2 (a) R. T. Bucher, C. P. Landee, M. M. Turnbull and F. Xiao, Inorg. Chim.
Acta, 2008, 361, 3658; (b) M. Conner, A. McConnell, J. Schleuter and
J. L. Manson, J. Low Temp. Phys., 2006, 142, 277.
3 R. C. E. Belford, D. E. Fenton and M. R. Truter, J. Chem. Soc., Dalton
Trans., 1974, 17.
4 (a) H. W. Richardson and W. E. Hatfield, J. Am. Chem. Soc., 1976,
98, 835; (b) A. Santoro, A. D. Mighell and C. W. Reimann, Acta
Crystallogr., 1970, B26, 979; (c) J. S. Valentine, A. J. Silverstein and
Z. G. Soos, J. Am. Chem. Soc., 1974, 96, 93; (d) D. B. Losee, H. W.
Richardson and W. E. Hatfield, J. Chem. Phys., 1973, 59, 3600.
5 M. S. Haddad, D. N. Hendrickson, J. P. Cannady, R. S. Drago and D.
S. Bieksza, J. Am. Chem. Soc., 1979, 101, 898.
H. Stone, S. H. Lapidus and P. W. Stephens, J. Appl. Crystallogr., 2009,
42, 385.
18 J. D. Bernal and R. H. Fowler, J. Chem. Phys., 1933, 1, 515.
19 (a) M. A. Harvey, S. Baggio, M. T. Garland and R. Baggio, Acta Cryst.
C, 2008, 64, o489; (b) M. A. Harvey, S. Baggio, M. T. Garland and R.
Baggio, J. Coord. Chem., 2005, 58, 243; (c) A. Singh, R. P. Sharma, V.
Ferretti, S. Rossetti and P. Venugopalan, J. Mol. Struct., 2009, 927, 111;
(d) L. S. Skogareva and N. A. Minaeva, Russ. J. Inorg. Chem., 2001,
46, 1636; (e) L. S. Skogareva, L. Kh. Minacheva, V. S. Sergienko, N.
A. Minaeva and T. V. Filippova, Russ. J. Inorg. Chem., 2004, 49, 938;
(f) L. S. Skogareva, N. A. Minaeva and T. V. Filippova, Russ. J. Inorg.
Chem., 2009, 54, 1341.
6 J. Darriet, M. S. Haddad, E. N. Deusler and D. N. Hendrickson, Inorg.
Chem., 1979, 18, 2679.
7 W. Grochala and R. Hoffmann, Angew. Chem., Int. Ed., 2001, 40, 2743.
8 (a) P. C. Leung and F. Aubke, Inorg. Chem., 1978, 17, 1765; (b) O. Ruff
and W. Giese, Chem. Ber., 1936, 69B, 598; (c) S. M. Cortez and R. M.
Raptis, Coord. Chem. Rev., 1998, 169, 363.
9 (a) B. Standke and M. Jansen, Angew. Chem., Int. Ed. Engl., 1986, 25,
77; (b) P. C. Leung, K. C. Lee and F. Aubke, Can. J. Chem., 1979,
57, 326; (c) P. J. Malinowski, M. Derzsi, Z. Mazej, Z. Jaglicˇic´, P. J.
Leszczyn´ski, T. Michałowski and W. Grochala, Eur. J. Inorg. Chem.,
2011, 2499.
10 P. J. Malinowski, M. Derzsi, B. Gaweł, W. Łasocha, Z. Jaglicˇic´, Z. Mazej
and W. Grochala, Angew. Chem., Int. Ed., 2010, 49, 1683.
11 (a) K. M. Mertes, Inorg. Chem., 1978, 17, 49; (b) R. P. Eckberg and
W. E. Hatfield, Inorg. Chem., 1975, 14, 1205; (c) D. Yamamoto and F.
Ozeki, Bull. Chem. Soc. Jpn., 1972, 45, 1408.
12 R. W. Matthews and R. A. Walton, Inorg. Chem., 1971, 10, 1433.
13 J. L. Manson, K. H. Stone, H. I. Southerland, T. Lancaster, A. J. Steele,
S. J. Blundell, F. L. Pratt, P. J. Baker, R. D. McDonald, P. Sengupta,
J. Singleton, P. A. Goddard, Ch. Lee, M.-H. Whangbo, M. M. Warter,
Ch. H. Mielke and P. W. Stephens, J. Am. Chem. Soc., 2009, 131, 4590.
14 The Ag–Ag–Ag angles are: 83.6◦, 86.5◦, and 88.2◦. The unit cell may
be transformed to the quasi-tetragonal one using the matrix (101, 020,
-101); the resulting cell has Z = 16 and b = 96.32◦.
15 (a) H. Po, Coord. Chem. Rev., 1976, 20, 171; (b) J. C. Evans, R. D.
