Article
J. Agric. Food Chem., Vol. 57, No. 15, 2009 7123
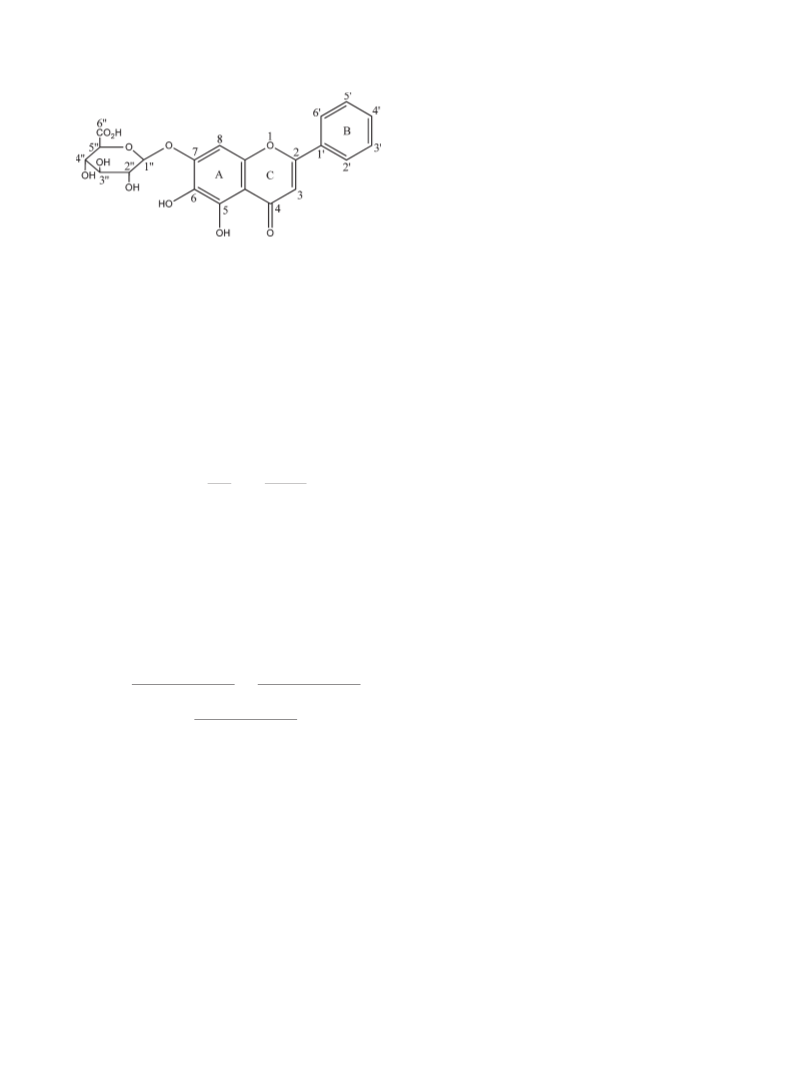
macroscopic property (29). Taken together, the differences in
chemical structures between baicalin and baicalein, the effects of
7-O-glycoside moiety on the conformation of flavones backbone,
and its steric hindrance against oxidative peroxyl radicals may all
account for the difference in the radical scavenging activities
between these two flavonoids.
(8) Wang, Q.; Wang, Y. T.; Pu, S. P.; Zheng, Y. T. Zinc coupling
potentiates anti-HIV-1 activity of baicalin. Biochem. Biophys. Res.
Commun. 2004, 324, 605–610.
(9) Li, B. Q.; Fu, T.; Yan, Y. D.; Baylor, N. W.; Ruscetti, F. W.; Kung,
H. F. Inhibition of HIV infection by baicalin;a flavonoid
compound purified from Chinese herbal medicine. Cell. Mol. Biol.
Res. 1993, 39, 119–124.
