Chromium(VI) Oxidation of L-Sorbose
J. Chin. Chem. Soc., Vol. 51, No. 2, 2004 407
therein. (b) Katz, S. A.; Salem, H. The Biological and Envi-
ronmental Aspects of Chromium; VCH: New York, 1994, pp.
65-119. (c) Cieslak-Golonka, M. Polyhedron 1996, 15,
3667. (d) Flora, S. De. Carcinogenesis 2000, 21, 533.
2. (a) Virtanen, P. O. I.; Lindroos-Heinanen, R. Acta. Chem.
Scand. Ser. 1988, B42, 411. (b) Gupta, M.; Saha, S. K.;
Banerjee, P. J. Chem. Soc. Perkin Trans II. 1985, 1781. (c)
Sengupta, K. K.; Basu, S. N. Carbohydr. Res. 1980, 80, 223.
(d) Sengupta, K. K.; Sengupta, S.; Basu, S. N. Carbohydr.
Res. 1979, 71, 75. (e) Sengupta, K. K.; Basu, S. N.
Carbohydr. Res. 1979, 72, 139. (f) Sengupta, K. K.; Bose, S.
N.; Sengupta, S. Carbohydr. Res. 1981, 97, 1. (g) Sala, L. F.;
Signorella, S. R.; Rizzotto, M.; Frascaroli, M. I.; Gandolfo,
F. Can. J. Chem. 1992, 70, 2046. (h) Rizzotto, M.;
Frascaroli, M. I.; Signorella, S.; Sala, L. F. Polyhedron 1996,
15, 1517. (i) Das, A. K.; Mondal, S. K.; Kar, D.; Das, M.
Inorg. React. Mech. 2001, 3, 63. (j) Das, A. K.; Roy, A.;
Saha, B.; Mohanty, R. K.; Das, M. J. Phys. Org. Chem. 2001,
14, 333. (k) Singh, S. V.; Saxena, O. C.; Singh, M. P. J. Am.
Chem. Soc. 1970, 92, 537. (l) Kumar, A.; Mehrotra, R. N. J.
Org. Chem. 1975, 40, 1248. (m) Daier, V.; Signorella, S.;
Frascaroli, M. I.; Palopoli, C.; Brondino, C.; Salas-Peregrin,
J. M.; Sala, L. F. Can. J. Chem. 1999, 77, 57. (n)
Kabir-ud-Din; Morshed, A. M. A.; Khan, Z. Inorg. React.
Mech. 2002, 3, 255. (o) Signorella, S.; Daier, V.; Garcia, S.;
Cargnello, R.; Gonzalez, J. C.; Rizzotto, M.; Sala, L. F.
Carbohydr. Res. 1999, 316, 14 and the references cited
therein.
3. (a) Das, A. K. Oxid. Commun. 2001, 24, 321 and the refer-
ences cited therein. (b) Lin, T. Y.; Chuo, C. M.; Zeng, H. W.
J. Chin. Chem. Soc. 1995, 42(1), 43. (c) Lin, T. Y. J. Chin.
Chem. Soc. 1981, 28(1), 21. (d) Lin, T. Y. J. Chin. Chem.
Soc. 1981, 28(3), 149. (e) Peng, T. Y.; Rocek, J. J. Am. Chem.
Soc. 1976, 98, 1026; 1977, 99, 7622. (f) Das, A. K.; Mondal,
S. K.; Kar, D.; Das, M. J. Chem. Res. (S) 1998, 574. (g) Das,
A. K.; Mondal, S. K.; Kar, D.; Das, M. Int. J. Chem. Kinet.
2001, 33, 173. (h) Mondal, S. K.; Das, M.; Kar, D.; Das, A.
K. Indian J. Chem. 2001, 40A, 352. (i) Srinivasan, C.;
Rajagopal, S.; Chellamani, A. J. Chem. Soc. Perkin Trans II.
1990, 1839.
