
Polyhedron p. 81 - 88 (2014)
Update date:2022-08-28
Topics:
 Smolentsev, Anton I.
Smolentsev, Anton I.
 Lider, Elisaveta V.
Lider, Elisaveta V.
 Lavrenova, Ludmila G.
Lavrenova, Ludmila G.
 Sheludyakova, Liliya A.
Sheludyakova, Liliya A.
 Bogomyakov, Artem S.
Bogomyakov, Artem S.
 Vasilevsky, Sergei F.
Vasilevsky, Sergei F.
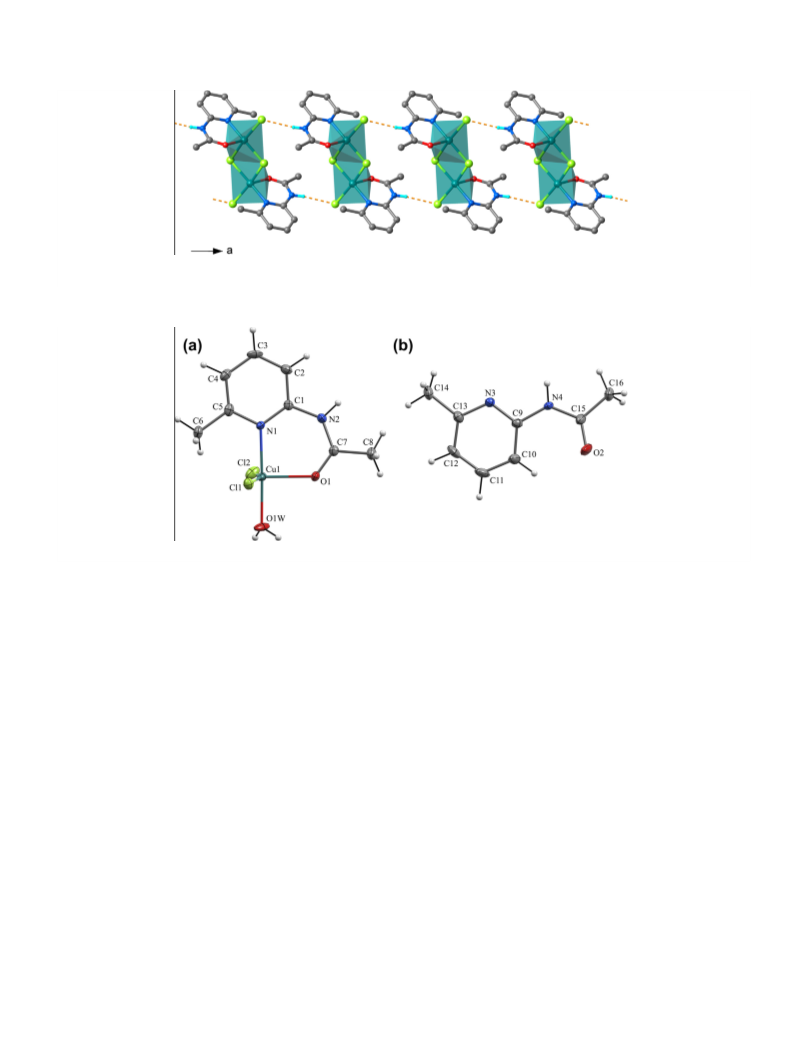
New coordination compounds of copper(II) chloride with 2-(N-acetylamino)-6- methylpyridine (L) of the compositions [Cu2L2Cl 4] (1), [CuL(H2O)Cl2]·L (1a), and [CuL2Cl][CuLCl3]·C2H5OH (2) have been prepared by the reactions between ethanol solutions of L and ethanol solutions of CuCl2·2H2O at different Cu:L ratios. The compounds have been characterized by a set of methods including elemental analysis, IR-spectroscopy, single-crystal X-ray diffraction analysis, and magnetic susceptibility measurements in the range 5-300 K (for 1). The crystal structures of 1 and 1a revealed distorted square-pyramidal geometry around Cu(II) centers, while in 2 both distorted square-pyramidal and trigonal-bipyramidal coordinations are present. L acts as a bidentate chelate ligand bound to the metal center through the pyridine N atom and the acetyl O atom. The results of the study demonstrate that the presence of 6-methyl substituent in L provides a high steric hindrance, precluding the formation of complexes with a distorted octahedral coordination of Cu(II).
View More





Beijing Stable Chemcial Co.ltd
Contact:86-10-63785052
Address:A1301 Technological Edifice. No.4 FuFeng Road,FengTai District, Beijing. China
Contact:+86-10-62651721
Address:29 Yongxing Road, Daxing District,Beijing China
Shanghai KFSL Pharmaceutical Technology Co.,Ltd.
Contact:+86-21-39971718
Address:859 jiadingchengliu shanghai
Yingkou Sanzheng Organic Chemical Co. Ltd.
Contact:+86-417-3638818
Address:25 Gengxinli Village, Daqing Road, Yingkou, Liaoning, China
Guangxi Shanyun Biochemical Science and Technology Co., Ltd
Contact:+86-0772--6828887
Address:#2 Industrial Park of Luzhai County, Liuzhou, Guangxi, China
Doi:10.1016/0022-1902(59)80100-7
(1959)Doi:10.1021/ol034334f
(2003)Doi:10.1002/jccs.201400377
(2015)Doi:10.1016/S0022-2860(99)00113-1
(1999)Doi:10.1039/jr9640003678
(1964)Doi:10.1016/j.tetasy.2003.09.014
(2003)