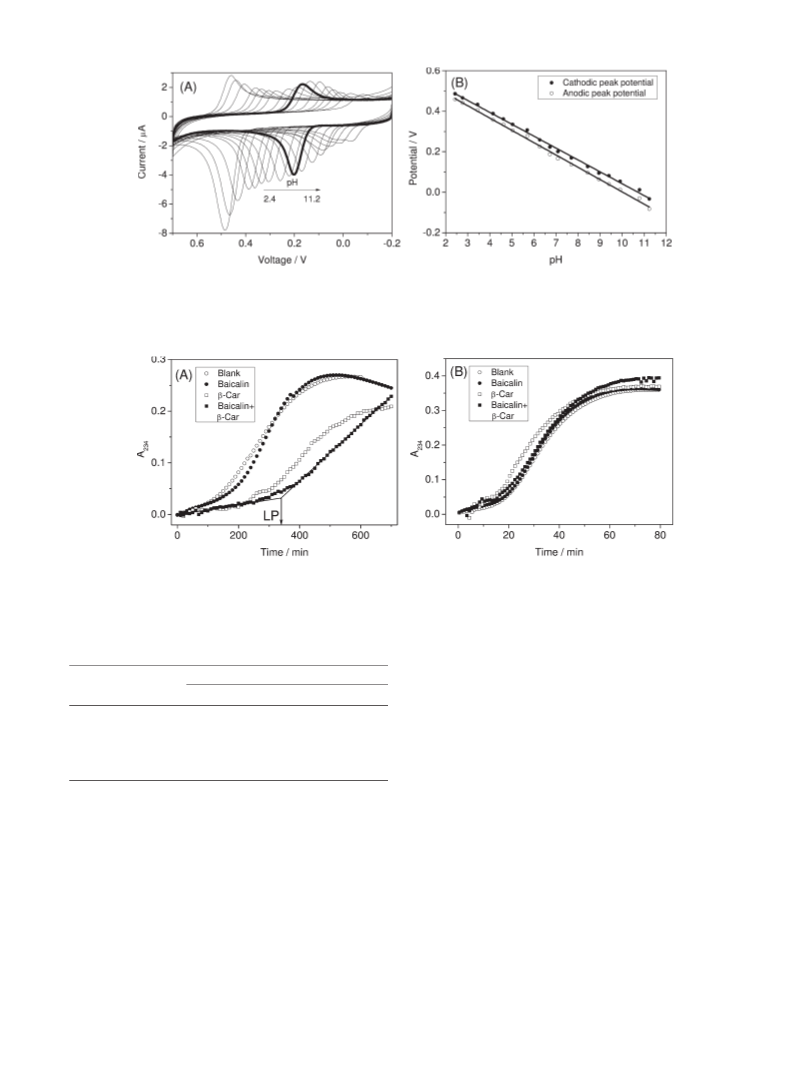
In summary, we have systematically investigated the physico-
chemical properties, the radical scavenging, and antilipoxida-
tion activities of baicalin, as well as the kinetics of regenerating
β-Car from β-Car•þ by baicalin. Baicalin as a glycoside-bearing
flavonoid is shown to be lipophilic and very reducing, but it is
of rather low radical scavenging activity and is almost ineffec-
tive in the retardation of AAPH- or AMVN-induced lipid
peroxidation. In homogeneous solution the conjugated bases
of baicalin are found to be capable of regenerating β-Car, whereas
neutral baicalin cannot. The present work has proven that
baicalin by itself shows little antilipoxidation activity; however,
it becomes very effective when combined with β-Car owing to
the synergistic interaction between its conjugated bases and β-Car
in liposomal membrane. The influence of the 7-O-glycoside
moiety on the radical scavenging activities of baicalin has been
discussed with reference to its metabolite baicalein, and the
radical scavenging and antilipoxidation properties of baicalin
have been discussed in terms of its physicochemical properties
and molecular structures.
(10) Kitamura, K.; Honda, M.; Yoshizaki, H.; Yamamoto, S.; Nakane,
H.; Fukushima, M.; Ono, K.; Tonkunaga, T. Baicalin, an inhibitor
of HIV-1 production in vitro. Antiviral Res. 1998, 37, 131–140.
(11) Park, S. W.; Lee, C. H.; Kim, Y. S.; Kang, S. S.; Jeon, S. J.; Son,
K. H.; Lee, S. M. Protective effect of baicalin against carbon
tetrachloride-induced acute hepatic injury in mice. J. Pharmacol.
Sci. 2008, 106, 136–143.
(12) Zhang, X. P.; Tian, H.; Chen, H. Q.; Chen, L.; Wang, Z. W.;
Wang, K. Y.; Yan, W.; Li, Y.; Li, Q. Y.; He, Q.; Wang, F. The
protecting effects and mechanisms of baicalin and octreotide on
heart injury in rats with SAP. Mediators Inflamm. 2007, 2007, 19469–
19479.
(13) Zhang, X. P.; Tian, H.; Lai, Y. H.; Chen, L.; Zhang, L.; Cheng,
Q. H.; Yan, W.; Li, Y.; Li, Q. Y.; He, Q.; Wang, F. Protective effects
and mechanisms of baicalin and octreotide on renal injury of rats
with severe acute pancreatitis. World J. Gastroenterol. 2007, 13,
5079–5089.
(14) Han, R. M.; Tian, Y. X.; Becker, E. M.; Andersen, M. L.; Zhang,
J. P.; Skibsted, L. H. Puerarin and conjugate bases as radical
scavengers and antioxidants: molecular mechanism and synergism
with β-carotene. J. Agric. Food Chem. 2007, 55, 2384–2391.
(15) Tian, Y. X.; Han, R. M.; Fu, L. M.; Zhang, J. P.; Skibsted,
L. H. Radical dynamics of puerarin as revealed by laser flash
photolysis and spin density analysis. J. Phys. Chem. B 2008, 112,
2273–2280.
