RSC Advances
Paper
Foundation of Hebei Province (B2015204003, B2016204131),
the Young Top-notch Talents Foundation of Hebei Provincial
Universities (BJ2016027), the Natural Science Foundation
of Agricultural University of Hebei (LG201404, ZD201506,
ZD201613), are gratefully acknowledged.
Notes and references
1 H. L. Jiang, S. K. Singh, J. M. Yan, X. B. Zhang and Q. Xu,
ChemSusChem, 2010, 3, 541–549.
2 M. Yadav and Q. Xu, Energy Environ. Sci., 2012, 5, 9698–9725.
3 Y. Ping, J.-M. Yan, Z.-L. Wang, H.-L. Wang and Q. Jiang, J.
Mater. Chem. A, 2013, 1, 12188–12191.
4 H. Dai, B. Xia, L. Wen, C. Du, J. Su, W. Luo and G. Cheng,
Appl. Catal., B, 2015, 165, 57–62.
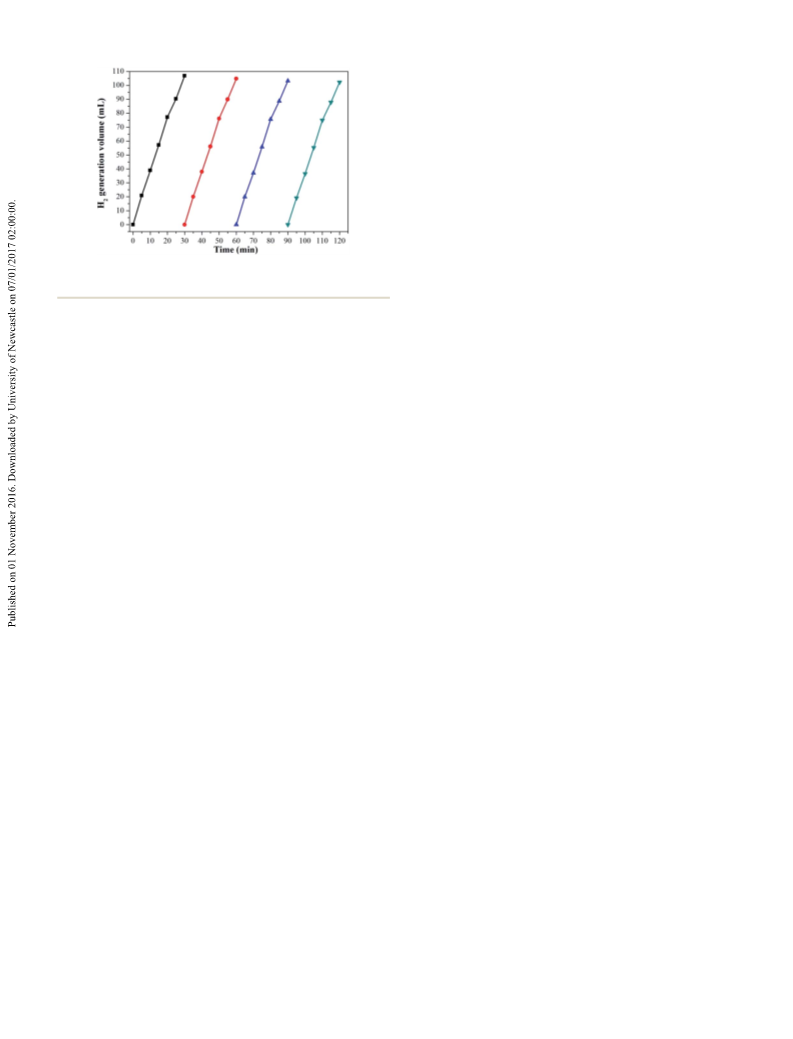
Fig. 8 Stability test on the Ag1Pd4@C-72 catalyst in the dehydroge-
nation of 0.26 M HCHO aqueous solution at 30 ꢁC.
