Slebocka-Tilk et al.
1349
–
1
hydroxide we assume in Scheme 1 that one of the solvating
waters has been lost to liberate a lone pair required for at-
tack on C=O, and that the developing O in TI is associated
with three solvating waters with fractionation factors similar
to those of hydroxide. We assume that the transition state for
estimated activation parameters of E = 22.5 kcal mol , ln A =
15.03 from a two-point Arrhenius plot.
a
–
–
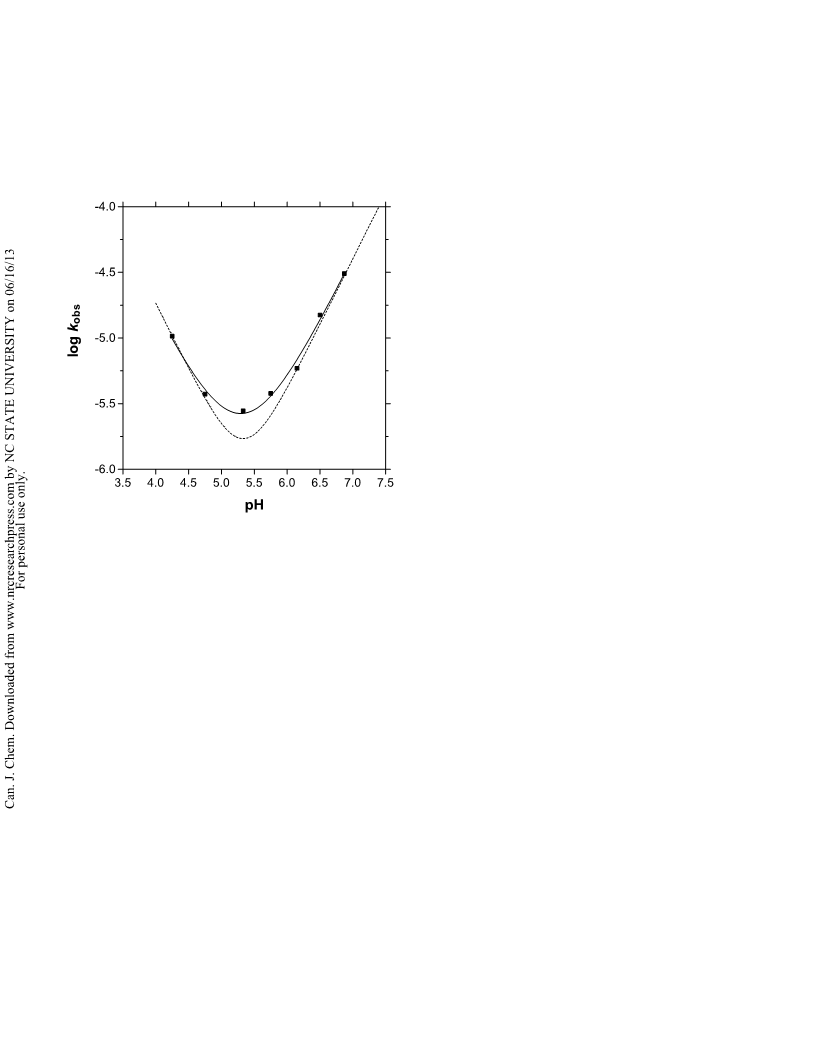
While it is apparent that a water reaction does exist at
56°C and at 120°C, in neither case does it contribute more
than about 40% to the observed rate at the respective pH
minima of 6.1 and 5.3. Hine et al. (14) also concluded that at
80°C, no more than 50% of the rate at the pH minimum can
be attributed to the water reaction. Taken together, these
studies indicate that it is improbable that conditions will be
found where the water term for hydrolysis of formamide can
be isolated (meaning contributing more than 95% at any
given pH) from the corresponding acid and base reactions.
This is an interesting consequence of two major factors: (1)
the relative activation parameters of all three processes for
this specific amide; and (2) the increase in the autoprotolysis
constant of water as a function of temperature that increases
attack (k ) is late, being ~70% along the reaction coordinate
1
to the tetrahedral species.
In the case of the general base mechanism given in
Scheme 2, we assume that the fractionation factor for the
proton in flight between the hydroxide and water is 0.4 (cor-
responding to a normal primary dkie of 2.5). All the as-
sumed values are shown in bold type in Schemes 1 and 2:
these give predicted solvent dkie’s of ~1.0 for the direct at-
tack of hydroxide and 2.2 for the general base mechanism.
The only substantial difference between the processes
shown in Schemes 1 and 2 concerns the presence of the pro-
ton in flight which is solely responsible for the predicted
normal dkie in the latter. Making the transition state in
Scheme 1 later gives only a small increase in the predicted
dkie because of the compensating effects of the loosening on
–
the [HO ] at any given pH, the net effect being to drive the
pH minimum for the reaction to lower values at higher tem-
peratures.
