60
P. Ascenzi et al. / Journal of Inorganic Biochemistry 144 (2015) 56–61
Table 2
A
Peroxynitrite scavenging by ferric heme–proteins and heme-model compounds.
0.9
0.6
0.3
0.0
Heme protein or heme-model compound
kon (M−1 s−1
)
Methanosarcina acetivorans Pgb Cys101(E20)Ser mutant a
Mycobacterium tuberculosis truncated-Hb N b
Pseudoalteromonas haloplanktis TAC125 truncated-Hb O c
Horse heart Mb d
3.8 × 104
6.2 × 104
2.9 × 104
2.9 × 104
1.6 × 104
5.8 × 106
4.8 × 106
5.7 × 104
5.2 × 104
9.0 × 103
1.2 × 104
4.1 × 105
3.5 × 104
4.3 × 105
5.8 × 104
3.2 × 105
6.8 × 104
5.3 × 105
~5 × 105
6.0 × 104
4.1 × 104
Sperm whale Mb e
Sperm whale Mb His64(E7)Ala mutant e
Sperm whale Mb His64(E7)Asp mutant e
Sperm whale Mb His64(E7)Leu mutant e
Sperm whale Mb Phe43(CD1)Trp/His64(E7)Leu mutant e
Sperm whale Mb His64(E7)Tyr/His97(F8)Gly mutant e
Human Hb f
3
4
5
6
pH
7
8
Human SA–heme g
Ibuprofen–human SA–heme h
Truncated human SA–heme i
Ibuprofen–truncated human SA–heme j
CL-cyt c k
B
12
8
CM-cyt c l
CL-CM-cyt c l
Fusarium oxysporum cytochrome P450 NO reductase m
FeTMPS n
MP11 o
a
pH = 7.4 and 20.0 °C. From [44].
pH 7.0 and 20.0 °C. From [42].
pH 7.0 and 20.0 °C. From [43].
pH 7.0 and 20.0 °C. From [24].
pH 7.5 and 20.0 °C. From [25].
pH 7.5 and 20.0 °C. From [24].
b
4
c
d
e
f
0
g
pH 7.2 and 22.0 °C. From [37].
3
4
5
6
7
8
h
pH 7.2 and 22.0 °C. Ibuprofen was 1.0 × 10−2 M. From [37].
i
pH
pH 7.0 and 20.0 °C. From [41].
j
pH 7.0 and 20.0 °C. Ibuprofen was 1.0 × 10−2 M. From [41].
k
pH 7.0 and 20.0 °C. CL was 1.6 × 10−4 M. From [12].
Fig. 6. Effect of pH on the first-order rate constant for peroxynitrite isomerization (i.e., k0;
A). Values of k0 are the average of those obtained in the absence and presence of MP11–
Fe(III) (see Table S1). The continuous line was calculated according to Eq. (4) with
pKa = 6.9 0.2 and klim(top) = 0.83 0.09 s−1. Effect of pH on the second-order rate
constant for MP11–Fe(III)-mediated peroxynitrite isomerization (i.e., kon; B). The continuous
l
pH 7.0 and 20.0 °C. CL was 1.6 × 10−4 M. From [13].
m
pH 8.0 and 12.0 °C. From [39].
pH = 7.6 and 25.0 °C. From [40].
pH = 7.2 and 20.0 °C. Present study.
n
o
line was calculated according to Eq. (5) with pKa = 6.9
0.2 and klim(top) = (1.1
0.1) × 105 M−1 s−1. The peroxynitrite concentration was 1.0 × 10−4 M. All data were obtain-
ed at 20.0 °C. Where not shown, standard deviation is smaller than the symbol. For details,
see text.
experimental conditions HOONO is the species that preferentially
undergoes isomerization.
As shown in Table 1, the spontaneous isomerization of peroxynitrite
yielded 80 6% NO3− and 20 4% NO2−, and (the NO3− and NO−2 yields
increased (91 5%) and decreased (8 3%), respectively, in the pres-
ence of MP11–Fe(III). These results agree with those previously obtain-
ed for the spontaneous isomerization of peroxynitrite and for the ferric
horse heart Mb-, human Hb-, and human SA–heme-catalyzed isomeri-
zation of peroxynitrite [24,37].
2.5 × 10−5 M to 1.0 × 10−3 M (according to Eq. (4)) (Figs. 3, panel A, 5,
panel B, and Table S1).
The pH dependence of k0 and kon for peroxynitrite isomerization, in
the absence and presence of MP11–Fe(III), was examined to identify
tentatively the species that preferentially react(s) with MP11–Fe(III).
