Organometallics
Article
2
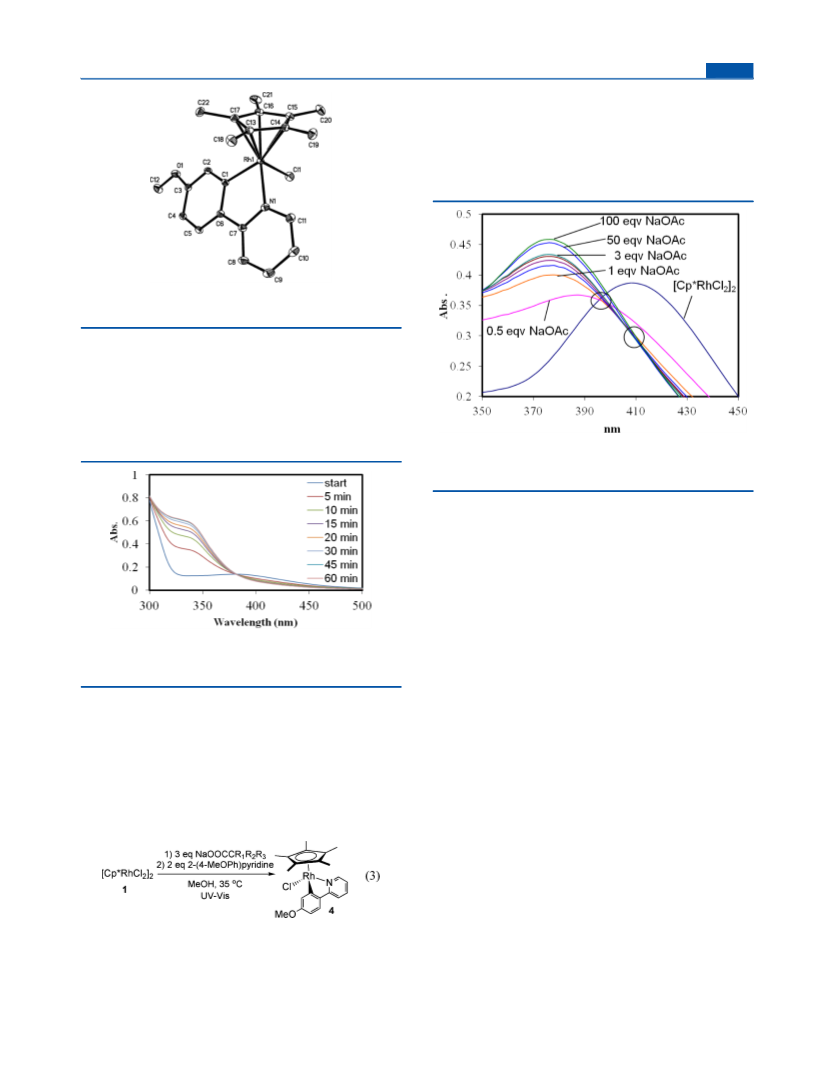
Figure 9. NMR experiment of Cp*RhCl(κ -OAc) and 4-picoline (1:1) before and after mixing at room temperature.
