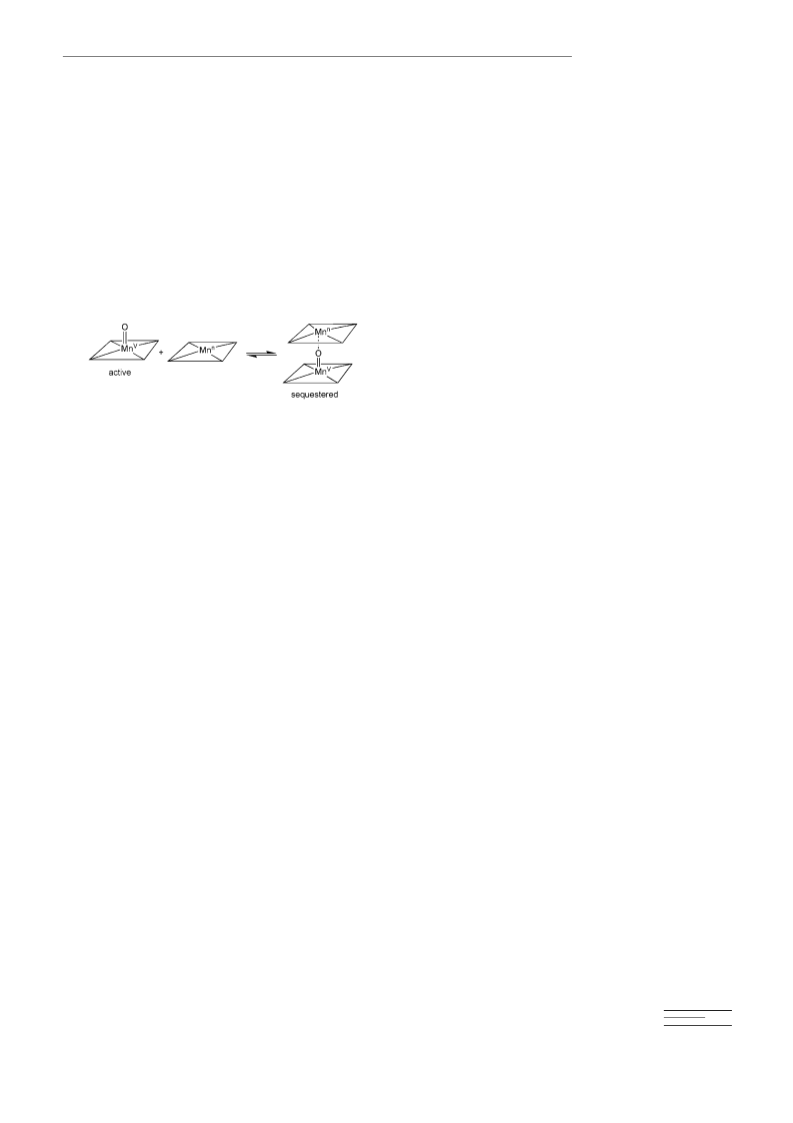M. Newcomb et al.
(BPMFC)MnIVCl (2b): LRMS (ESI): m/z: 788.2 [MꢀCl]+.
(TPC)MnIVCl (2c):[15] LRMS (ESI): m/z: 578.5 [MꢀCl]+.
Corrole–manganese(iv) chlorates (3): The compounds were prepared in
situ by stirring (Cor)MnIVCl with excess amounts of silver chlorate
(AgClO3) in CH2Cl2 followed by filtration. The formation of chlorate
products 3 was indicated by the change of UV/Vis absorption (Table 1).
The resulting solutions were used for LPF studies immediately after
preparation.
Acknowledgements
This work was supported by a grant from the National Institutes of
Health (GM 48722).
[1] B. Meunier, Chem. Rev. 1992, 92, 1411–1456. J. P. Collman, X.
Zhang, V. J. Lee, E. Uffelman, J. I. Brauman, Science 1993, 261,
1404–1411; Metalloporphyrins in Catalytic Oxidations (Ed.: R. A.
