Journal of Catalysis 196, 345–351 (2000)
Zeolites for the Production of Fine Chemicals: Synthesis
of the Fructone Fragrancy
M. J. Climent, A. Corma, ,1 A. Velty, and M. Susarte†
Instituto de Tecnolog´ıa Quimica (UPV-CSIC), Universidad Polite´cnica de Valencia, Avda. de los Naranjos s/n, 46022 Valencia, Spain;
and †ACEDESA, Carretera de Mazarro´n, Km. 1.8, 30120 El Palmar, Murcia, Spain
Received May 4, 2000; revised August 10, 2000; accepted August 24, 2000
of the methods mentioned above present limitations due to
the use of expensive reagents, the tedious work-up proce-
dure, and the necessity of neutralisation (with the exception
of solid catalysts) of the strong acid media producing unde-
sired wastes. Consequently, there is a genuine need for an
efficient heterogeneous catalytic procedure for these reac-
tions. In this sense zeolites appear to be promising catalysts
with the obvious advantages of easy separation, controlled
acidity, shape selectivity, and reusability.
Preparation of fructone (ethyl 3,3-ethylendioxybutyrate)
(8), a flavouring material with apple scent, involves the
acetalization of ethyl acetoacetate with ethylene glycol.
This process is catalysed by strong acids such as p-
toluenesulfonic acid. It is known that in homogeneous me-
dia a strong acid and the water formed during the acetal-
ization can cause the hydrolysis of the ester, producing the
corresponding 3,3-ethylenedioxy-butanoic acid. The for-
mation of this product not only reduces the yield of fructone
but also, when it is present in amounts 3% , can alter the
organoleptic characteristic of the final product. Besides the
formation of 3,3-ethylenedioxy-butanoic acid, the use of
strong acid catalysts in liquid media presents the problem
derived from the hydrolysis of the ethyl acetoacetate that
results in a yield of fructone below 75% .
Fructone (ethyl 3,3-ethylendioxybutyrate), a flavouring material,
has been obtained by acetalization of ethyl acetoacetate with ethy-
lene glycol using different microporous and mesoporous alumi-
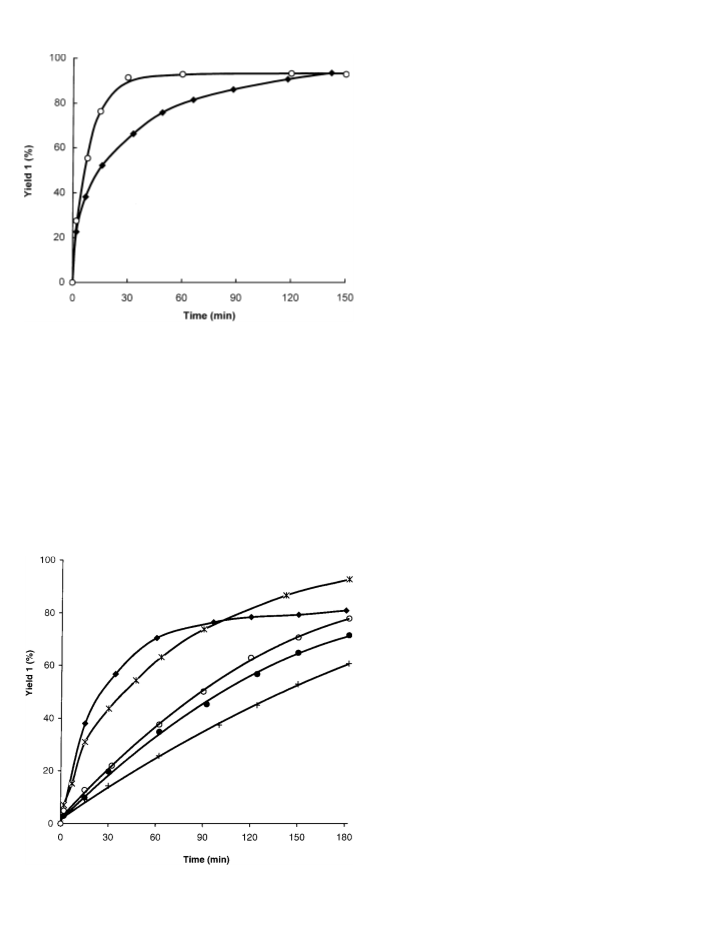
nosilicates as catalysts. Tridirectional zeolites (Y and Beta) are very
effective and selective for carrying out this reaction. It has been
found that a higher concentration of acid sites does not guarantee
a better catalytic performance and the hydrophobic–hydrophilic
properties of the material play a determinant role in this reaction
due to the different polarity of the reactants. The coupling between
the concentration of active sites and the adequate adsorption prop-
erties is achieved for Beta zeolites with a Si/Al ratio between 25 and
50. Optimisation of reaction conditions and the appropriate cata-
lyst allow us to obtain fructone with conversions and selectivities
c
close to 100%.
2000 Academic Press
Key Words: fructone; zeolites; acetalization.
INTRODUCTION
The acetalization reaction is sometimes necessary to pro-
tect carbonyl groups in the presence of other functional
groups when processing multifunctional organic molecules
(1). Besides the interest in acetals as protecting groups,
many of them have found direct applications as fragrances,
in cosmetics, as food and beverage additives, in pharmaceu-
ticals, in detergents, and in lacquer industries (2).
The most general method for the synthesis of acetals in-
volves the reaction of carbonyl compounds with an alcohol
or an orthoester in the presence of acid catalysts. A number
of acetalization procedures include the use of protic acids,
Lewis acids (zinc chloride) (3), alumina (4), montmoril-
lonite (5), zeolites (6), and mesoporous aluminosilicates (7)
and have had variable success. Acetalization of aldehydes
can be performed in the presence of weak acids, while ke-
tones generally need stronger conditions such as sulphuric,
hydrochloric, or p-toluenesulfonic acids and larger amounts
of catalyst than when reacting aldehydes. However, many
In the work described in this paper we have studied
the possibilities of zeolites as catalysts for the synthesis
of fructone from ethyl acetoacetate and ethylene glycol.
It will be shown that by controlling the acidity, textural,
and hydrophobic–hydrophilic properties of the zeolite, it is
possible to design a successful catalyst that, when coupled
with an adequate process design, maximises conversion and
selectivity to fructone (9).
EXPERIMENTAL
Catalysts
Beta zeolite ( -1) and ZSM-5 were supplied by PQ Corp.,
in the acidic and in the ammonium form, respectively. The
ZSM-5 sample was calcined at 823 K for 3 h to obtain the
1 To whom correspondence should be addressed. Fax: 34 96 387 78 09.
E-mail: acorma@itq.upv.es.
345
0021-9517/00 $35.00
c
Copyright
2000 by Academic Press
All rights of reproduction in any form reserved.

 Climent
Climent
 Corma
Corma
 Velty
Velty
 Susarte
Susarte