We have presented solvent deuterium kinetic isotope data
for the base-catalyzed reaction at high [HO ] where the ki-
–
–
waters solvating the attacking HO , and tightening of the
–
–
netics are largely determined by the first step leading to the
tetrahedral intermediate. The observed inverse value of
kOH/kOD = 0.77 gives little evidence for a significant contri-
bution of a proton in flight and strongly supports the mecha-
nism involving the direct attack of hydroxide, similar to that
believed operative for other amides, and at variance with a
waters solvating the developing O in TI . By converse, an
earlier transition state for the direct attack of hydroxide, say
–
5
0% along the way to TI , gives a predicted dkie of 0.85,
close to the experimentally observed value of 0.77 ± 0.06.
That value seems to rule out the pathway given in Scheme 2,
or any other pathway where there is much contribution from
one or more protons in flight. In the limit of that mecha-
nism, where the proton is nearly fully transferred from the
–
suggested water attack mediated by HO .
–
attacking water to the general base HO , its associated frac-
Acknowledgements
tionation factor would be close to unity leading to a drop in
the predicted dkie toward the observed value. In the end this
simply becomes a mechanism of direct hydroxide attack and
The authors gratefully acknowledge the financial support
of the Natural Sciences and Engineering Research Council
of Canada (NSERC) and Queen’s University. In addition
they acknowledge the generous donation of NMR time and
expertise by Bruker Canada. In addition they are grateful for
the helpful comments of the referees and for insightful per-
sonal communications from Professor J. P. Guthrie, Univer-
sity of Western Ontario.
6
cannot be distinguished from the process in Scheme 1.
Conclusions
The main goals of the above study were to provide infor-
mation about the water reaction of formamide, and provide
the solvent dkie for the base reaction. Several important con-
clusions can be advanced.
References
1
. (a) W.P. Jencks. In Catalysis in chemistry and enzymology.
McGraw-Hill Inc., New York. 1969. pp. 523–527; (b) T.H.
Lowry and K.S. Richardson. In Mechanism and theory in or-
ganic chemistry. 3rd. ed. Harper and Row Inc., New York.
1987. pp. 714–717; (c) C.J. O’Connor. Quart. Rev. Chem. Soc.
4, 553 (1970); (d) M.L. Bender. Chem. Rev. 60, 53 (1960);
e) N. Isaacs. In Physical organic chemistry. 2nd. ed. Longman
Scientific and Technical and J. Wiley and Sons, New York.
995. pp. 529–531; ( f ) J. March. In Advanced organic chem-
We have determined the activation parameters for the
+
–
H O and HO promoted hydrolyses of formamide. The ex-
3
‡
‡
–1
perimental ꢀH and ꢀ S values are (17.0 ± 0.4) kcal mol
and (–18.8 ± 1.3) cal mol–1
–1
+
K
for the H O catalyzed reaction
3
–
1
–1
–1
and (17.9 ± 0.2) kcal mol and (–11.1 ± 0.5) cal mol
for the HO process. The values fit within the general range
determined for acid- and base-catalyzed hydrolysis of other
simple amides. However, the ꢀH and ꢀ S values for the
HO process give an experimental ꢀG2 of 21.2 kcal mol
K
2
(
–
‡
‡
1
–
‡
5
–1
istry. 4th. ed. Wiley-Interscience, New York. 1992.
which is substantially lower than the recently computed
value of 27.3 kcal mol–1 (17) which should prompt further
efforts to refine the computational approaches.
2
. (a) R.S. Brown, A.J. Bennet, and H. Slebocka-Tilk. Acc.
Chem. Res. 25, 481 (1992); (b) A.J. Bennett and R.S. Brown.
In Comprehensive biological catalysis: A mechanistic refer-
ence. Vol. 1. Edited by M. Sinnott. Academic Press, New
York. 1997. pp 293–326; (c) R.S. Brown. In The amide link-
We have determined k values of (1.09 ± 0.29) × 10 s–1
–
6
w
–
9
–1
at 120°C and (3.6 ± 0.1) × 10
s
at 56°C, from which we
6
This analysis differs from the one used by Mata-Segreda in assessing the mechanism for hydroxide attack on ethyl acetate (25). In that anal-
–
ysis the assumed transition structure involved HO ···H-(OH)···C=O with no solvating waters and a proton in flight between the attacking
–
water and the general base HO . A future publication from these laboratories will present our analysis of the proton inventory study of the
base hydrolysis of formamide.
©
2002 NRC Canada

 Slebocka-Tilk
Slebocka-Tilk
 Sauriol
Sauriol
 Monette, Martine
Monette, Martine
 Brown
Brown