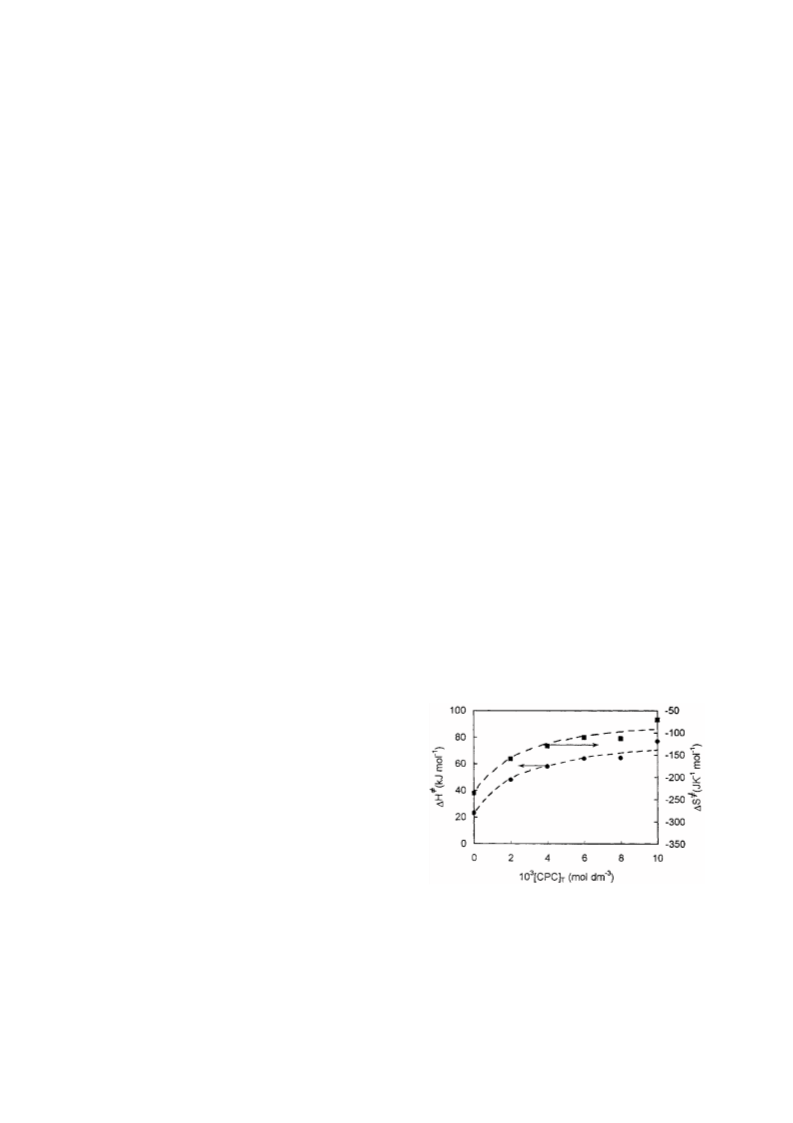
Here, kobs = observed rate constant in the presence of total
surfactant concentration denoted by [D]T; kw = observed rate
constant in the absence of surfactant; km = corresponding ob-
served rate constant in the micellar pseudo-phase; n is the in-
dex of cooperativity and KD gives the dissociation constant of
the micellized surfactant back to its components. The treat-
ment on the kinetic data does not require the knowledge of
cmc (critical micelle concentration) of the surfactant. In fact,
the cmc values of the surfactants are highly sensitive to the
reaction conditions. This is the advantage of the Piszkiewicz
model. Here it may be mentioned that although the present
equation was originally applied for the micelle catalysed re-
actions showing a maximum rate followed by inhibition, but
it has been applied in the cases20,21,23 where the micelles show
the rate retarding or enhancing effect throughout the range of
surfactant concentrations used. Here, for the CPC inhibited
reaction, km may be taken approximately as zero. By using the
equation, the different parameters n, log[D]50 (which indi-
cates the concentration of the surfactant needed for half-
maximum catalysis or inhibition) and logKD have been deter-
mined from the plot of log(P) vs. log[D]T (cf. Fig. 6). The
log[D]50 values obtained from this equation nicely agree with
those obtained experimentally. The values of n »1, far less
than the aggregation number (20-100) of the surfactants indi-
cate the formation of catalytically productive submicellar ag-
gregates.22 Thus, the reactant induced micellisation before
the cmc value is strongly hinted. In fact, the non-integral
value of n suggests the existence of multiple equilibria in the
formation of active submicellar aggregates at the premicellar
concentration range. When the interaction is fairly high
(logKD » 2.5), it is reasonable to consider n as representing
the average stoichiometry of the detergent-reactant aggre-
gate.
ACKNOWLEDGEMENTS
4. cf. (a) Virtanen, P. O. I.; Lindroos.-Heinanen, R.; Oikarinen,
E.; Vaskuri, J. Carbohydr. Res. 1987, 167, 29. (b) Virtanen,
P. O. I.; Pohjola, S. Finn. Chem. Lett. 1984, 155. (c)
Virtanen, P. O. I.; Kurkisuo, S.; Nevala, H.; Pohjola, S. Acta.
Chem. Scand. Sect. A 1986, 40, 200. (d) Das, A. K.; Roy, A.;
Saha, B. Transition. Met. Chem. 2001, 26, 630 (e) Lin, T. Y.;
Mao, Y. L.; Chuo, C. M. J. Chin. Chem. Soc. 1991, 38(2),
167.
The authors are thankful to CSIR, New Delhi and Visva
Bharati for providing financial assistance.
Received March 14, 2003.
5. Lyons, L. A Practical Guide to Data Analysis for Physical
Science Students; Cambridge University Press; 1991.
6. Ferrir, R. J.; Collins, P. M. Monosaccharide Chemistry; Pen-
guin: London, 1972; p 82.
7. (a) Feigl, F. Spot Tests in Organic Analysis; Elsevier: Am-
sterdam, 1956; p. 331. (b) Vishnoi, M.; Sharma, K.;
REFERENCES
1. (a) Codd, R.; Dillon, C. T.; Levina, A.; Lay, P. A. Coord.
Chem. Rev. 2001, 216-217, 537 and the references cited

 Saha, Bidyut
Saha, Bidyut
 Das, Mahua
Das, Mahua
 Mohanty, Rajani K.
Mohanty, Rajani K.
 Das, Asim K.
Das, Asim K.