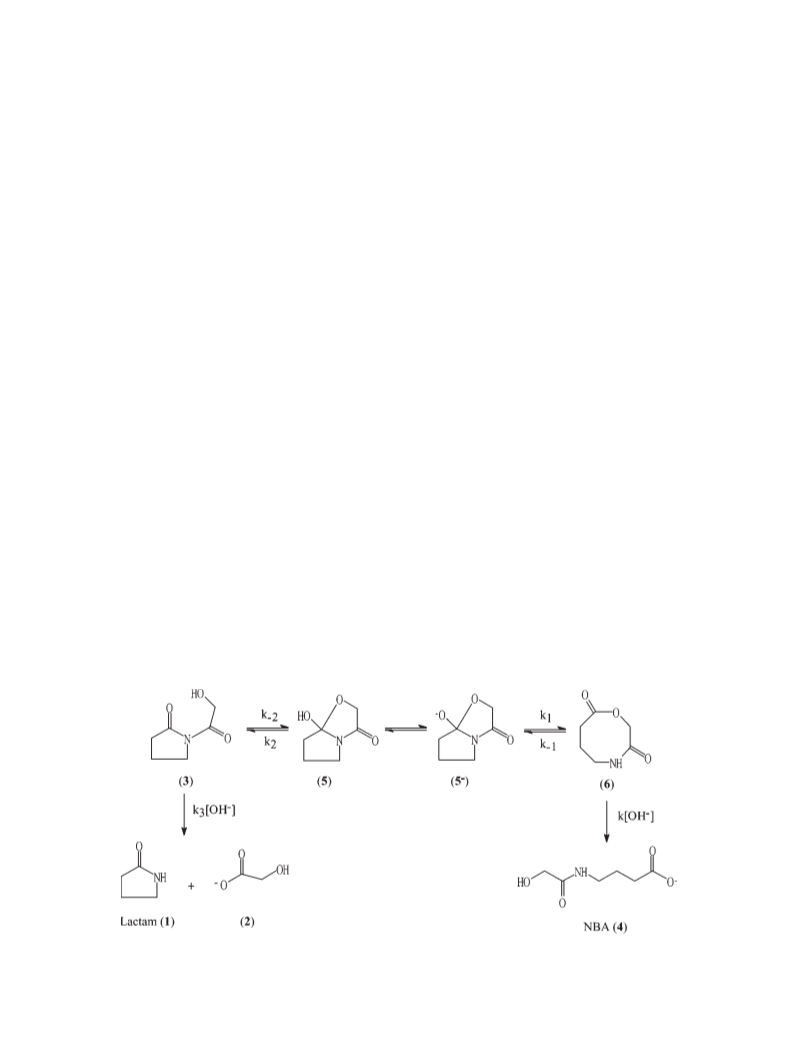
Identification of hydrolysis products
The lactam 2-pyrrolidone (1) and glycolic acid produced
during the reaction were identified by adding these
commercially available compounds to the reaction
mixture and identifying the signals whose intensity
increased. Signals of NBA (4) were identified by
comparison with the NMR signals of the methyl ester
of NBA that was synthesized in this work. Cyclol 5 and
macrocycle 6 NMR signals were identified by changing
the equilibrium toward the N-(2-hydroxyacetyl)-2-pyrro-
lidone (3) by decreasing the pH at ca. 2 of the reaction at
pH ca. 12 and observing the disappearance of these
signals. The cyclol and macrocycle signals were then
assigned by analogy with similar systems previously
identified1,2 in our laboratory. Magnetization transfer
experiments were also used to identify the corresponding
methylene signals. In Table 1, the NMR signals for the
reactants and products are shown. In Fig. 1, NMR spectra
at pD ¼ 3.0 and pD ¼ 12.7 are shown.
Sample preparation
1H NMR, was used to follow the hydrolysis of compound
3 at different pH values. The samples for each experiment
were prepared by dissolving 25 mg of compound 3 in
0.7 mL of D2O or in a solution of phosphate buffer in
D2O. NMR tubes of 5 mm were used to record the NMR
spectra. According to the required pH, the following
buffer solutions were used: 0 < pD < 2, H3PO4;
DISCUSSION
The product pair 2-pyrrolidone (lactam) (1) and glycolic
acid (2) is formed in the hydrolysis of N-(2-hydro-
xyacetyl)-2-pyrrolidone (3) via reaction of its exocyclic
Copyright # 2006 John Wiley & Sons, Ltd.
J. Phys. Org. Chem. 2006; 19: 737–743

 Borboa, Lisaedy Garcia
Borboa, Lisaedy Garcia
 Nunez, Oswaldo
Nunez, Oswaldo