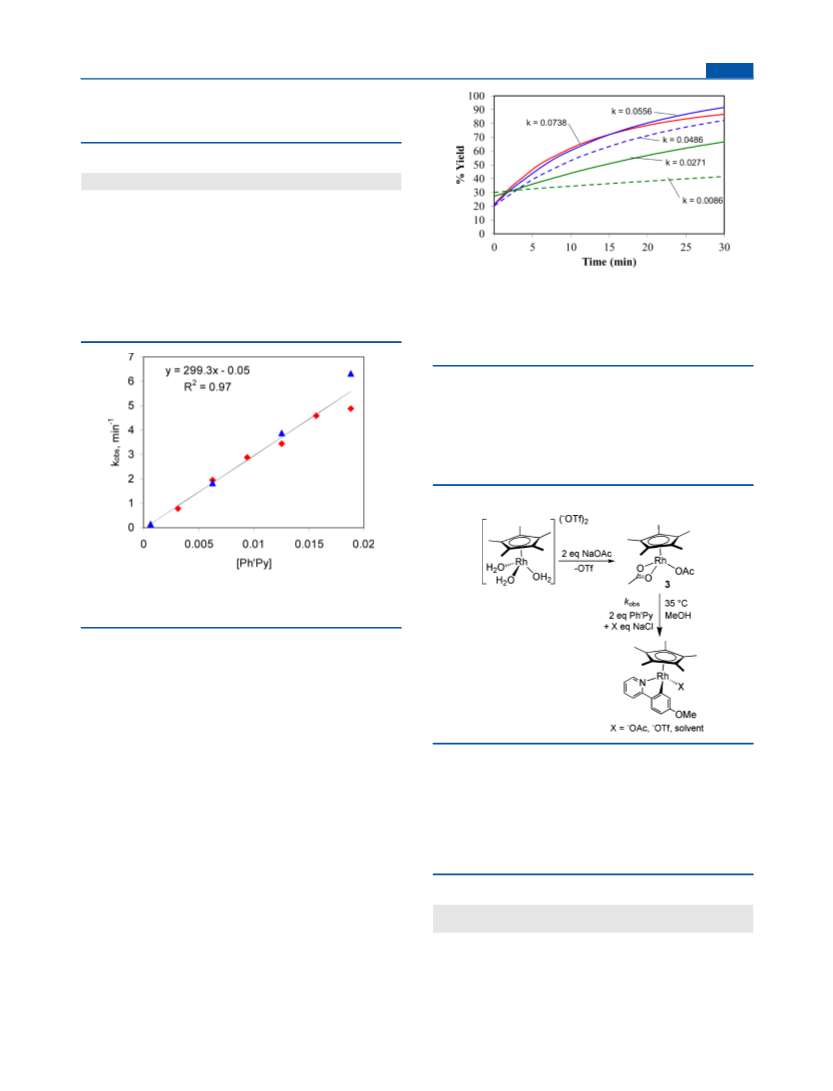
while studying the kinetics of the reaction under stoichiometric
conditions provides useful information, it does not necessarily
mean that the reaction follows those observed kinetics under
catalytically relevant conditions. The evalution of the
mechanism using kinetic studies allowed for a mechanistic
proposal in which a second Ph′Py coordinates prior to the rate-
determining C−H activation.
(400 MHz, CDCl
J = 8.4 Hz, 1H), 7.36 (d, J = 2 Hz, 1H), 7.03 (dt, J = 5.8, 1.6 Hz, 1H),
): δ 8.65 (d, J = 5.2 Hz, 1H), 7.62 (m, 2H), 7.53 (d,
3
1
3
1
6
.61 (dd, J = 8.4, 2 Hz, 1H), 3.9 (s, 3H), 1.62 (s 15H). C { H} NMR
(125 MHz, CDCl ): δ 180.9, 180.6, 165.3, 160.6, 151.2, 137.0 (d),
3
1
21.3, 120.9, 118.4, 109.4, 96.0 (d), 55.2, 9.3. Anal. Calcd (found) for
C H ClNORh: C, 57.72 (57.81); H, 5.504 (5.40); N, 3.06 (3.04).
22
25
Synthesis of Sodium Difluoroacetate. Sodium carbonate (562.7
mg, 5.3 mmol) was placed in a round-bottom flask followed by a stir
bar and 10 mL of THF. Difluoroacetic acid (0.67 mL, 10.6 mmol) was
added with stirring, and then a reflux condenser was attached and the
flask was submerged in an oil bath and refluxed for 3 h. The reaction
mixture was cooled and filtered through a frit while warm to give a
EXPERIMENTAL SECTION
■
3
General Procedures, Materials, and Instrumentation. RhCl3·
H O was purchased from Pressure Chemical Co., and [Cp*RhCl ]
was prepared according to the literature. Chloroacetic acid,
phenylacetic acid, pivalic acid, sodium iodoacetate, sodium acetate,
sodium trifluoroacetate and sodium tert-butoxide (2 M in THF) were
purchased from Sigma-Aldrich. Cyanoacetic acid, difluoroacetic acid,
2
2
2
1
8
white solid, that was dried under vacuum overnight; yield 1.21 g
−1
(
(
97%). IR (solid): 1634 cm (CO). Mp: 172−174 °C. Anal. Calcd
found) for C HF NaO : C, 20.35 (20.25); H, 0.85 (0.78).
2
2
2
Synthesis of Sodium Cyanoacetate. Cyanoacetic acid (372.4
mg, 4.3 mmol) was placed in a round-bottom flask followed by a stir
bar and 10 mL of THF. The flask was sealed with a septum, the
solution was stirred, and the flask was submerged in an ice−water bath.
