Please do not adjust margins
New Journal of Chemistry
Page 8 of 9
DOI: 10.1039/C7NJ05058E
PAPER
NJC
3
a) Y. Du, D.‐L. Kong, H.‐Y. Wang, F. Cai, J.‐S. Tian, J.‐Q. Wang, L.‐N. He,
J. Mol. Catal. A: Chem., 2005, 241, 233; b) L. Plasseraud, H. Cattey, P.
Richard; Z. Naturforsch., B: Chem. Sci., 2009, 64, 831.
N. Germain, I. Müller, M. Hanauer, R. A. Paciello, R. Baumann, O.
Trapp, T. Schaub, ChemSusChem, 2016, 9, 1586.
a) L. Plasseraud, D. Ballivet‐Tkatchenko, H. Cattey, S. Chambrey, R.
Ligabue, P. Richard, R. Willem, M. Biesemans, J. Organomet. Chem.,
2010, 695, 1618; b) G. Laurenczy, M. Picquet, L. Plasseraud, J.
Organomet. Chem., 2011, 696, 1904; c) G. Laurenczy, A. F. Dalebrook,
M. Picquet, L. Plasseraud, J. Organomet. Chem., 2015, 796, 53.
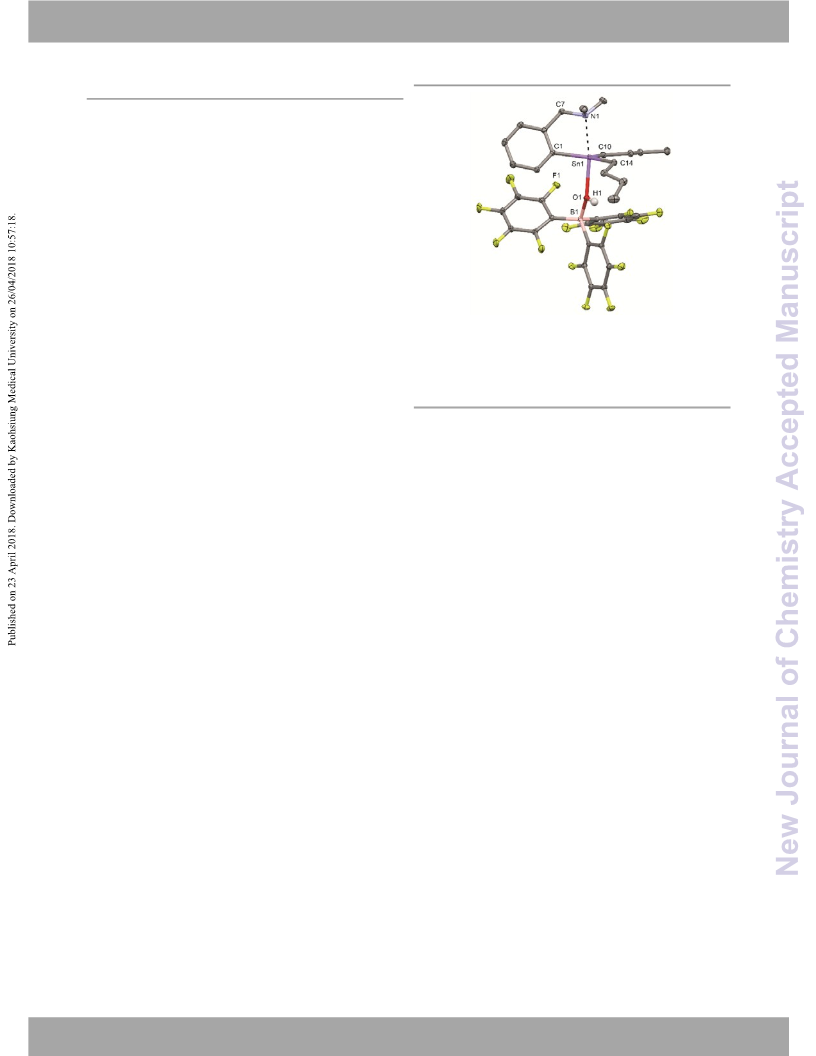
Single ingle crystals of 7 were grown from a methanolic solution
which was recovered from the reactor after a catalytic run using
1
4
as a catalyst. M. p. > 220 °C (dec.). H NMR (THF‐d8, 295 K,
4
5
ppm): 2.72 (br, 18H, OCH3); 2.10 (br, 2H, CcageH); 1.65 (br m,
20H,
‐CH2); 1.49‐1.38 (m, 40H,
‐ and γ‐CH2); 0.95 (t, 60H,
‐
CH3, J(1H, H) = 7.0 Hz); broad BH resonances with overall
integral intensity of 22 display a typical pattern ranging from
2.05 to 1.05 ppm and some of them are overlapped by
resonances of the n‐Bu moieties and residual THF signal by
coincidence. 11B{1H} NMR (THF‐d8, 295 K, ppm): ‐7.6 (br s, 2B,
B(12)); ‐13.9 (br s, 10B, B(7‐11)); ‐17.0 (br s, 10B, B(2‐6)).