Values of k0 and kon for peroxynitrite isomerization in the absence and
presence of MP11–Fe(III), respectively, decrease on increasing pH
from 3.6 to 8.1 (Fig. 6). The analysis of data reported in Fig. 6, according
to Eq. (5), indicates that values of pKa for the pH dependence of
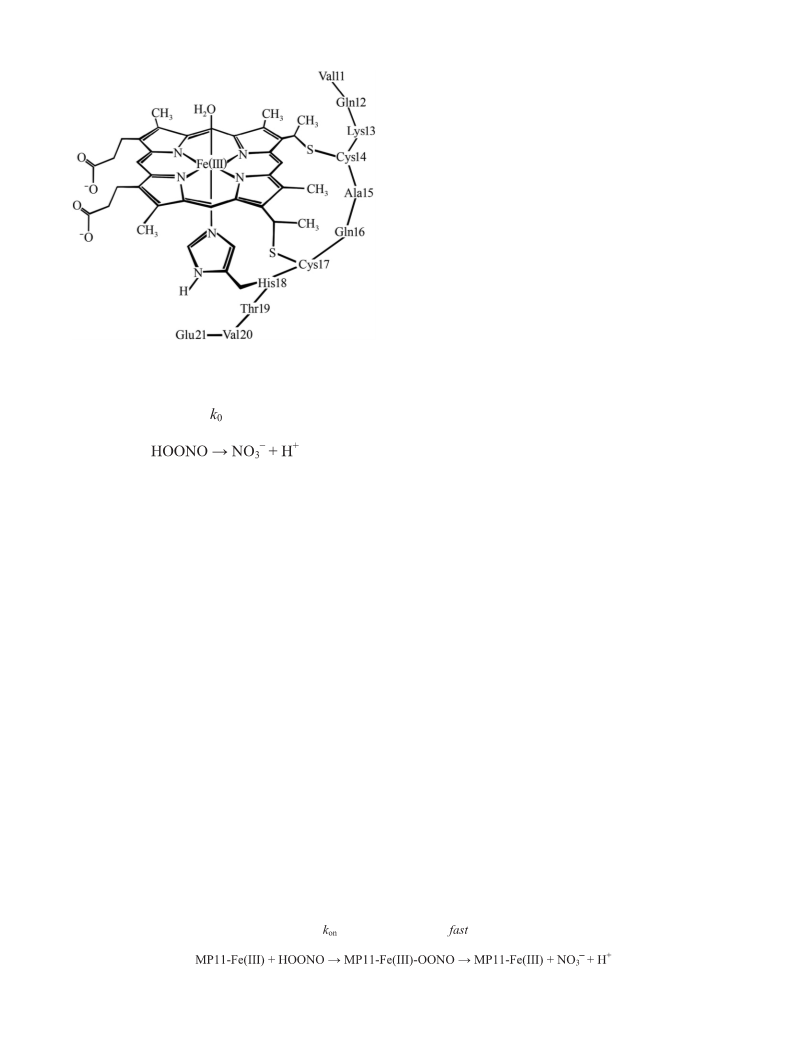
Peroxynitrite isomerization is catalyzed by penta-coordinated MP11–
Fe(III), while hexa-coordinated MP11–Fe(III)–CN is non-reactive (Figs. 3
and 4). This demonstrates that the efficiency of the catalytic process
reflects the access to and the coordination of the heme–Fe(III) atom.
The values of kon for peroxynitrite isomerization by ferric heme–proteins
and heme-model compounds range between 9.0 × 103 M−1 s−1 and
5.8 × 106 M−1 s−1 (Table 2) (see [12,13,24,25,37,39–44] and present
study), reflecting the coordination of the heme–Fe(III) atom. In fact,
cardiolipin (CL) binding facilitates peroxynitrite isomerization by wild-
type and carboxymethylated ferric horse heart cyt c inducing the cleav-
age or the severe weakening of the sixth coordination bond of the
heme–Fe(III) atom [12,13,45]. In contrast, ibuprofen binding to the FA2
site of ferric wild-type and truncated human SA-heme induces the
hexa-coordination of the heme–Fe(III) atom, with the consequent inhi-
bition of the heme-based reactivity [37,41,46]. Also, hexa-coordinated
ferric horse heart cyt c and human neuroglobin [47,48] do not catalyze
the peroxynitrite conversion to NO−3 [12,49]. Moreover, the analysis of
kon values for peroxynitrite isomerization by wild-type and mutants of
sperm whale Mb(III) (see Table 2) suggests that the heme–Fe(III) reac-
tivity is regulated not only by steric factors modulating the ligand access
to the metal center, but also by the Lewis acidity of the heme–Fe(III)
k0 (pKa = 6.9
0.2) (Fig. 6, panel A) and of kon (pKa = 6.9
0.2)
(Fig. 6, panel B) are essentially the same within the error limit. The
pKa values for the pH dependence of k0 and kon here determined are
in excellent agreement with pKa values reported in the literature [24,
25,33,37,38]. Accordingly, the close similarity of the pH dependence of
k0 (Fig. 6, panel A) and of kon (Fig. 6, panel B) suggests that under all
Table 1
NO−3 and NO−2 distribution of peroxynitrite isomerization in the absence and presence of
MP11–Fe(III), and MP11–Fe(III)–CN, at pH 7.2 and 20.0 °C. a
.
MP11–Fe(III)
(M)
MP11–Fe(III)–CN b
(M)
[NO−3
(%)
]
[NO−2
(%)
]
[NO−3 ] + [NO−2
(%)
]
–
–
81
91
79
6
6
5
17
8
22
4
3
3
98 10
1.0 × 10−5
–
99
9
8
–
1.0 × 10−5
101
a
The MP11–Fe(III) concentration was 5.0 × 10−5 M, and the peroxynitrite concentra-
tion was 2.0 × 10−4 M.
b
The cyanide concentration was 5.0 × 10−4 M.

 Ascenzi, Paolo
Ascenzi, Paolo
 Leboffe, Loris
Leboffe, Loris
 Santucci, Roberto
Santucci, Roberto
 Coletta, Massimo
Coletta, Massimo