5 Z.-L. Wang, J.-M. Yan, Y.-F. Zhang, Y. Ping, H.-L. Wang and
Q. Jiang, Nanoscale, 2014, 6, 3073–3077.
6 X. Gu, Z.-H. Lu, H.-L. Jiang, T. Akita and Q. Xu, J. Am. Chem.
Soc., 2011, 133, 11822–11825.
7 Z. L. Wang, J. M. Yan, Y. Ping, H. L. Wang, W. T. Zheng and
Q. Jiang, Angew. Chem., Int. Ed., 2013, 52, 4406–4409.
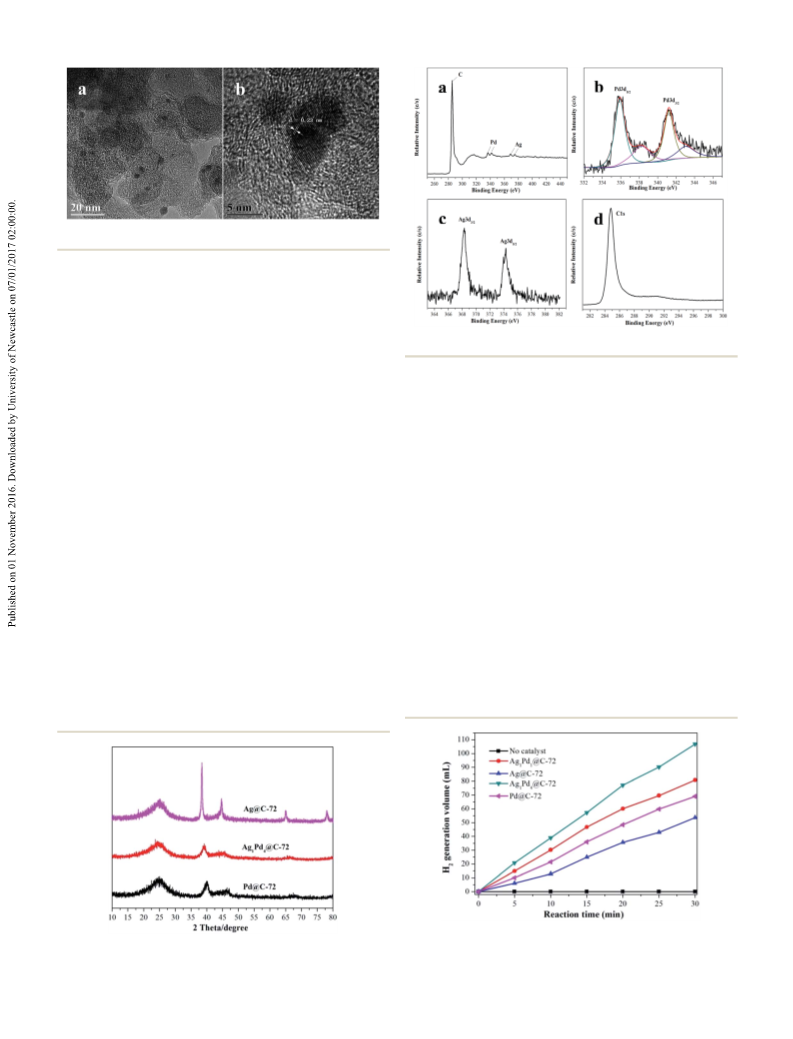
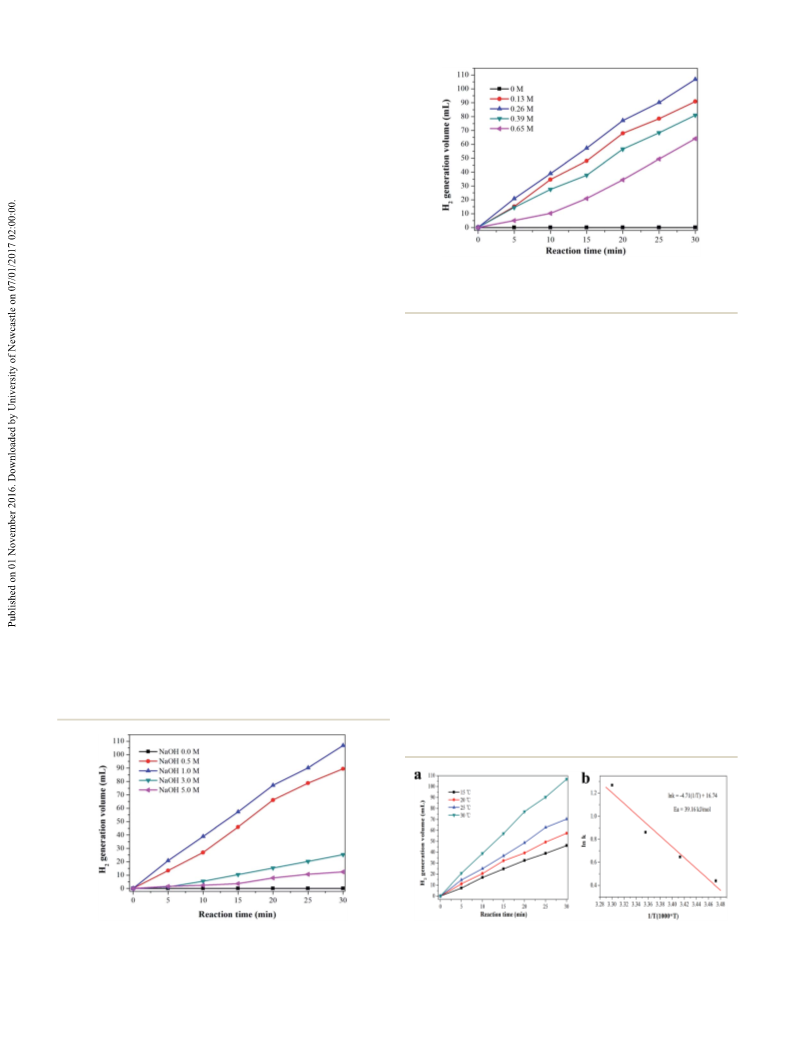
activity of Ag1Pd4@C-72 catalyst has no obvious decline aer
four runs, suggesting that the catalyst has a quite good stability,
which should attribute to the fact that loading AgPd nano-
particles on the carbon matrix could efficiently prevent the
agglomeration of metal nanoparticles. Thereby, Ag1Pd4@C-72
could efficiently catalyze formaldehyde to produce H2 with an
excellent catalytic activity and stability in alkaline aqueous
solutions.
A possible mechanism for the high catalytic activity of the
Ag1Pd4@C-72 is proposed. Firstly, formaldehyde is hydrated to
form methylene glycol in the presence of NaOH, then the
methylene glycol is transformed to hydrogen and sodium
formate catalyzed by Ag1Pd4@C-72.25 We think that the high
catalytic activity of the Ag1Pd4@C-72 may be attributed to the
AgPd nanoalloy structure, strong electron-donating effects of
Vulcan XC-72 carbon on Pd, and the high adsorption ability of
XC-72 carbon for formaldehyde.36
¨
˙
8 M. S. Izgi, O. ¸Sahin and C. Saka, Int. J. Hydrogen Energy, 2016,
41, 1600–1608.
9 H. Cai, P. Lu and J. Dong, Fuel, 2016, 166, 297–301.
10 J. Mahmood, S.-M. Jung, S.-J. Kim, J. Park, J.-W. Yoo and
J.-B. Baek, Chem. Mater., 2015, 27, 4860–4864.
11 T. Liu, Q. Wang, B. Yan, M. Zhao, W. Li and H. Bie, J.
Nanomater., 2015, 2015, 1–5.
