1278
D.C. Menezes et al. / European Journal of Medicinal Chemistry 40 (2005) 1277–1282
activity towards C. albicans we have synthesised and char-
acterised a series of tin(IV) complexes with pyrrolidinedithio-
carbamate. Here in we describe the outcomes of our investi-
gation.
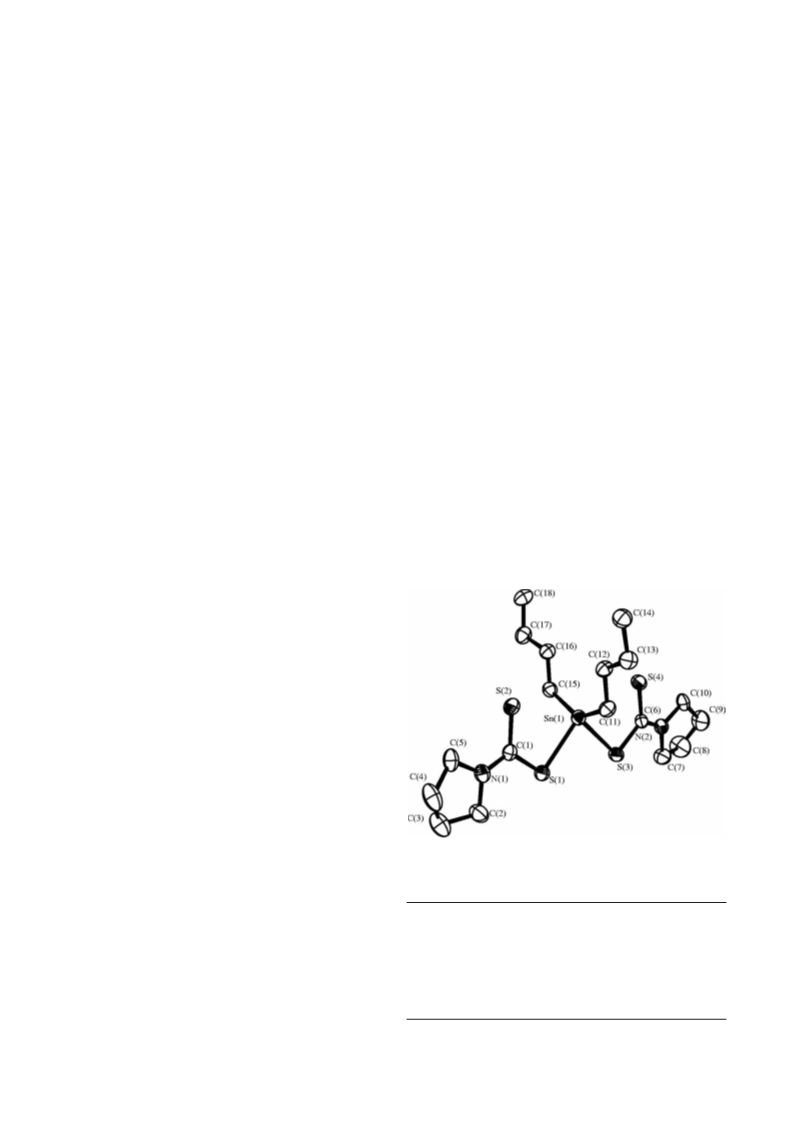
yielding an orange solid, [Sn{S2CN(CH2)4}2Cl2]. It
was re-crystallised in a mixture of hexane/THF and
dried in vacuum. Yield 80%. Elemental analysis for:
C10H16SnN2S4Cl2. Found: C, 24.72%; H, 3.30%; N, 5.65%.
Calc: C, 24.91%; H, 3.32%; N, 5.81%. M.p. (decomposi-
tion) = 200 °C. IR (cm−1, KBr): 303 (m, mSn–Cl); 315 (w, mSn–
S
); 949 (s, mC–S); 1327 (m, mC–N); 1459 (s, mC–N + mC=N). 1H
2. Experimental
NMR (d, CDCl3): 1.86 (m, 2CH2); 3.63 (t, 2NCH2).13C{1H}
NMR (d, CDCl3): 26.01 (2CH2); 55.32 (2NCH2); 202.85
(SCN). 119Sn{1H} NMR (d, CHCl3): –521.
2.1. Materials
Carbon disulphide, pyrrolidine, ammoniun hydroxide, tin
compounds, dichloromethane, diethylether and ethanol were
purchased from Merck, Aldrich, Synth and Quimex and used
with no previous treatment. Solvents such as tetrahydrofuran
(THF) and hexane, purchased from Synth. were previously
dried by standard methods.
2.4.2. [Sn{S2CN(CH2)4}2Ph2] (2)
[NH4{S2CN(CH2)4}] (0.98 g, 6 mmol) was dissolved in
20 cm3 of ethanol and added to a solution of [Sn(C6H5)2Cl2]
(1.03 g, 3 mmol) in the same solvent. The mixture was stirred
for 2 h and then the solvent was removed by filtration and a
white solid [Sn{S2CN(CH2)4}2Ph2] (2), was obtained. The
ammonium salt was washed with water at 80 °C. Yield 85%.
Elemental analysis for: C22H26SnN2S4. Found: C, 46.62%;
H, 4.54%; N, 4.84%. Calc.: C, 46.75%; H, 4.60%; N, 4.95%.
M.p.: 222.5–223.1 °C. IR (cm−1, KBr): 385 (m, mSn–S); 943
(s, mC–S); 1247 and 1328 (m, mC–N); 1495 (s, mC–N + mC=N).
