DOI: 10.1039/C4RA13285H
Page 3 of 4
RSC Advances
indicated that the Au-Pt NCs had a satisfactory tolerance toward
the carbonaceous species formed during the oxidation of
electrocatalytic activity over both methanol and formic acid
oxidation (Figure S3). When the growth temperature was
methanol, and were much more electroactive and stable during 50 increased to 90 °C, the electrocatalysis CVs for methanol and
the methanol oxidation reactions.
formic acid gradually changed (Figure S4-S8; Au/Pt 3/11-2/23
mol/mol, Table S1). Note that, the typical double peaks for these
two specimen electrocatalytic oxidations are formed when using
the NCs grown at 30 min or longer time, which evidently
55 revealed that the Pt nanobranch is very critical to trigger the
electrocatalytic activity of the NPs. A comparison of the NCs
grown at 5 h with those grown at 2 h indicated that there is an
optimum size of nanobranch for their electrocatalytic activity, i.e.,
the overwhelm longer nanobranch may decrease their
60 electrocatalytic activity, owing to the faded influence of Au NPs
(Figure S8).
In conclusion, we here reported a facile approach for the
synthesis of Au-Pt NCs with controllable composition and
morphologies. We also found an optimized Au-to-Pt molar ratio
65 of NCs, which ensures the most efficient eletrochemical activtiy
and stability. The synthesized NCs were found to have enhanced
electrocatalytic activity for direct oxidation of methanol and
formic acid, which is promising for application in fuel cells.
5
Figure 4. (A) CVs, (B) electrochemical impedance spectra (EIS), (C)
chronoamperometric results and (D) LSV curves of formic acid
oxidation by Au-Pt and commercial JM-Pt/C catalysts in N2-saturated
Acknowledgments
70 This work was financially supported by the National Natural
Science Foundation of China (No. 21373006, 51402203), Natural
Science Foundation of Jiangsu Province for Young Scholars
(BK20140326) and the Priority Academic Program Development
of Jiangsu Higher Education Institutions (PAPD).
75
0.5 M H2SO4 + 0.5 M HCOOH. Scan rate: 50 mVs−1
.
10
We also applied these NCs for electrocatalysis of formic acid
oxidation. Figure 4A shows typical CVs for this reaction. The
exhibited characteristic double anodic peaks in the forward and
reverse scans correspond to the typical feature for the oxidation
of formic acid. The first peak at 0.3 V on the CV is related to the
15 direct oxidation of HCOOH to form CO2 while the second peak
at 0.66 V is related to the oxidation of the CO generated from the
dissociative adsorption.25 The mass-normalized current density
for these two oxidations reveals that the latter mechanism
dominates this electrocatalysis for both NCs and the commercial
20 catalyst. In addition, for the current density of Au-Pt NCs (137.1
mA mg-1 Pt) is 1.41 times larger than that for the commercial
catalyst (97.5 mA mg-1 Pt), showing a significant increase in
mass current density.
aKey Laboratory of Organic Synthesis of Jiangsu Province, College of
Chemistry, Chemical Engineering and Materials Science & Collaborative
Innovation Center of Suzhou Nano Science and Technology, Soochow
80 University, Suzhou, China 215123.
bAnalysis and Testing Center, Soochow University, Suzhou, China 215123
Fax: +86-65880905; Tel: +86-65880905
†
Electronic Supplementary Information (ESI) available: [Details of
85 general exeprimental procedures]. See DOI: 10.1039/b000000x/
As shown in Figure 4B, the alternating current impedance of
25 the Au-Pt NCs is far smaller than that of the commercial JM-Pt/C
catalysts; this means that the intrinsic electrocatalytic activity of
the Au-Pt NCs is much lower than that of the commercial catalyst.
Figure 4C shows the electrochemical activity and durability of
the nanocrystals, as measured using chronoamperometry, in an
30 N2-saturated solution containing 0.5 M H2SO4 and 0.5M
HCOOH at a constant voltage of 0.65 V for 900 s. The current
decay for Au-Pt NCs is from 596.1 mA mg-1 Pt to 102.7 mA mg-1
Pt, higher than that of the commercial catalyst (from 179.4 mA
mg-1 Pt to 8.6 mA mg-1 Pt) over the whole duration of the
35 experiment. Besides, such NCs also exhibited the lowest potential
for the formic acid oxidation reaction at a given oxidation current
density. Besides, linear sweep voltammogram (LSV) curves also
reveal that the CO oxidation dominates the process and the
electroactivity is ~4.1 times higher than that of the commercial
40 catalyst, for this type of oxidation (Figure 4D). These results
indicated that the Au-Pt NCs had a satisfactory tolerance toward
the carbonaceous species formed during the oxidation of formic
acid, and were much more electroactive and stable during the
formic acid oxidation reactions.