119Sn{1H} NMR (THF‐d8, 295 K, ppm): ‐164.3 (s, 2Sn); ‐177.2 (s,
2Sn); ‐207.6 (s, 4Sn), ‐211.8 (s, 2Sn). IR (ATR, selected bands, cm‐
3
1
6
7
S. R. Sanapureddy, L. Plasseraud, Appl. Organomet. Chem., 2017, 31,
2335.
a) P. Švec, A. Eisner, L. Kolářová, T. Weidlich, V. Pejchal, A. Růžička,
Tetrahedron Lett., 2008, 49, 6320; b) P. Švec, A. Růžička, Main Group
Met. Chem., 2011, 34, 7; c) J. Turek, I. Panov, P. Švec, Z. Růžičková, A.
Růžička, Dalton Trans., 2014, 43, 15465.
T. Weidlich, L. Dušek, B. Vystrčilová, A. Eisner, P. Švec, A. Růžička,
Appl. Organomet. Chem., 2012, 26, 293.
a) P. Švec, Z. Padělková, A. Růžička, T. Weidlich, L. Dušek., L.
Plasseraud, J. Organomet. Chem., 2011, 696, 676; b) P. Švec, R.
Olejník, Z. Padělková, A. Růžička, L. Plasseraud, J. Organomet. Chem.,
2012, 708, 82.
1): 2956 ν(C
ν(B H), 1461, 1426, 1409, 1377, 1048, 1019, 964, 870, 836, 670,
H), 2924 ν(CH), 2857 ν(CH), 2819 (OCH), 2535
8
9
619, 580. Elemental analysis (%): found: C, 35.84; H, 7.48. Calcd.
for C90H222B22O16Sn10 (2985.65): C, 36.21; H, 7.49.
10 For example see: a) P. Švec, K. Bartoš, Z. Růžičková, P. Cuřínová, L.
Dušek, J. Turek, F. De Proft, A. Růžička, New. J. Chem., 2016, 40, 5808;
b) P. Švec, Z. Růžičková, P. Vlasák, J. Turek, F. De Proft, A. Růžička, J.
Organomet. Chem., 2016, 801, 14; c) C. Coza, A. Stegarescu, R. Şuteu,
A. Silvestru, J. Organomet. Chem., 2015, 777, 71; d) R. A. Varga, A.
Rotar, M. Schürmann, K. Jurkschat, C. Silvestru, Eur. J. Inorg. Chem.,
2006, 1475; e) R. A. Varga, M. Schurmann, C. Silvestru, J. Organomet.
Chem., 2001, 623, 161.
4.6 High‐pressure experiments
Safety Warning: Experiments involving pressurized gases can
be hazardous and must only be conducted with suitable
equipment and following appropriate safety considerations.48
General procedure for reactions of methanol with carbon
dioxide: The reaction was carried out in a 125 mL stainless steel
reactor equipped with a magnetic stirrer. The reactor was
purged with argon and a 20 mL solution of the appropriate C,N‐
11 a) J. Plešek, T. Jelínek, E. Drdáková, S. Heřmánek, B. Štíbr, Collect.
Czech. Chem. Commun., 1984, 49, 1559; b) S. Körbe, P. J. Schreiber, J.
Michl, Chem. Rev., 2006, 106, 5208.
chelated organotin(IV) species (4 and 5) in anhydrous methanol
12 N. J. Patmore, C. Hague, J. H. Cotgreave, M. F. Mahon, C. G. Frost, A.
S. Weller, Chem.‐Eur. J., 2002, 8, 2088.
was introduced by syringe. Then, CO2 was admitted to the
desired amount (see Table 1). The reaction temperature (150
°C) was controlled by an internal thermocouple. After a reaction
time of 24 hours the reactor was cooled down to 0°C, the
pressure was gently released and the liquid phase was
transferred to a Schlenk tube. Trap‐to‐trap distillation under
vacuum at ambient temperature allowed separation of volatile
compounds that were quantitatively analyzed by GC (DEC
external standard, Fisons 8000, J&W Scientific DB‐WAX 30 m
capillary column, FID detector). Tin‐based residue was firstly
characterized by IR and multinuclear NMR spectroscopy.