1H NMR (d, CHCl3): 1.99 (m, 2CH2); 3.64 (t, 2NCH2); 7.23–
7.90 (m, 2 C6H5). 13C{1H} NMR (d, CDCl3): 26.72; 54.88;
128.10 (4J119Sn–13C = 83 Hz); 128.34; 134.30 (2J119Sn–
13C = 621 Hz); 151.62; 194.49. 119Sn{1H} NMR (d, CHCl3):
–500 (1J119Sn–13C = 811 Hz).
2.2. Instruments and techniques
Elemental analyses were performed using a Perkin–Elmer
Model PE 2400CHN. Melting points were determined with a
digital melting point from Mettler model FP90 with cell of
heating model FP82 HT and microscopy from Olympus CH-2.
IR spectra were obtained as KBr plates on a Mattson Gal-
axy model ST 3000 spectrometer in the 4000–200 cm−1 range.
NMR spectra were recorded in CDCl3 at 25 °C on a Brucker
Avance DRX 400. The values were referenced to internal
SiMe4 and SnMe4. 119Sn Mössbauer measurements were per-
formed on a conventional apparatus with the samples at liq-
uid N2 temperature and a CaSnO3 source kept at room tem-
perature.
2.4.3. [Sn{S2CN(CH2)4}Ph3] (3)
Prepared accordingly using [NH4{S2CN(CH2)4] (0.49 g,
3 mmol) and [Sn(C6H5)3Cl] (1.26 g, 3 mmol) dissolved in
20 cm3 of ethanol. Yield 89%. Elemental analysis for:
C23H23SnNS2. Found: C, 55.47%; H, 4.73%; N, 2.81%. Calc.:
C, 55.68%; H, 4.64%; N, 2.82%. M.p.: 171–171.4 °C. IR
(cm−1, KBr): 349 (m, mSn–S); 949 (s, mC–S); 1246 (m, mC–N);
1479 (s, mC–N + mC=N). 1H NMR (d, CDCl3): 1.91 (m, 2CH2);
3.67 (t, 2NCH2); 7.28–7.90 (m, 3 C6H5). 13C{1H} NMR (d,
CDCl3): 26.64; 55.05; 128.46; 129.07 (3J119Sn–
13C = 105 Hz); 136.72 (2J119Sn–13C = 276 Hz); 142.07
(1J119Sn–13C = 1737 Hz); 191.57. 119Sn{1H} NMR (d,
CHCl3): –173 (1J119Sn–13C = 1897 Hz).
2.3. Synthesis of [NH4{S2CN(CH2)4}]
To a solution of pyrrolidine (0.64 g, 9 mmol, in 20 cm3 of
diethylether), at 0 °C, was dropped carbon disulphide (0.68 g,
9 mmol). This mixture was stirred for 1 h and ammoniun
hydroxide was added. The white solid formed,
[NH4{S2CN(CH2)4}], was filtered and re-crystallised from a
mixture of water and THF. Yield 85%. Analysis for:
NH4S2C5H8N. Found: C, 36.51%; H, 7.28%; N, 8.41%. Calc:
C, 36.59%; H, 7.32%; N, 8.54%. M.p. 142–143 °C. IR (cm−1,
KBr): 998 (s, mC–S); 1240 and 1325 (m, mC–N); 1450 (s,
mC–N + mC=N) . 1H NMR (d, D2O): 1.85 (m, 2CH2); 3.58 (t,
2NCH2). 13C{1H} NMR (d, D2O): 25.01 (2CH2); 55.27
(2 NCH2); 202.80 (SCN).
2.4.4. [Sn{S2CN(CH2)4}2n-Bu2] (4)
Prepared accordingly employing [NH4{S2CN(CH2)4}]
(0.98 g, 6 mmol) and [Sn(C4H9)2Cl2] (0.91 g, 3 mmol) dis-
solved in 20 cm3 of dichloromethane. X-ray quality colour-
less crystals were obtained by cooling an ethanol solution of
the compound. Yield 75%. Elemental analysis for:
C18H34SnN2S4. Found: C, 40.99%; H, 6.38%; N, 5.28%.
Calc.: C, 41.17%; H, 6.48%; N, 5.34%. M.p.: 110–111 °C.
IR (cm−1, KBr): 347 (m, mSn–S); 949 (s, mC–S); 1010 and 1038
2.4. Syntheses of complexes
2.4.1. [Sn{S2CN(CH2)4}2Cl2] (1)
1
In a Schlenk flask, [NH4{S2CN(CH2)4}] (0.98 g, 6 mmol)
was dissolved in 50 cm3 of THF and added to a solution of
SnCl2 (0.57 g, 3 mmol) in the same solvent. The mixture was
kept stirring for 2 h in an atmosphere of nitrogen. The white
solid formed, composed of SnO and NH4Cl, was separated
by filtration from an orange solution. The solvent was removed
(s, mC=S); 1643 (m, mC–N). H NMR (d, CDCl3): 0.93 (t,
2CH2CH3); 1.34 (m, 2CH3); 1.86 (m, CH2CH2CH2); 1.97 (t,
SnCH2); 2.05 (m, 2CH2); 3.76 (t, 2NCH2). 13C{1H} NMR
(d, CDCl3): 13.81; 26.23; 26.48; 28.53 (2J119Sn–
13C = 144 Hz); 34.21 (1J119Sn–13C = 1168 Hz); 54.01; 196.04.
119Sn{1H} NMR (d, CHCl3): –307.

 Menezes
Menezes
 Vieira
Vieira
 De Lima
De Lima
 Porto
Porto
 Cortes
Cortes
 Ardisson
Ardisson
 Albrecht-Schmitt
Albrecht-Schmitt