Green Chemistry
Paper
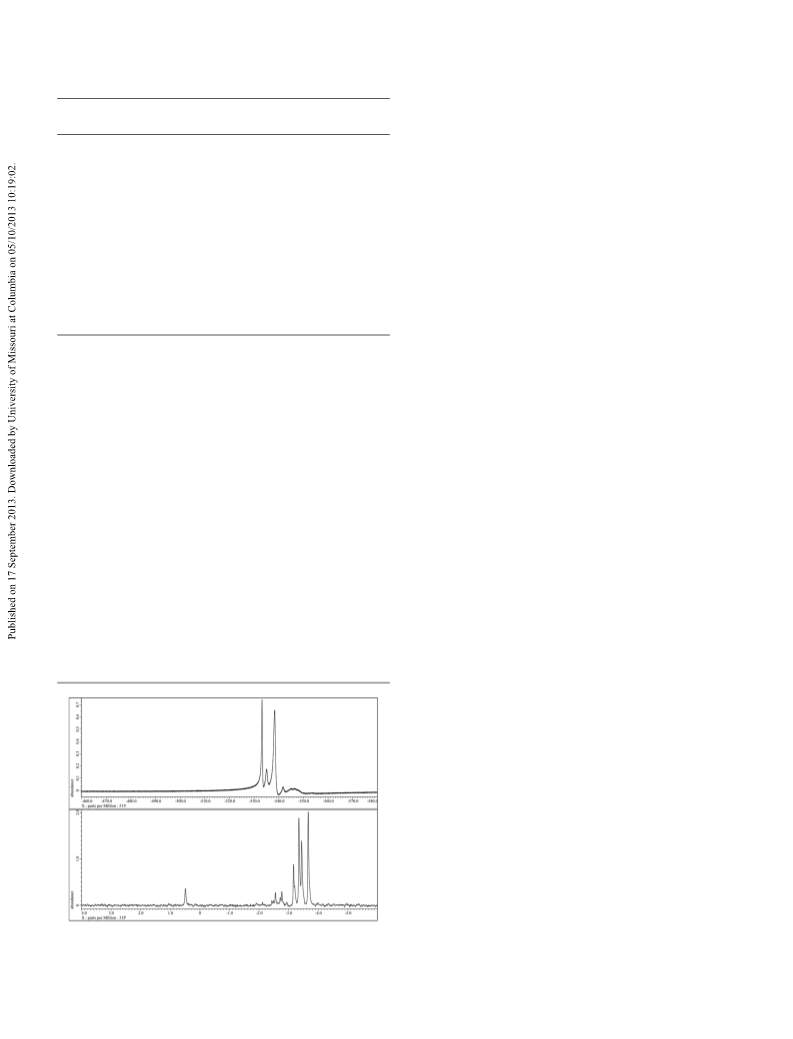
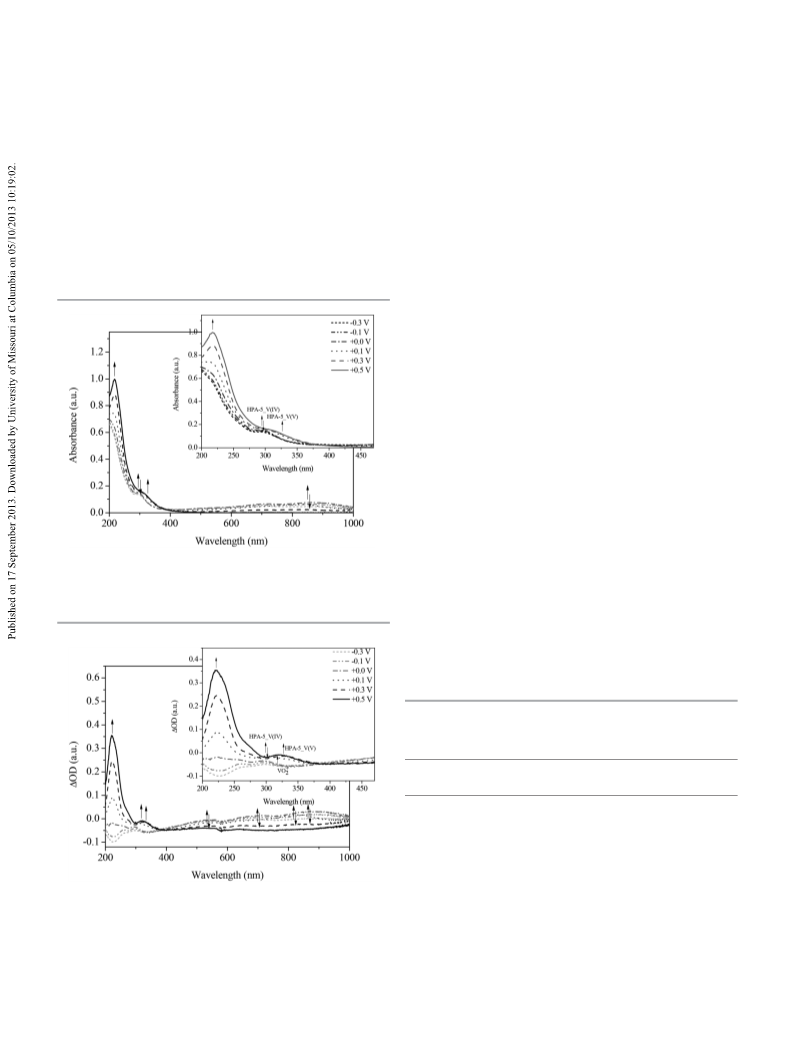
Optical spectroscopy
8 C. Tuck, E. Pérez, I. Horváth, R. Sheldon and M. Poliakoff,
Science, 2012, 337, 695.
9 J. Geboers, S. van de Vyver, K. Carpentier, P. Jacobs and
B. Sels, Green Chem., 2011, 13(8), 2167.
10 Y. Ogasawara, S. Itagaki, K. Yamaguchi and N. Mizuno,
ChemSusChem, 2011, 4(4), 519.
11 B. Op de Beeck, J. Geboers, S. van de Vyver, J. Van Lishout,
J. Snelders, W. Huijgen, C. Courtin, P. Jacobs and B. Sels,
ChemSusChem, 2013, 6, 199.
12 J. Geboers, S. van de Vyver, K. Carpentier, K. de Blochouse,
P. Jacobs and B. Sels, Chem. Commun., 2010, 46, 3577.
13 L. Wang, C. L. Weller, D. D. Jones and M. A. Hanna,
Biomass Bioenergy, 2008, 32, 573.
14 M. M. Yung, W. S. Jablonski and K. A. Magrini-Bair, Energy
Fuels, 2009, 23, 1874.
15 A. A. Petersen, F. Vogel, R. P. Lachance, M. Froling,
J. M. Antal and J. W. Tester, Energy Environ. Sci., 2008, 1, 32.
16 L. J. Guo, Y. J. Lu, X. M. Zhang, C. M. Ji, Y. Guan and
A. X. Pei, Catal. Today, 2007, 129, 275.
17 R. R. Davda, J. W. Shabaker, B. W. Huber, R. D. Cortright
and J. A. Dumesic, Appl. Catal., B, 2005, 56, 171.