13 J. Turek, Z. Padělková, Z. Černošek, M. Erben, A. Lyčka, M. S. Nechaev,
I. Císařová, A. Růžička, J. Organomet. Chem., 2009, 694, 3000.
14 For example see: a) P. Novák, I. Císařová, L. Kolářová, A. Růžička, J.
Holeček, J. Organomet. Chem., 2007, 692, 4287; b) B. Kašná, R.
Jambor, L. Dostál, A. Růžička, I. Císařová, J. Holeček, Organometallics,
2004, 23, 5300.
15 Z. Padělková, T. Weidlich, L. Kolářová, A. Eisner, I. Císařová, T. A.
Zevaco, A. Růžička, J. Organomet. Chem., 2007, 692, 5633.
16 Z. Růžičková, J. Holub, P. Melichar, J. Moncol, D. A. Wann, J. Fanfrlík,
A. Růžička, D. Hnyk, Eur. J. Org. Chem., 2016, 4473.
17 Unpublished results.
18 a) Bruker Almanac, ISBN‐13 978‐3‐929431‐25‐4, 2011; b) B.
Wrackmeyer, Annu. Rep. NMR Spectrosc., 1988, 20, 61.
19 a) J. Bareš, P. Novák, M. Nádvorník, R. Jambor, T. Lébl, I. Císařová, A.
Růžička, J. Holeček, Organometallics, 2004, 23, 2967; b) P. Švec, P.
Novák, M. Nádvorník, Z. Padělková, I. Císařová, L. Kolářová, A. Růžička,
J. Holeček, J. Fluorine Chem., 2007, 128, 1390.
20 P. Brown, M. F. Mahon, K. C. Molloy, J. Chem. Soc., Dalton Trans.,
1992, 3503.
21 A. S. Larsen, J. D. Holbrey, F. S. Tham, C. A. Reed, J. Am. Chem. Soc.,
2000, 122, 7264.
Acknowledgement
The Czech authors would like to thank the Czech Science Foundation
(project 16‐01618S) for the financial support of this work. The French
authors gratefully acknowledge the Centre National de la Recherche
Scientifique (CNRS, France) and the University of Bourgogne
Franche‐Comté (Dijon, France).
22 J. Dymon, R. Wibby, J. Kleingardner, J. M. Tanski, I. A. Guzei, J. D.
Holbrey, A. S. Larsen, Dalton Trans., 2008, 2999.
23 a) A. G. Davies, Organotin Chemistry, Chapters 12‐114, pp 179‐243,
Wiley‐VCH, Weinheim, Germany, FRG 1997 & 2004; b) M. Gielen, in:
Tin Chemistry: Fundamentals, Frontiers, and Applications, A. G.
Davies, K. Pannell, E. R. T. Tiekink (eds.), ISBN: 978‐0‐470‐51771‐0,
Chapters 2.3 and 2.10, 2008.
References
1
a) J. Kizlink, Collect. Czech. Chem. Commun., 1993, 58, 1399; b) J.
Kizlink, I. Pastucha, Collect. Czech. Chem. Commun., 1995, 60, 687.
a) K.‐I. Tominaga, Y. Sasaki, Synlett 2002, 307; b) S. Pulla, C. M. Felton,
24 a) E. R. T. Tiekink, Appl. Organomet. Chem., 1991, 5, 1; b) R. R. Holmes,
2
Acc. Chem. Res., 1989, 22, 190; c)V. Chandrasekhar, S. Nagendran, V.
Baskar, Coord. Chem. Rev., 2002, 235, 1; d) R. Murugavel, N. Gogoi, J.
Organomet. Chem., 2008, 693, 3111.
Y. Gartia, P. Ramidi, A. Ghosh, ACS Sustainable Chem. Eng., 2013, 1,
309.
8 | New J. Chem., 2012, 00, 1‐3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 ?vec, Petr
?vec, Petr
 Cattey, Hélène
Cattey, Hélène
 R??i?ková, Zdeňka
R??i?ková, Zdeňka
 Holub, Josef
Holub, Josef
 R??i?ka, Ale?
R??i?ka, Ale?
 Plasseraud, Laurent
Plasseraud, Laurent