Scheldon), Marcel Dekker, New York, 1994; Metal-Oxo and Metal-
Peroxo Species in Catalytic Oxidations (Ed.: B. Meunier), Springer,
Berlin, 2000.
[2] For an early review about metallocorrole complexes, see: C. Erlen,
S. Will, K. M. Kadish, in Porphyrin Handbook, Vol. 2 (Eds.: K. M.
Kadish, K. M. Smith, R. Guilard), Academic Press, New York, 2000,
pp. 233–300.
[3] Z. Gross, N. Galili, I. Saltsman, Angew. Chem. 1999, 111, 1530–
1533; Angew. Chem. Int. Ed. 1999, 38, 1427–1429.
[4] Z. Gross, H. B. Gray, Adv. Synth. Catal. 2004, 346, 165–170, and ref-
erences therein.
[5] Z. Gross, G. Golubkov, L. Simkhovich, Angew. Chem. 2000, 112,
4211–4213; Angew. Chem. Int. Ed. 2000, 39, 4045–4047.
[6] A. Mahammed, Z. Gross, J. Am. Chem. Soc. 2005, 127, 2883–2887.
[7] G. Golubkov, J. Bendix, H. B. Gray, A. Mahammed, I. Goldberg,
A. J. DiBilio, Z. Gross, Angew. Chem. 2001, 113, 2190–2192;
Angew. Chem. Int. Ed. 2001, 40, 2132–2134.
Instrumentation: UV/Vis spectra were recorded on an Agilent 8453 spec-
trophotometer. Laser flash photolysis studies were conducted on an LK-
60 kinetic spectrometer (Applied Photophysics) at ambient temperature
(20 ꢁ 28C). Solutions of (Cor)MnIV(ClO3) with concentrations of ca. 2
10ꢀ5 m (after mixing) in methylene chloride were irradiated with 355 nm
light from a Nd/YAG laser (ca. 7 ns pulse). Data was acquired and ana-
lyzed with the Applied Photophysics software. Oversampling (64:1) was
employed in some cases to improve the signal to noise ratios. For kinetic
studies, an SC-18 mV stopped-flow mixing unit affixed to the kinetic
spectrometer was employed. For generation of (Cor)MnV(O) species in
mixing studies, an RX2000 rapid kinetics spectrometer accessory (Ap-
plied Photophysics) coupled with the above UV spectrometer was em-
ployed with 400 ms to seconds time scales; solutions of (Cor)MnIII com-
plexes were treated with equivalent amounts of MCPBA or PhIO.
Quantum yields: The values were determined relative to the yield of a
standard by the general method of Hoshino.[17]
A solution of the
(Cor)MnIV(ClO3) complex 3 with an absorbance of 0.5 at 355 nm was ir-
radiated with the third harmonic of the Nd/YAG laser (355 nm) at 30 mJ
of power per pulse, and the absorption at lmax for the ca. 350 nm band of
(Cor)MnV(O) product 4 was measured. The molar yield of 4 was deter-
mined from the extinction coefficients found in the mixing studies
(Table 2). The yield of 4 was compared with the yield of benzophenone
triplet formed by 355 nm irradiation of a solution with an absorbance of
0.5 at 355 nm. In this method, the quantum yield for excitation of benzo-
phenone is taken to be 1.0.[17] The quantum yield was calculated from the
ratio of molar yields and ratio of molar extinction coefficients for the
product of interest and the benzophenone triplet.
[8] H. Y. Liu, T. S. Lai, L. L. Yeung, C. K. Chang, Org. Lett. 2003, 5,
617–620.
[9] B. S. Mandimutsira, B. Ramdhanie, R. C. Todd, H. L. Wang, A. A.
Zareba, R. S. Czernuszewicz, D. P. Goldberg, J. Am. Chem. Soc.
2002, 124, 15170–15171.
