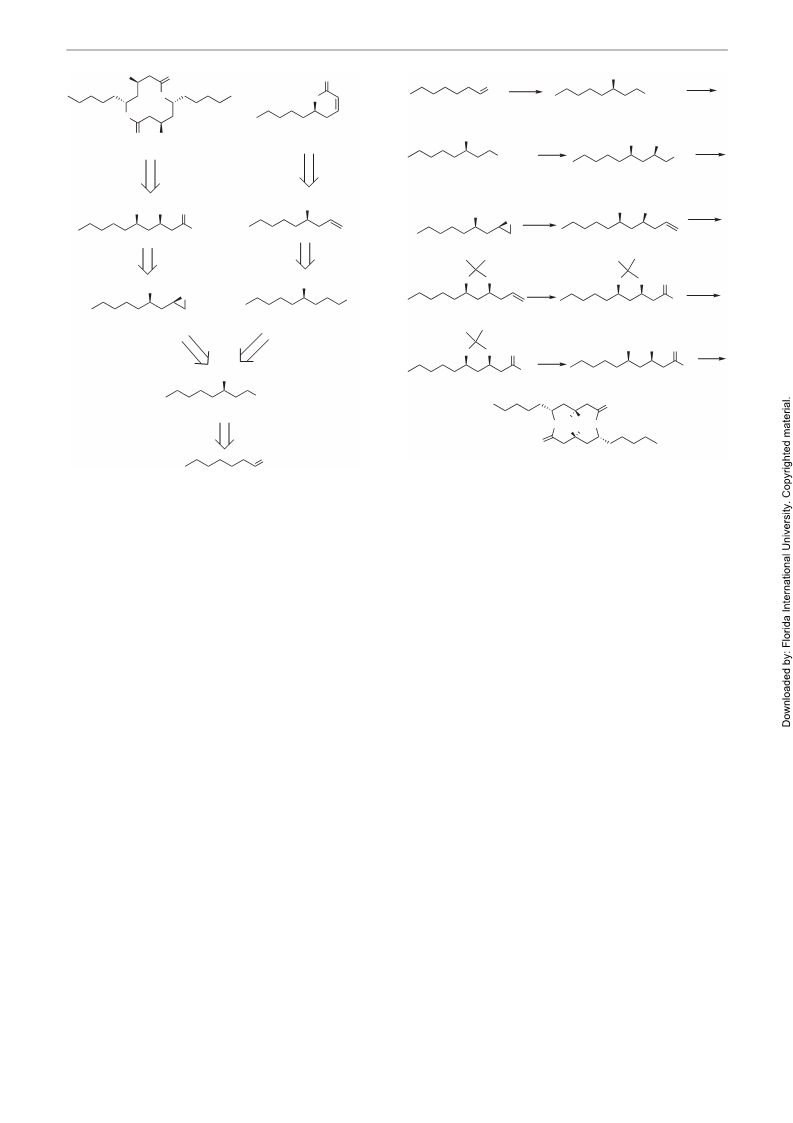
1958
A. Harbindu, P. Kumar
PAPER
1H NMR (400 MHz, CDCl3): d = 0.89 (t, J = 6.7 Hz, 3 H), 1.40 (s,
3 H), 1.44 (s, 3 H), 1.24–1.54 (m, 10 H), 2.46 (dd, J = 15.4, 5.6 Hz,
1 H), 2.58 (dd, J = 15.4, 6.8 Hz, 1 H), 3.79–3.88 (m, 1 H), 4.26–4.35
(m, 1 H).
Na2S2O3 (40 mL) and extracted with EtOAc (2 × 30 mL). The com-
bined organic layer was dried (Na2SO4) and concentrated under re-
duced pressure, then used without purification for the next step.
(R)-4-(tert-Butyldimethylsilyloxy)non-1-ene (15)
13C NMR (100 MHz, CDCl3): d = 14.0, 19.8, 22.5, 24.5, 30.2, 31.8,
To the crude iodo compound 14 (2.8 g, 7.29 mmol) dissolved in
benzene (25 mL) was added 1 N KtBD (10.4 mL, 10.4 mmol) and
the mixture was stirred at r.t. for 30 min. Then, the mixture was
poured into H2O and the organic layer was separated. The aqueous
layer was extracted with Et2O (3 × 20 mL) and the combined organ-
ic layer was washed with brine (2 × 20 mL), dried (Na2SO4) and
concentrated. Purification of the crude product by silica gel column
chromatography (PE) afforded 15 as a colorless liquid; yield: 1.29
g (69%).