Gillard, R. J. Lancashire and P. H. Morgan, J. Chem. Soc., Dalton
Trans., 1980, 1277.
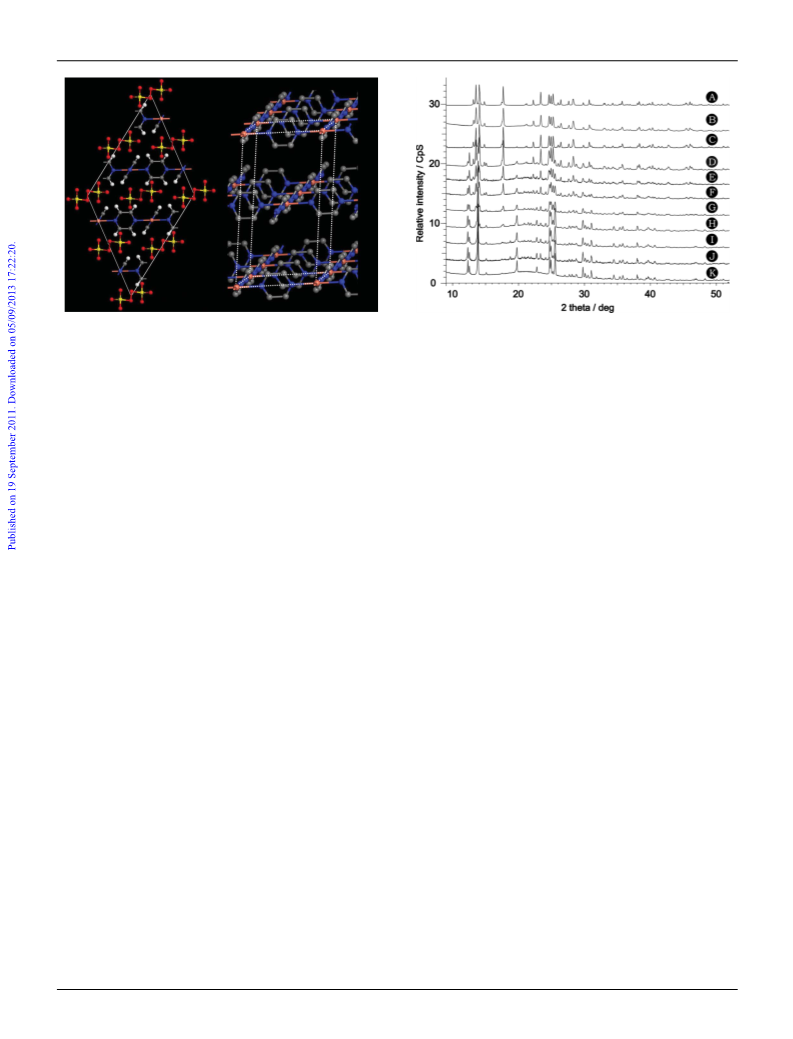
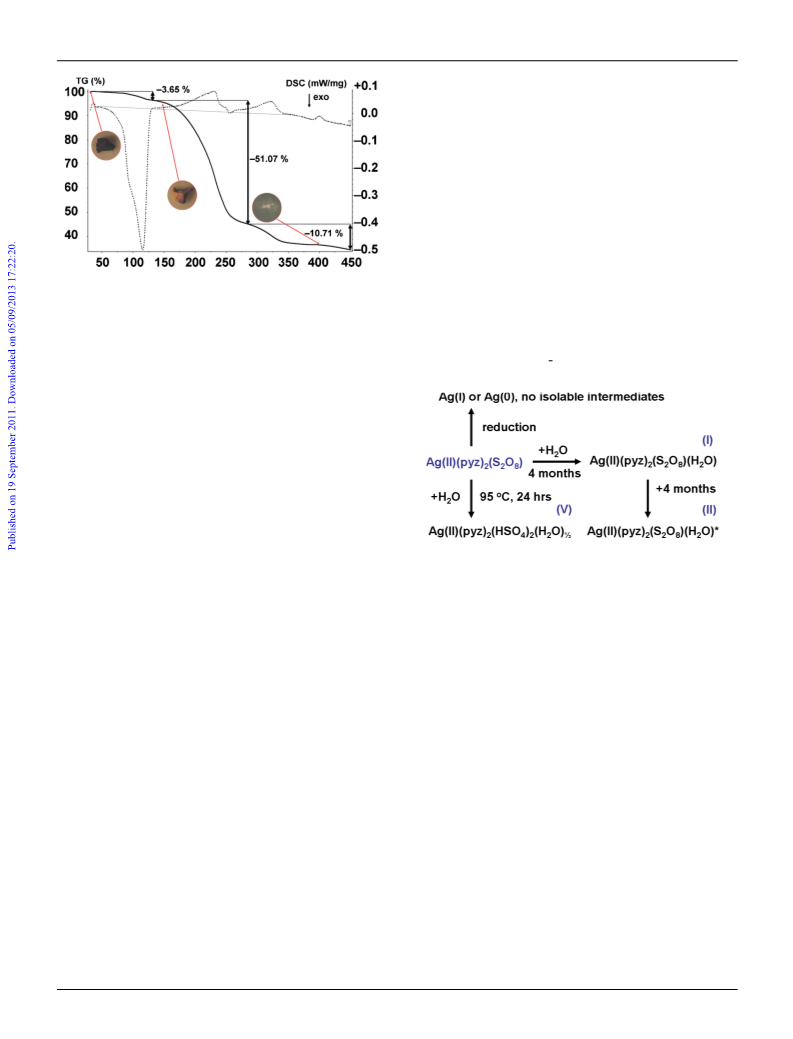
16 The changes in XRDP are independent of the synthetic pathway
used to prepare the title compound, which indicates that a slow but
spontaneous chemical decomposition takes place.
