
Journal of the American Chemical Society p. 9883 - 9889 (1994)
Update date:2022-08-17
Topics:
 Bilski, Piotr
Bilski, Piotr
 Reszka, Krzysztof
Reszka, Krzysztof
 Chignell, Colin F.
Chignell, Colin F.
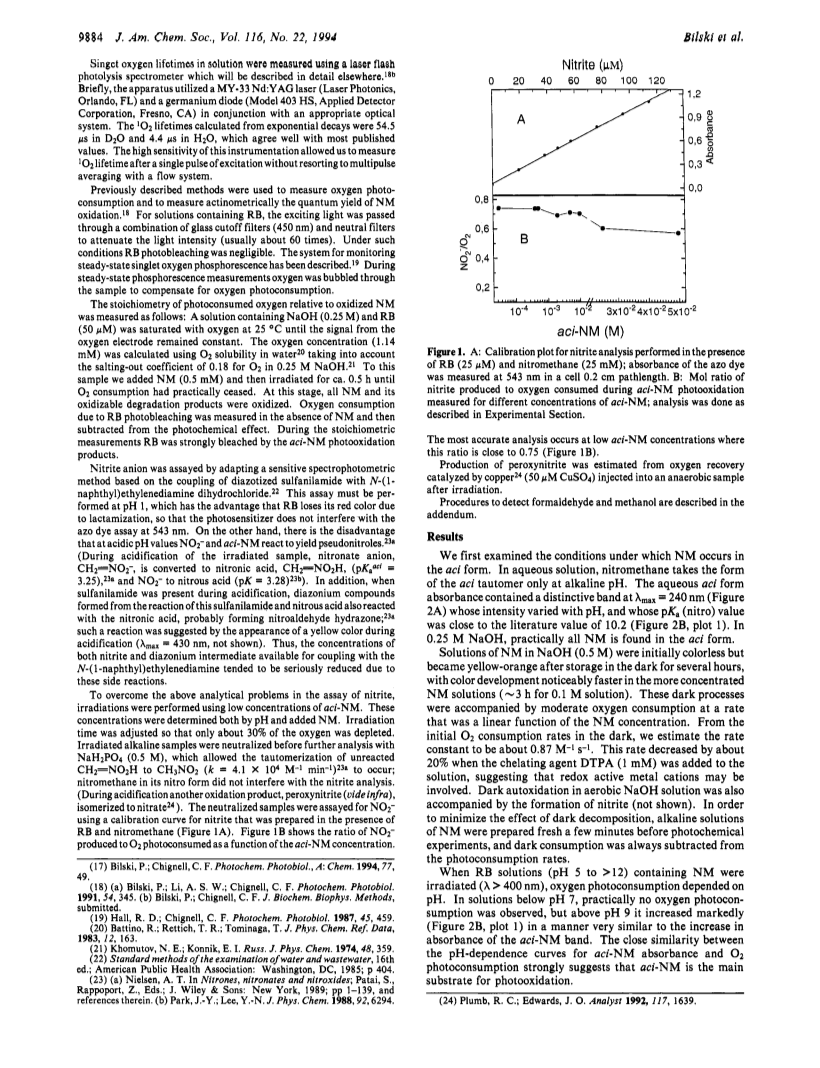
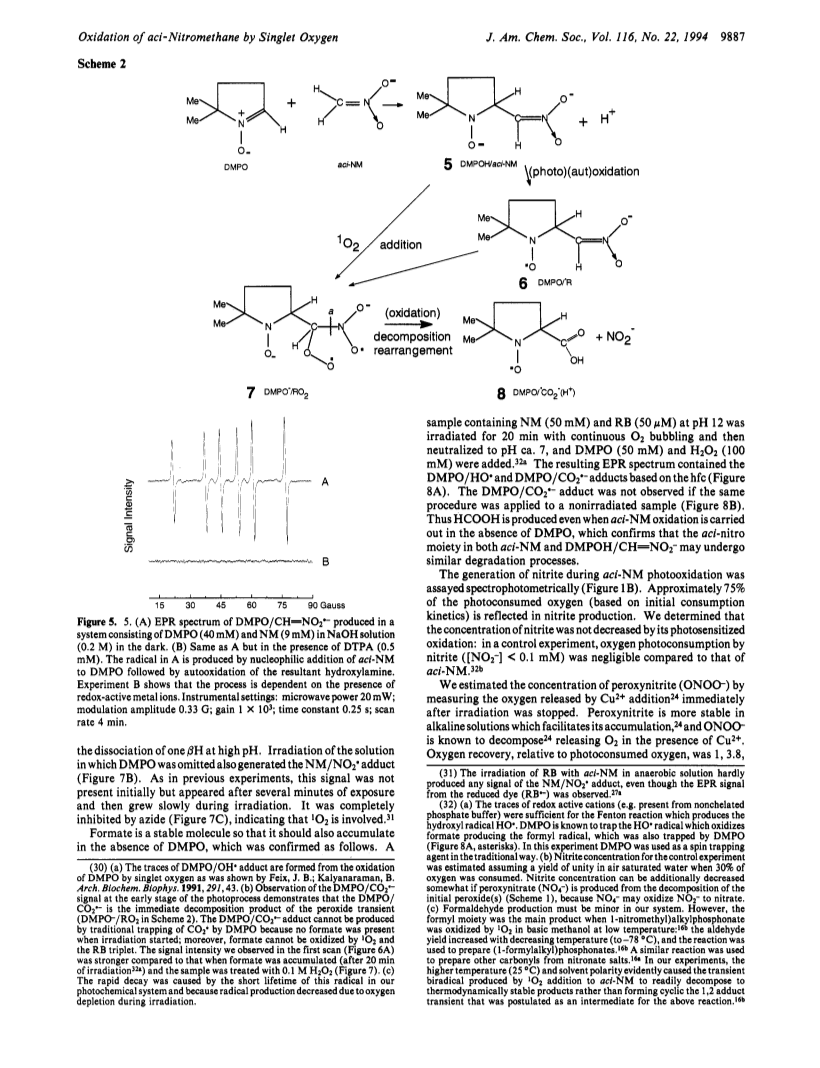
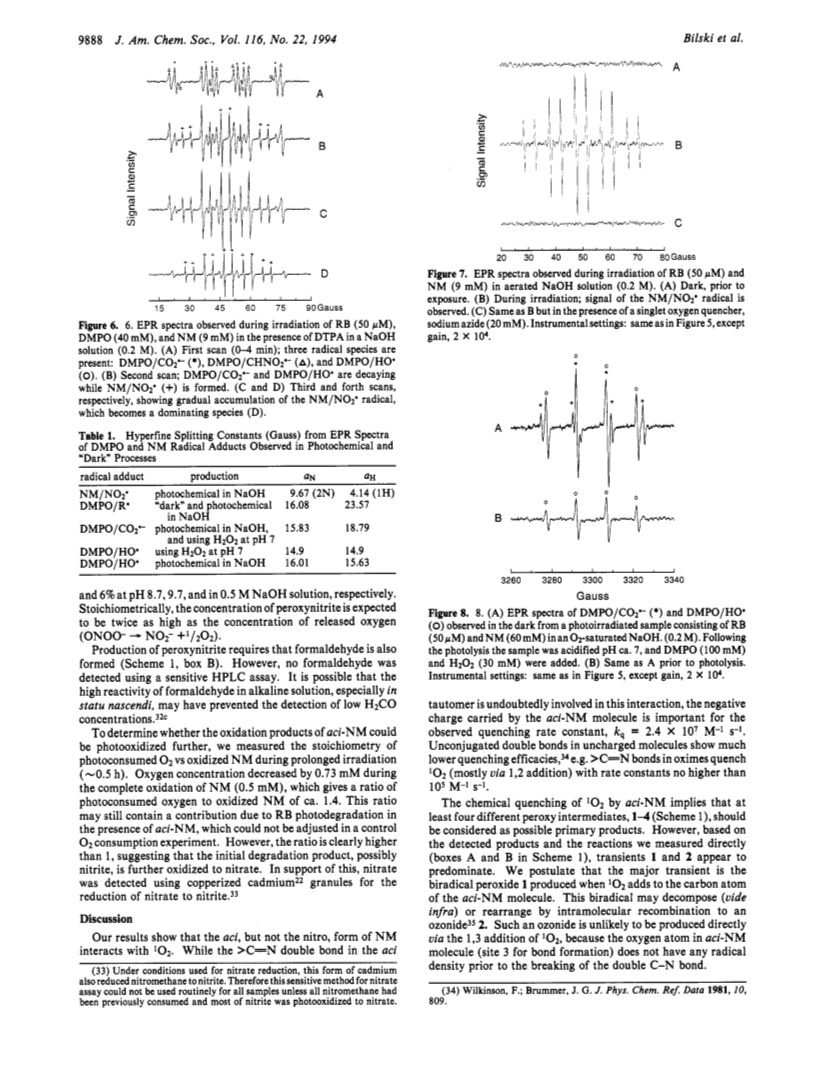
In alkaline solution nitromethane (NM) forms a stable act tautomer (H2C=NO2-) which may be used as a spin trap for radicals, including NO? and NO2?. We have noticed previously that in aerated photochemical systems NM can undergo photosensitized degradation (Bilski et al., J. Am. Chem. Soc. 1992, 114, 549), possibly via singlet oxygen (1O2). We have now confirmed that aci-NM does indeed quench 1O2 phosphorescence (kq = 2.4 × 107 M-1 s-1), and that the addition of 1O2 to aci-NM is solely responsible for this chemical quenching, as confirmed by the high quantum yield of O2 photoconsumption (φ = 0.65 for [NM] = 35 mM). To obtain "labeled" fragments from the decomposition of the resultant