18 B. M. Kabyemela, T. Adschiri, R. M. Malaluan and K. Arai,
Ind. Eng. Chem. Res., 1999, 38, 2888.
Infrared spectra of the solid catalysts were recorded using
FT-IR spectrophotometer Shimadzu Prestige-21 in combi-
nation with a diamond crystal ATR unit (L.O.T., Golden Gate)
between 4000 and 500 cm−1 region. Steady state UV-Vis
measurements of the catalysts in hydrochloric aqueous solu-
tion were performed using a Specord S 600 UV-Vis spectro-
meter (Analytic Jena) at room temperature in a 10 × 10 mm
quartz cuvette between 200 and 1000 nm.
Electrochemistry
Electrochemical data were obtained by cyclic voltammetry and
square wave voltammetry using a FRA 2 µAutolab Type III
Potentiostat/Galvanostat (with impedance unit) (Metrohm).
The usual three-electrode arrangement included a glassy
carbon electrode as a working electrode, Pt wire as a counter
electrode and a silver wire as a pseudo reference electrode, see
ESI.† The different voltammetric measurements were carried
out in argon saturated hydrochloric-aqueous solution in a
potential range between −0.60 and +1.00 V.
In situ UV-Vis electrochemistry
Spectroelectrochemical experiments were carried out using a 19 A. Kruse and A. Gawlik, Ind. Eng. Chem. Res., 2002, 42, 267.