20 U. C. Singh and K. Venkatarao, Int. J. Chem. Kinet., 1981, 13, 555.
21 A. Budzianowski, M. Derzsi, P. J. Leszczyn´ski, M. K. Cyran´ski and W.
Grochala, Acta Cryst. B, 2010, 66, 451.
22 In this reaction H2SO4 is eliminated to balance the equation. A more
realistic equation necessitates even more water as substrate to yield
stable H2SO4 hydrates.
23 C. L. Perrin, Acc. Chem. Res., 2010, 43, 1550.
24 Y. Noda, S. Uchiyama, K. Kafuku, H. Kasatani, H. Terauchi and J.
Phys, J. Phys. Soc. Jpn., 1990, 59, 2804.
25 The auxiliary Ag(I) . . . O interactions are present in
a similar
[Ag(I)(pyz)](NO3): R. G. Vranka and E. L. Amma, Inorg. Chem., 1966,
5, 1020.
26 D. Sun, C. –F. Yang, H. –R. Xu, H. –X. Zhao, Z. –H. Wei, N. Zhang,
L. –J. Yu, R. Huang and L. Zheng, Chem. Commun., 2010, 46, 8168.
27 See: T. Buch, J. Chem. Phys., 1965, 43, 761, and references therein.
28 L. Carlucci, G. Ciani, D. M. Prosperio and A. Sironi, Angew. Chem.,
Int. Ed. Engl., 1995, 34, 1895.
29 (a) W. Grochala, J. Mater. Chem., 2009, 19, 6949; (b) W. Grochala, J.
Mol. Model., 2011, 17, 2237.
30 It is surprising that reaction between Ag(II)(pyz)2(S2O8) and Hg metal
(liquid or vapour) seems not to occur even after a prolonged reaction
time. We found that a small droplet of mercury generating dense
vapour helps to protect Ag(II)(pyz)2(S2O8) from decomposition in the
atmosphere.
31 G. Bhosekar, I. Jess and C. Na¨ther, Inorg. Chem., 2006, 45, 6508.
32 C. Na¨ther, I. Jess and J. Greve, Polyhedron, 2001, 20, 1017.
33 See: (a) J. E. House and C. Flentge, Thermochim. Acta, 1978, 24, 117;
(b) V. M. Gorbachev, F. R. Verzhbitsky, K. G. Myakishev and O. G.
Potapova, J. Therm. Anal., 1984, 29, 19.
34 F. A. Cotton, B. A. Frenz and D. L Hunter, Acta Cryst. B, 1975, 31,
302.
35 R. C. Keen, Z. Kristallogr., 1935, 91, 129.
36 Despite its aromatic character, and lack of susceptibility to oxidation,
pyrazine carries substantial spin density when attached to powerful
Ag(II) oxidizer; rendering free radical reactions likely: W. Grochala, J.
Mol. Model., 2008, 14, 887.
17 We notice that other authors have also observed an impurity in their
˚
˚
samples to which a small monoclinic cell (a = 14.342 A, 7.185 A, 7.412
◦
3
˚
˚
A, b = 107.2 , V = 729.63 A ) was later assigned. It turns out that the
transformation of this cell via the matrix (1 0 2, 0 -1 0, 0 0 -2) yields a
cell with unit cell vectors essentially identical to those of our phase II;
this suggests the same chemical identity of impurities observed. See: K.
402 | Dalton Trans., 2012, 41, 396–402
This journal is
The Royal Society of Chemistry 2012
©

 Leszczynski, Piotr J.
Leszczynski, Piotr J.
 Budzianowski, Armand
Budzianowski, Armand
 Dobrzycki, Lukasz
Dobrzycki, Lukasz
 Cyranski, Michal K.
Cyranski, Michal K.
 Derzsi, Mariana
Derzsi, Mariana
 Grochala, Wojciech
Grochala, Wojciech