1H NMR (200 MHz, CDCl3): d = 0.05 (s, 6 H), 0.90 (s, 12 H), 1.27–
1.45 (m, 8 H), 2.18–2.25 (m, 2 H), 3.63–3.74 (m, 1 H), 5.00–5.09
(m, 2 H), 5.72–5.93 (m, 1 H).
13C NMR (50 MHz, CDCl3): d = –4.5, –4.4, 14.0, 18.1, 22.6, 25.0,
25.8, 31.9, 36.7, 41.9, 72.0, 116.5, 135.5.
36.3, 41.4, 65.8, 68.9, 99.3, 176.2.
ESI-MS: m/z = 267 [M + Na]+.
Anal. Calcd for C13H24O4: C, 63.91; H, 9.90. Found: C, 63.83; H,
9.67.
Verbalactone (1)
A soln of acid 12 (120 mg, 0.49 mmol) and CSA (5.70 mg, 24.6
mmol) in anhyd MeOH (10 mL) was stirred at r.t. for 1 h. A citric
acid–sodium hydroxide buffer soln (pH 6) was added and the mix-
ture was extracted with EtOAc (5 × 20 mL). The combined extract
was dried (Na2SO4) and concentrated to afford deprotected 3 (80
mg) as a colorless syrup which was found to be unstable (on stand-
ing it undergoes lactonization to form monomeric lactone) and was
directly taken for the next reaction without further purification.
ESI-MS: m/z = 257 [M + H]+.
To a soln of seco acid 3 (24 mg, 117.5 mmol) in THF (4 mL) was
added Et3N (18.02 mL, 129.24 mmol) at r.t. The mixture was treated
with 2,4,6-trichlorobenzoyl chloride (18.36 mL, 117.5 mmol) and
stirred at r.t. for 2 h. The mixture was diluted with anhyd toluene (4
mL), then added dropwise over a period of 3 h to a refluxing soln of
DMAP (187.7 mg, 1.536 mmol) in anhyd toluene (40 mL). After
completion of the addition, the mixture was refluxed for an addi-
tional hour and then concentrated under reduced pressure to provide
crude product which on purification over silica gel (light PE–
EtOAc, 5:1) furnished verbalactone (1); yield: 11 mg (53%).
Anal. Calcd for C15H32OSi: C, 70.24; H, 12.57. Found: C, 69.88; H,
12.83.
(R)-Non-1-en-4-ol (16)
To a stirred soln of compound 15 (500 mg, 1.95 mmol) in anhyd
THF (10 mL) was added 1 M TBAF in THF (2.15 mL, 2.15 mmol)
at r.t. After completion of the reaction (2 h), some ice flakes were
added to the mixture which was then extracted with EtOAc (3 × 15
mL). The combined organic layer was dried (Na2SO4) and the sol-
vent was removed under reduced pressure to afford crude homoal-
lylic alcohol. Purification of the crude product by silica gel column
chromatography (PE–EtOAc, 9:1) gave 16 as a colorless liquid;
yield: 244 mg (88%).
[a]D25 +8.7 (c 0.9, CHCl3) [Lit.1 [a]D20 +7.3 (c 0.9, CHCl3)].
1H NMR (200 MHz, CDCl3): d = 0.86 (t, J = 6.9 Hz, 6 H), 1.16–
1.31 (m, 12 H), 1.45–1.60 (m, 4 H), 1.97 (td, J = 15.1, 4.5 Hz, 2 H),
2.02–2.10 (ddd, J = 14.5, 9.7, 3.1 Hz, 2 H), 2.67 (d, J = 3.6 Hz, 4
H), 3.73 (br s, 2 H), 4.03–4.08 (m, 2 H), 4.90–4.97 (m, 2 H).
[a]D25 +12.5 (c 0.9, CHCl3).
IR (CHCl3): 3351, 2926, 2854, 1641, 1589, 1457, 1378, 1259, 1156,
999, 836 cm–1.
1H NMR (500 MHz, CDCl3): d = 0.89 (t, J = 6.4 Hz, 3 H), 1.26–
1.47 (m, 8 H), 1.70 (br s, 1 H), 2.09–2.19 (m, 1 H), 2.27–2.35 (m, 1
H), 3.65–3.69 (m, 1 H), 5.12–5.16 (m, 2 H), 5.77–5.91 (m, 1 H).