[10] R. Zhang, M. Newcomb, J. Am. Chem. Soc. 2003, 125, 12418–
12419.
[11] R. Zhang, J. H. Horner, M. Newcomb, J. Am. Chem. Soc. 2005, 127,
6573–6582.
[12] R. Zhang, R. E. P. Chandrasena, E. Martinez II, J. H. Horner, M.
Newcomb, Org. Lett. 2005, 7, 1193–1195.
[13] R. Paolesse, S. Nardis, F. Sagone, R. G. Khoury, J. Org. Chem. 2001,
66, 550–556.
[14] D. T. Gryko, B. Koszarna, Synthesis 2004, 2205–2209, and references
therein.
[15] E. Steene, T. Wondimagegn, A. Ghosh, J. Phys. Chem. B 2001, 105,
11406–11413.
[16] A. D. Alder, F. R. Longo, F. Kampas, J. Kim, J. Inorg. Nucl. Chem.
1970, 32, 2443–2445.
[17] M. Hoshino, S. Arai, M. Yamaji, Y. Hama, J. Phys. Chem. 1986, 90,
2109–2111.
[18] J. P. Collman, L. Zeng, R. A. Decrꢁau, Chem. Commun. 2003, 2974–
2975.
[19] K. S. Suslick, R. A. Watson, New J. Chem. 1992, 16, 633–642; H.
Hennig, Coord. Chem. Rev. 1999, 182, 101–123.
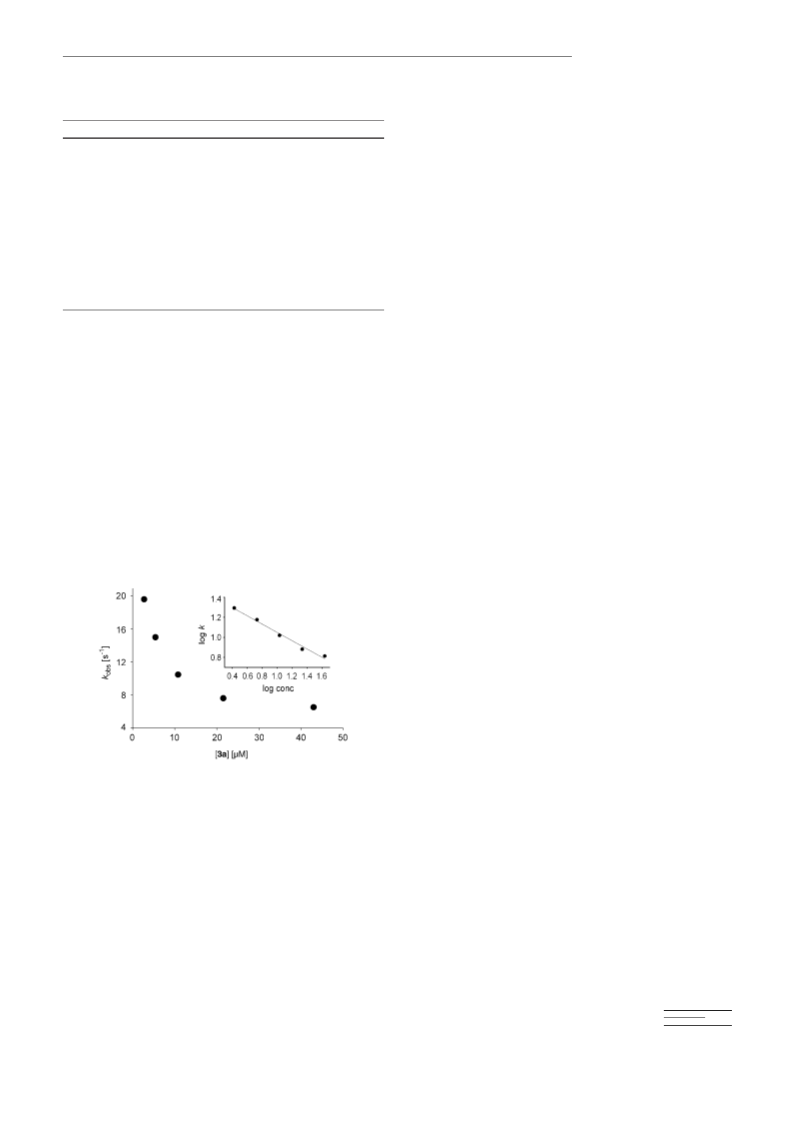
Kinetics: In LFP studies, solutions of precursor 3 were irradiated by the
laser, and decay of 4 at lmax was monitored. For reactions with substrates,
solutions of 3 were mixed with solutions of substrate at varying concen-
trations, the resulting mixtures were irradiated by the laser after a delay
of < 1 s, and decay of (Cor)MnV(O) (4) was followed at lmax. For stop-
ped-flow mixing studies, 1a in CH2Cl2 solution was oxidized with 1.0
equivalent of MCPBA, and the resulting solution containing 4a was
mixed with CH2Cl2 solutions of cis-cyclooctene. The kinetic results were
complex but could be fit reasonably well for single exponential processes.