12 J. Liao, H. Li, X. Zhang, K. Feng and Y. Yao, Catal. Sci.
Technol., 2016, 6, 3893–3899.
13 N.-Z. Shang, C. Feng, S.-T. Gao and C. Wang, Int. J. Hydrogen
Energy, 2016, 41, 944–950.
14 J. K. Sun and Q. Xu, ChemCatChem, 2015, 7, 526–531.
15 Q.-L. Zhu, D.-C. Zhong, U. B. Demirci and Q. Xu, ACS Catal.,
2014, 4, 4261–4268.
16 S.-J. Li, Y. Ping, J.-M. Yan, H.-L. Wang, M. Wu and Q. Jiang, J.
Mater. Chem. A, 2015, 3, 14535–14538.
Conclusions
In this work, a novel bimetallic catalyst, the AgPd nanoalloy
supported on Vulcan XC-72 carbon, has been successfully fabri-
cated and applied as a stable and low-cost catalyst for H2 gener-
17 X. Yang, P. Pachfule, Y. Chen, N. Tsumori and Q. Xu, Chem.
Commun., 2016, 52, 4171–4174.
ation from formaldehyde aqueous solution at room temperature. 18 C. Feng, Y. Wang, S. Gao, N. Shang and C. Wang, Catal.
The results show that the Ag1Pd4@C-72 catalyst exhibit high
Commun., 2016, 78, 17–21.
catalytic activity and good stability. The hydrogen generation 19 Q. Lv, L. Feng, C. Hu, C. Liu and W. Xing, Catal. Sci. Technol.,
rates could reach up to 237.4 mL minꢀ1 gꢀ1, which could attri-
2015, 5, 2581–2584.
bute to the synergistic effect of bimetal AgPd, distinct interaction 20 L. E. Heim, N. E. Schlorer, J.-H. Choi and M. H. Prechtl, Nat.
¨
between bimetal and support, and high dispersion of metal
nanoparticles. The excellent performance of the new catalyst
Commun., 2014, 5, 1–8.
21 Y. Bi and G. Lu, Int. J. Hydrogen Energy, 2008, 33, 2225–2232.
renders it quite attractive as a superior competitor for efficient 22 H. Gao, J. Zhang, R. Wang and M. Wang, Appl. Catal., B,
hydrogen production from formaldehyde. This work might open
2015, 172, 1–6.
up a new way to further develop cost-effective and highly efficient 23 Y. Bi, H. Hu, Q. Li and G. Lu, Int. J. Hydrogen Energy, 2010, 35,
bimetallic catalysts for the generation of H2 from formaldehyde
7177–7182.
aqueous solution to meet the requirement of practical applica- 24 X. Pan, L. Wang, F. Ling, Y. Li, D. Han, Q. Pang and L. Jia, Int.
tion of formaldehyde as a H2 storage/generation material.
J. Hydrogen Energy, 2015, 40, 1752–1759.
25 Y. Li, T. Chen, T. Wang, Y. Zhang, G. Lu and Y. Bi, Int. J.
Hydrogen Energy, 2014, 39, 9114–9120.
26 H. Hu, Z. Jiao, J. Ye, G. Lu and Y. Bi, Nano Energy, 2014, 8,
103–109.
Acknowledgements
Financial supports from the National Natural Science Founda-
tion of China (31671930, 21603054), the Natural Science 27 S. Li, H. Hu and Y. Bi, J. Mater. Chem. A, 2016, 4, 796–800.
105642 | RSC Adv., 2016, 6, 105638–105643
This journal is © The Royal Society of Chemistry 2016

 Gao, Shutao
Gao, Shutao
 Feng, Tao
Feng, Tao
 Wu, Qiuhua
Wu, Qiuhua
 Feng, Cheng
Feng, Cheng
 Shang, Ningzhao
Shang, Ningzhao
 Wang, Chun
Wang, Chun