aci-NM-O2 peroxy species we have applied another spin trap, 5,5-dimethyl-1-pyrroline N-oxide (DMPO), in a novel way: we used an EPR silent hydroxylamine adduct (DMPOH/CH=NO2-) formed by nucleophilic addition of aci-NM to DMPO. Reaction of DMPOH/CH=NO2- with 1O2 resulted in the generation of the DMPO/CO2?- radical, suggesting that in the absence of DMPO the following mechanism may occur: 1O2 + CH2=NO2- → NO2- + HCOOH. We have found that nitrite and formate production accounts for most of the oxygen consumed. This suggests that 1O2 reacts mainly with the carbon atom of NM producing a biradical transient which decomposes yielding thermodynamically stable products nitrite and formate. A minor product is peroxynitrite (OONO-) which must be produced via an ozonide-type intermediate formed by intramolecular recombination of the biradical. Prolonged irradiation of Rose Bengal and aci-NM resulted in accumulation of nitrite which was then photooxidized to NO2?, trapped by unreacted aci-NM and identified as the NM/NO2? spin adduct.
View More



EASYBUYER Co.,LTD.( Chen-Chen )
Contact:+86-21-51816013/51816067
Address:1306,Bldg 8 Hi-Shanghai Center,No.950 Dalian Rd
Tianjin Emulsion Science&Technology Development Co.,Ltd
Contact:13901380442
Address:Vake Garden New Town New Yi Bai Road Beichen District Tianjin,China
Yangling Ciyuan biotech Co., Ltd.
Contact:86-15802970736
Address:2-1804, International Park Mansion, No.2, South Fengdeng Road, Lianhu District
Zibo Jujin Chemical Industry Co., Ltd.(Dongming Jujin Chemical Industry Co., Ltd. )
Contact:+86-533-2975022
Address:No.99 Shanquan Road, Zhangdian District
Shanghai Send Pharmaceutical Technology Co., Ltd.
website:http://www.shsendpharma.com
Contact:021-58088081, +8613585868794
Address::Room A601, Building 1,NO. 800 Qingdai Road Pudong District Shanghai,China
Doi:10.1016/j.phytochem.2011.01.013
(2011)Doi:10.1039/c5ob00078e
(2015)Doi:10.1515/znb-2005-1211
(2005)Doi:10.1039/c1dt10671f
(2011)Doi:10.1016/j.tet.2008.09.026
(2008)Doi:10.1016/j.tet.2003.08.067
(2003)