13C NMR (50 MHz, CDCl3): d = 13.9, 22.5, 25.2, 31.8, 36.7, 41.8,
70.7, 117.6, 134.9.
13C NMR (50 MHz, CDCl3): d = 14.0, 22.4, 25.4, 31.3, 31.6, 36.2,
39.4, 64.7, 72.5, 172.9.
ESI-MS: m/z = 373 [M + H]+.
(R)-4-(tert-Butyldimethylsilyloxy)nonan-1-ol (13)
To a soln of ester 5 (3.0 g, 9.48 mmol) in CH2Cl2 (40 mL) was added
1.87 M DIBAL-H in toluene (12.67 mL, 23.69 mmol) at –78 °C un-
der argon atmosphere. The reaction mixture was stirred at this tem-
perature for 2 h. Then, a sat. soln of tartaric acid in H2O (10 mL) was
added. The resulting mixture was stirred for 15 min and the organic
layer was separated. The aqueous phase was extracted with CH2Cl2
(3 × 20 mL), and the combined organic layer was dried (Na2SO4),
filtered and concentrated under reduced pressure. Purification over
silica gel (light PE–EtOAc, 9:1) afforded alcohol 13 as a colorless
oil; yield: 2.16 g (83%).
1H NMR (200 MHz, CDCl3): d = 0.06 (s, 6 H), 0.91 (s, 12 H), 1.20–
1.27 (m, 6 H), 1.45–1.67 (m, 6 H), 3.55–3.77 (m, 3 H).
13C NMR (50 MHz, CDCl3): d = –4.5, 14.0, 18.0, 22.6, 25.1, 25.8,
31.9, 33.3, 36.4, 41.9, 63.1, 72.1.
(R)-Non-1-en-4-yl Acrylate (17)
Acryloyl chloride (0.95 g, 0.86 mL, 10.55 mmol) was added drop-
wise under argon to a soln of homoallylic alcohol 16 (1.5 g, 10.55
mmol) and Et3N (4.27 g, 5.9 mL, 42.18 mmol) in anhyd CH2Cl2 (15
mL) at 0 °C. The mixture was stirred at 0 °C for 6 h. Then, the mix-
ture was filtered through a pad of Celite® and poured into H2O (15
mL), and the organic layer was separated. The aqueous layer was
extracted with CH2Cl2 (3 × 30 mL) and the combined organic layer
was washed with brine (2 × 20 mL), dried (Na2SO4) and concentrat-
ed. Purification of the crude product by silica gel column chroma-
tography (PE–EtOAc, 19:1) afforded 17 as a colorless liquid; yield:
1.70 g (82%).
ESI-MS: m/z = 297 [M + Na]+.
[a]D25 +19.93 (c 1.1, CHCl3).
Anal. Calcd for C15H34O2Si: C, 65.63; H, 12.48. Found: C, 65.31;
H, 12.57.
IR (CHCl3): 2956, 2930, 2859, 1742, 1725, 1640, 1620, 1549, 1406,
1296, 1271, 1195, 1048, 986, 917, 809 cm–1.
1H NMR (200 MHz, CDCl3): d = 0.88 (t, J = 6.9 Hz, 3 H), 1.18–
1.29 (m, 8 H), 2.33–2.37 (m, 2 H), 4.96–5.11 (m, 3 H), 5.72–5.83
(m, 2 H), 6.10 (dd, J = 10.5, 17.2 Hz, 1 H), 6.42 (d, J = 18 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 13.9, 22.5, 24.9, 31.6, 33.5, 38.6,
73.6, 117.5, 128.9, 130.0, 132.7, 165.4.
(R)-4-(tert-Butyldimethylsilyloxy)-1-iodononane (14)
To a cooled (0 °C), stirred soln of Ph3P (2.10 g, 8.01 mmol) in THF–
MeCN (6:5, 30 mL) were added imidazole (0.60 g, 8.74 mmol) and
I2 (2.03 g, 8.01 mmol). The mixture was stirred for 2 h and then a
soln of alcohol 13 (2 g, 7.29 mmol) in THF (10 mL) was added at
0 °C. The mixture was stirred for 2 h, then diluted with 10% aq
Synthesis 2011, No. 12, 1954–1959 © Thieme Stuttgart · New York

 Harbindu, Anand
Harbindu, Anand
 Kumar, Pradeep
Kumar, Pradeep