For each reaction with substrate, four or five concentrations of substrate
were studied. All kinetic runs are the average of three independent deter-
minations. Apparent second-order rate constants for reactions of 4 with
substrates were determined from the observed decay rate constants with
Equation (1). All errors listed are 2s.
[20] J. T. Groves, J. Lee, S. S. Marla, J. Am. Chem. Soc. 1997, 119, 6269–
6273.
Competitive oxidations: Solutions containing cis-stilbene (0.25 mmol),
cis-cyclooctene (0.25 mmol) and (Cor)MnIII (5 mmol) in CH2Cl2 were pre-
pared. PhIO (0.125 mmol) was added, and the mixture was stirred under
a nitrogen atmosphere at ca. 228C until the PhIO was consumed. The re-
action mixture was passed through a short silica gel column and analyzed
by GC after addition of an internal standard or, after concentration, by
1H NMR spectroscopy of a CDCl3 solution. The products were identified
by comparison to authentic samples. The products from cis-stilbene were
cis- and trans-stilbene oxide (cis/trans 5:1), quantitated using their charac-
teristic singlets at d 4.3 and 3.8, respectively, and a trace of benzaldehyde
(<5%). For cis-cyclooctene, the only product detected was cis-cyclooc-
tene oxide. Total yields based on PhIO were ca. 60% as determined by
quantitative GC. The product ratios listed in the text (determined by GC
and/or NMR integration) are the averages of three determinations.
[21] N. Jin, J. T. Groves, J. Am. Chem. Soc. 1999, 121, 2923–2924.
[22] W. Nam, I. Kim, M. H. Lim, H. J. Choi, J. S. Lee, H. G. Jang, Chem.
Eur. J. 2002, 8, 2067–2071.
[23] G. Golubkov, Z. Gross, J. Am. Chem. Soc. 2005, 127, 3258–3259.
[24] P. E. Ellis, J. W. Lyons, Coord. Chem. Rev. 1990, 105, 181–193.
[25] M. W. Grinstaff, M. G. Hill, J. A. Labinger, H. B. Gray, Science 1994,
264, 1311–1313.
[26] D. Dolphin, T. G. Traylor, L. Y. Xie, Acc. Chem. Res. 1997, 30, 251–259.
[27] S. H. Wang, B. S. Mandimutsira, R. Todd, B. Ramdhanie, J. P. Fox,
D. P. Goldberg, J. Am. Chem. Soc. 2004, 126, 18–19.
Received: February 8, 2005
Revised: May 8, 2005
Published online: July 20, 2005
5720
ꢀ 2005 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 2005, 11, 5713 – 5720

 Zhang, Rui
Zhang, Rui
 Harischandra, Dilusha N.
Harischandra, Dilusha N.
 Newcomb, Martin
Newcomb, Martin