PGSTAT 101 (Metrohm) and a Specord S 600 UV-Vis spectro- 20 S. van de Vyver, J. Geboers, P. Jacobs and B. Sels, Chem-
meter (Analytic Jena). The measurements were performed in a
quartz glass cuvette with an optical path length 1.0 mm in 21 H. Kobayashi, H. Ohta and A. Fukuoka, Catal. Sci. Technol.,
hydrochloric-aqueous solution under argon atmosphere. 2012, 2, 869.
Light-transparent platinum gauze served as a working elec- 22 J. Geboers, S. van de Vyver, R. Ooms, B. Op de Beeck,
CatChem, 2011, 3, 82.
trode, a silver wire as a reference electrode and a platinum
wire as a counter electrode.
P. Jacobs and B. Sels, Catal. Sci. Technol., 2011, 1, 714.
23 H. Kobayashi and A. Fukuoka, Green Chem., 2013, 15, 1740.
24 R. Wölfel, N. Taccardi, A. Bösmann and P. Wasserscheid,
Green Chem., 2011, 13, 2759.
25 J. Albert, R. Wölfel, A. Bösmann and P. Wasserscheid,
Energy Environ. Sci., 2012, 5, 7956.
Acknowledgements
The authors like to thank Dr Nicola Taccardi for performing 26 W. Reutemann and H. Kieczka, in Ullmann’s Encyclopedia
the ICP measurements. Additionally, we have to thank Patrick
Preuster and Stephanie Bajus for performing TGA analysis.
of Industrial Chemistry, Wiley-VCH Verlag GmbH & Co.
KGaA, Weinheim, 2005, DOI: 10.1002/14356007.a12_013.
The Energie Campus Nürnberg is gratefully acknowledged for 27 F. S. Dickson, J. Bakker and A. Kitai, “Acedic Acid” in Chemi-
financial support.
cal Economics Handbook, 1982.
28 W. Leitner, Angew. Chem., 1995, 107, 2391.
29 N. Taccardi, D. Assenbaum, M. E. M. Berger, A. Bösmann,
F. Enzenberger, R. Wölfel, S. Neuendorf, V. Goeke,
N. Schödel, H. J. Maass, H. Kistenmacher and
P. Wasserscheid, Green Chem., 2010, 12, 1150.
30 G. Mc Ginnis, S. Prince, C. Biermann and J. Lowrimore,
Carbohydr. Res., 1984, 128, 51.
Notes and references
1 G. Huber, S. Iborra and A. Corma, Chem. Rev., 2006, 106,
4044.
2 D. Alonso, J. Bond and J. Dumesic, Green Chem., 2010, 12,
1493.
31 K. Niemelä, Biomass, 1988, 15, 223.
3 J. Chheda, G. Huber and J. Dumesic, Angew. Chem., Int. Ed., 32 D. Yu, M. Aihara and M. Antal, Energy Fuels, 1993, 7, 574.
2007, 46, 7164.
33 F. Jin, J. Yun, G. Li, A. Kishita, K. Tohji and H. Enomoto,
Green Chem., 2008, 10, 612.
34 J. Li, D. J. Ding, Q. X. Guo and Y. Fu, ChemSusChem, 2012,
5, 1313.
35 A. Boddien, F. Gärtner, R. Jackstell, H. Junge,
A. Spannenberg, W. Baumann, R. Ludwig and M. Beller,
Angew. Chem., 2010, 122, 9177.
4 R. Palkovits, K. Tajvidi, A. Ruppert and J. Procelewska,
Chem. Commun., 2011, 47, 576.
5 D. Klemm, B. Heublein, H. Fink and A. Bohn, Angew.
Chem., Int. Ed., 2005, 44, 3358.
6 M. Holm and S. Saravanamurugan, Science, 2010, 328, 602.
7 P. Dhepe and A. Fukuoka, ChemSusChem, 2008, 1, 969.
This journal is © The Royal Society of Chemistry 2013
Green Chem.

 Albert, Jakob
Albert, Jakob
 Lueders, Daniela
Lueders, Daniela
 Boesmann, Andreas
Boesmann, Andreas
 Guldi, Dirk M.
Guldi, Dirk M.
 Wasserscheid, Peter
Wasserscheid, Peter