Then sodium tert-butoxide (2.16 mL, 4.3 mmol) was added to the
mixture, dropwise, over the course of 10 min. Instant precipitation of a
white solid occurred, and the mixture was stirred for 2 h. The mixture
was then filtered through a frit and washed with three portions of cold
diethyl ether (25 mL total). The solid was dried under high vacuum
2-(4-methoxyphenyl)pyridine, methyl fluoroacetate, and 2-phenyl-
pyridine were purchased from TCI Chemicals. Isobutyric acid and
valeric acid were purchased from Acros Organics. Sodium hydroxide
and methanol were purchased from Fisher Scientific. 4-Picoline was
purchased from Alfa Aesar. Propionic acid was purchased from
Eastman Kodak. Sodium carbonate was purchased from Macron
Chemicals. All purchased materials were used as received. Tetrahy-
drofuran, diethyl ether, and dichloromethane were purchased from
Fisher Scientific and purified by passage through activated alumina
columns in an Innovative Technology, Inc., PS-MD-6 solvent
purification system. Deuterated solvents were purchased from
−1
overnight; yield 400.4 mg (99%). IR (solid): 2256 cm (CN), 1598
−
1
cm
(CO). Mp: 175−176 °C. Anal. Calcd (found) for
2
C H NaNO : C, 33.66 (33.55); H, 1.88 (1.95); N, 13.08 (12.72).
3
2
Synthesis of Sodium Fluoroacetate. Ground sodium hydroxide
399.5 mg, 10 mmol) was placed in a round-bottom flask followed by
Cambridge Isotope Laboratories. Chloroform-d (δ 7.26) was used
1
(
as received, and methanol-d (δ 3.31) was dried over 4 Å molecular
4
a stir bar and 10 mL of THF. Methyl fluoroacetate (0.78 mL, 10
mmol) was then added, a reflux condenser was attached, and the flask
was submerged in an oil bath and refluxed for 2 h with stirring. The
mixture was cooled to room temperature, and the solvent was
evaporated to give a white solid which was dried under high vacuum
sieves (activated under vacuum while heating overnight) and distilled
1
under vacuum. H NMR spectra were recorded using an Avance 400
MHz spectrometer, and 1 C{ H} NMR spectra were recorded using
3
1
an Avance 500 MHz spectrometer. UV−vis kinetics were carried out
using a Hewlett-Packard 8452 diode array spectrophotometer with a
Hewlett-Packard 89090A Peltier temperature controller. Elemental
analyses were determined by the CENTC Elemental Analysis Facility
at the University of Rochester using a PerkinElmer 2400 SeriesII
analyzer equipped with a PerkinElmer Model AD-6 autobalance by Dr.
William W. Brennessel. IR spectra were recorded using a Shimadzu IR
Prestige-21 FTIR spectrophotometer with a PIKE Technologies
MIRacle single-reflection ATR. X-ray diffraction data were collected
using a Bruker SMART APEX II CCD Platform diffractometer.
Synthesis of Cp*RhCl(2-(4-methoxyphenyl)pyridine) (4). The
−1
overnight; yield 946.1 mg (94%). IR (solid): 1608 cm (CO). Mp:
>170 °C dec. Anal. Calcd (found) for C H FNaO : C, 24.01 (23.87);
2 2 2
H, 2.015 (1.97).
Synthesis of Sodium Chloroacetate. The synthesis is analogous
−
1
to NaO
CCH
CN; yield 99%. IR (neat): 1591 cm (CO). Mp:
H ClNaO : C, 20.62 (20.24);
2 2 2
2
2
>170 °C dec. Anal. Calcd (found) for C
H, 1.73 (1.65).
Synthesis of Sodium Phenylacetate. Sodium carbonate (75.6
mg, 0.713 mmol) was placed in a round-bottom flask followed by a stir
bar. Then phenylacetic acid (194.7 mg, 1.43 mmol) was added,
followed by 5 mL of distilled water, and the solution was stirred
vigorously for 30 min. The solvent was evaporated and the solid white
1
3
synthesis was analogous to the previously published procedure. The
crude material was crystallized via vapor diffusion of cyclohexane into a
1
saturated CH Cl solution; purified yield 39.7 mg (80%). H NMR
2
2
3
406
Organometallics 2015, 34, 3400−3407

 Walsh, Aaron P.
Walsh, Aaron P.
 Jones, William D.
Jones, William D.