Notes and references
1. S. Guo and E. Wang, Nano Today, 2011, 6, 240-264.
90 2. A. Chen and P. Holt-Hindle, Chem. Rev, 2010, 110, 3767-3804.
3. P. K. Jain, X. Huang, I. H. El-Sayed and M. A. El-Sayed, Acc. Chem.
Res., 2008, 41, 1578-1586.
4. J. Zhu, Y. Lu, Y. Li, J. Jiang, L. Cheng, Z. Liu, L. Guo, Y. Pan and H.
Gu, Nanoscale, 2014, 6, 199-202.
95 5. C. Wang, B. Peng, H.-N. Xie, H.-X. Zhang, F.-F. Shi and W.-B. Cai, J.
Phys. Chem. C, 2009, 113, 13841-13846.
6. R. Mu, Q. Fu, H. Xu, H. Zhang, Y. Huang, Z. Jiang, S. Zhang, D. Tan
and X. Bao, J. Am. Chem. Soc., 2011, 133, 1978-1986.
7. X. Yang, Q. Yang, J. Xu and C.-S. Lee, J. Mater. Chem., 2012, 22,
100
8057-8062.
8. C. Bartual‐Murgui, L. Salmon, A. Akou, N. A. Ortega‐Villar, H. J.
Shepherd, M. C. Muñoz, G. Moln á r, J. A. Real and A.
Bousseksou, Chem. Eur. J., 2012, 18, 507-516.
9. S. Zhou, G. S. Jackson and B. Eichhorn, Adv. Funct. Mater., 2007, 17,
3099-3104.
45
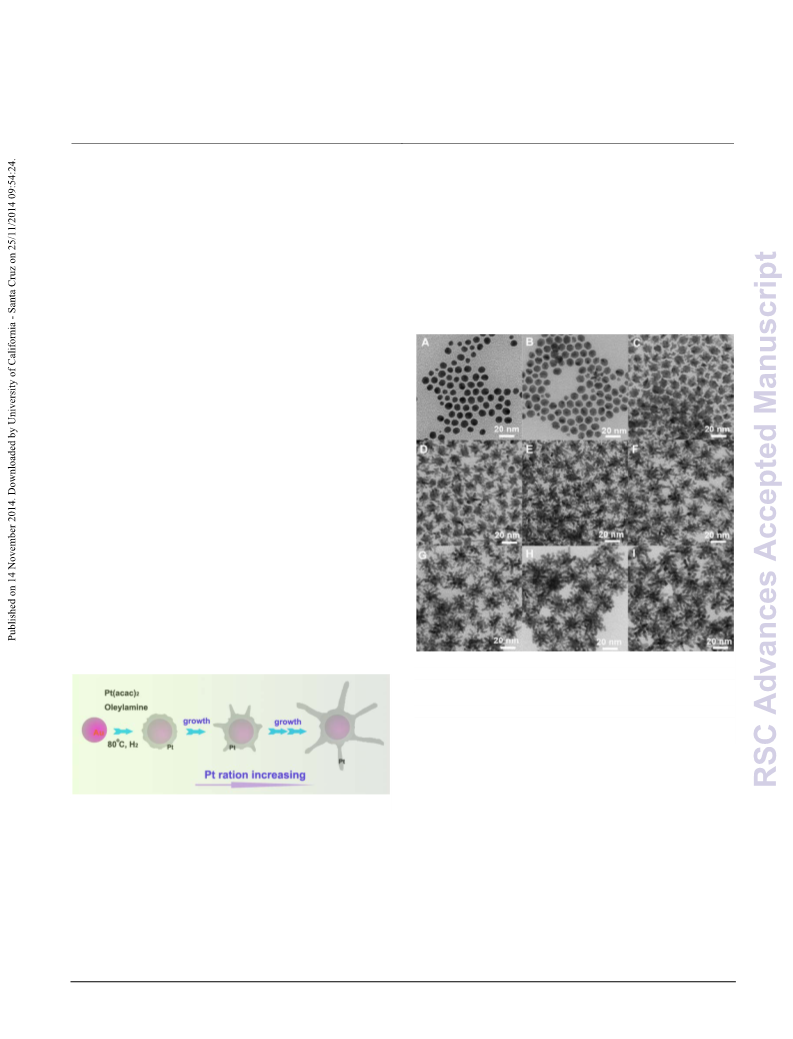
We also investigated the Au-Pt NCs grown under other
conditions. Firstly, the NCs grown at 80 °C exhibited no branches
(Au/Pt 211/18 mol/mol, Table S1), which induce the absence of
105
This journal is © The Royal Society of Chemistry [year]
Journal Name, [year], [vol], 00–00 | 3

 Xie, Ruigang
Xie, Ruigang
 Chen, Muzi
Chen, Muzi
 Wang, Jiaqing
Wang, Jiaqing
 Mei, Sujuan
Mei, Sujuan
 Pan, Yue
Pan, Yue
 Gu, Hongwei
Gu, Hongwei