
Journal of the American Chemical Society p. 5514 - 5519 (1987)
Update date:2022-08-17
Topics:
 Chari
Chari
 Whitman
Whitman
 Kozarich
Kozarich
 et al.
et al.
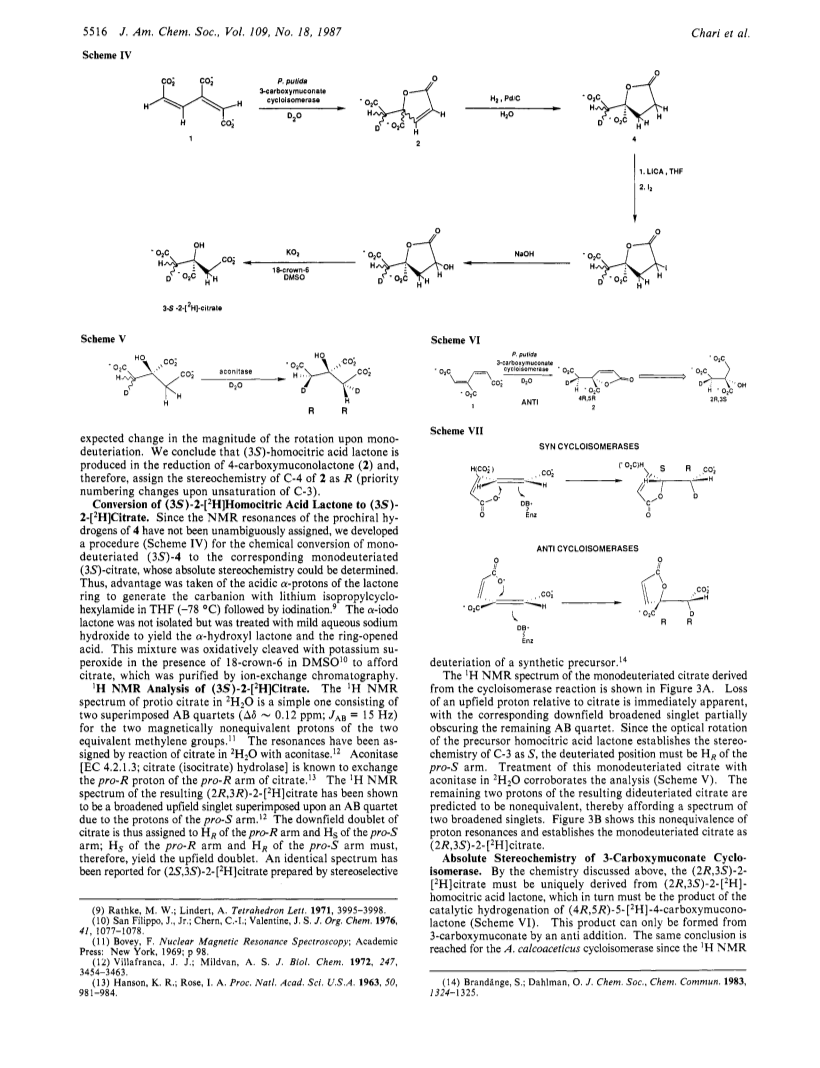
The absolute stereochemical course of the 3-carboxymuconate cycloisomerases [EC 5.5.1.2; 2-carboxy-5-oxo-2,5-dihydrofuran-2-acetate lyase (decyclizing)] from Pseudomonas putida and Acinetobacter calcoaceticus has been determined by chemical and 1H NMR methods. The product of the enzyme-catalyzed reaction in 2H2O was detected by NMR and trapped by catalytic hydrogenation to afford 5-[2H]homocitrate lactone. Subsequent chemical degradation of the monodeuteriated homocitrate lactone gave (2r,3S)-2-[2H]citrate as determined by 1H NMR analysis. The product of the cycloisomerase reaction was established as (4R,5R)-5-[2H]-4-carboxymuconate, indicating that the lactonization proceeded by an anti addition - the mechanistic and stereochemical antipode of the previously studied muconate cycloisomerase from P. putida and 3-carboxymuconate cycloisomerase from Neurospora crassa. The anti addition probably represents the lower energy pathway for the reaction and suggests that the evolutionary relationship between the two classes of cycloisomerases is more remote than previously believed.
View More




Zhejiang Kangfeng Chemical Co.,LTD.
Contact:+86-579-86709687
Address:Xueshizhai Industrial Zone, Weishan Town,Dongyang City, Zhejiang Province ,China
Contact:+86-15527136576
Address:Wuhan city
Anhui Redstar Pharmaceutical Corp., Ltd
Contact:+86-563-5120837
Address:Jingxian Industrial Development Zone, Anhui , China
Huaian Double Win Chemicals Co.,Ltd.
Contact:+86-13511538872
Address:Blk 43,Greenland Century Town,Huaian District, Huaian City,Jiangsu Province,China
Taizhou KEDE Chemical.Co.,Ltd.
Contact:86-576-84613060
Address:Jiangkou Chemical Zoon
Doi:10.1039/a903876k
(1999)Doi:10.1016/S0040-4039(01)87617-6
(1973)Doi:10.1016/j.tetlet.2016.05.062
(2016)Doi:10.1016/j.ssc.2005.03.003
(2005)Doi:10.1016/j.ejmech.2016.01.036
(2016)Doi:10.1039/c5gc00580a
(2015)