ABBREVIATIONS USED
AAPH, 2,20-azobis(2-methylpropionamidine) dihydrochlor-
ide; AMVN, 2,20-azobis(2,4-dimethylvaleronitrile); ABTS, 2,
20-azinobis(3-ethylbenzothiazoline-6-sulfonic acid); β-Car, β-car-
otene; DMSO, dimethyl sulfoxide; TEAC, Trolox equivalent
(16) Tian, Y. X.; Han, R. M.; Wang, P.; Wu, Y. S.; Zhang, J. P.; Skibsted,
L. H. Puerarin as an antioxidant fluorescence probe. Chem. Phys.
Lett. 2008, 452, 253–258.
antioxodation capacity; PC, -R-phosphatidylcholine.
L
(17) McKenzie, H. A.; Dawson, R. M. C. pH, buffers, and physiological
media. In Data for Biological Research, 2nd ed.; Clarendon Press:
Oxford, U.K., 1974; pp 475-507.
ACKNOWLEDGMENT
(18) Albert, A.; Serjeant, E. P. Ionization Constants of Acids and Bases: A
We thank C.-H. Chen and C.-B. An for their support in the LP
experiments. Continuing support from LMC, Centre for Ad-
vanced Food Studies to the Food Chemistry group at University
of Copenhagen, is acknowledged.
Laboratory Manual; Wiley: Chichester, U.K., 1962.
(19) Arts, M. J. T. J.; Dallinga, J. S.; Voss, H. P.; Haenen, G. R. M. M.;
Bast, A. A new approach to assess the total antioxidant capacity
using the TEAC assay. Food Chem. 2004, 88, 567–570.
(20) Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-
Evans, C. Antioxidant activity applying an improved ABTS radical
cation decolorization assay. Free Radical Biol. Med. 1999, 26, 1232–
1237.
(21) Roberts, W. G.; Gordon, M. H. Determination of the total anti-
oxidant activity of fruits and vegetables by a liposome assay. J. Agric.
Food Chem. 2003, 51, 1486–1493.
(22) Hwang, J.; Sevanian, A.; Hodis, H. N.; Ursini, F. Synergistic
inhibition of LDL oxidation by phytoestrogens and ascorbic acid.
Free Radical Biol. Med. 2000, 29, 79–89.
(23) Han, R. M.; Tian, Y. X.; Wu, Y. S.; Wang, P.; Ai, X. C.; Zhang, J. P.;
Skibsted, L. H. Mechanism of radical cation formation from the
excited states of zeaxanthin and astaxanthin in chloroform. Photo-
chem. Photobiol. 2006, 82, 538–546.
(24) Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb,
M. A.; Cheeseman, J. R.; Montgomery, J. A., Jr.; Vreven, T.; Kudin,
K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.;
Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.;
Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.;
Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.;
Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H.
P.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.;
Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.;
Ochterski, J. W.; Ayala, P. Y.; Morokuma, K.; Voth, G. A.;
Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.;
Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck,
A. D.; Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.;
Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.;
LITERATURE CITED
(1) Hollman, P. C. H.; Katan, M. B. Dietary flavonoids: intake, health
effects and bioavailability. Food Chem. Toxicol. 1999, 37, 937–942.
(2) Galati, G.; O’Brien, P. J. Potential toxicity of flavonoids and
other dietary phenolics: significance for their chemopreventive
and anticancer properties. Free Radical Biol. Med. 2004, 37, 287–
303.
(3) Chen, G.; Zhang, H.; Ye, J. Determination of baicalein, baicalin and
quercetin in Scutellariae radix and its preparations by capillary
electrophoresis with electrochemical detection. Talanta 2000, 53,
471–479.
(4) Shi, H.; Zhao, B.; Xin, W. Scavenging effects of baicalin on free
radicals and its protection on erythrocyte membrane from free
radical injury. Biochem. Mol. Biol. Int. 1995, 35, 891–894.
(5) Shieh, D. E.; Liu, L. T.; Lin, C. C. Antioxidant and free radical
scavenging effects of baicalein, baicalin and wogonin. Anticancer
Res. 2000, 20, 2861–2865.
(6) Kim, Y. S.; Kim, J. J.; Cho, K. H.; Jung, W. S.; Moon, S. K.; Park, E.
K.; Kim, D. H. Biotransformation of ginsenoside Rb1, crocin,
amygdalin, geniposide, puerarin, ginsenoside Re, hesperidin, pon-
cirin, glycyrrhizin, and baicalin by human fecal microflora and its
relation to cytotoxicity against tumor cells. J. Microbiol. Biotechnol.
2008, 18, 1109–1114.
(7) Gao, Z.; Huang, K.; Yang, X.; Xu, H. Free radical scavenging and
antioxidant activities of flavonoids extracted from the radix of
Scutellaria baicalensis Georgi. Biochim. Biophys. Acta 1999, 1472,
643–650.

 Liang
Liang
 Rui-Min
Rui-Min
 Li-Min
Li-Min
 Xi-Cheng
Xi-Cheng
 Zhang, Jian-Ping
Zhang, Jian-Ping
 Skibsted, Leif H.
Skibsted, Leif H.