160
E.M.M. Ewais et al. / Journal of Alloys and Compounds 649 (2015) 159e166
hydrothermal [27,28], microwave-assisted combustion processing
29], microemulsion [30], metal-organic processing [31e34], spray
drying [35], freeze drying [36], and mechanochemical synthesis
37,38] techniques. The chemical co-precipitation method [39e42]
Table 1
Chemical composition of Al scraps.
[
Elements
Al
Zn
Mg
Cu
[
Percentage, %
92
3.96
2.35
1.7
ensures proper distribution of various metal ions resulting into
stoichiometric and smaller particle sized product compared to
some of the others. Moreover, the process is a low cost technique
suitable for the mass production compared to the other mentioned
methods.
An aqueous solution of magnesium nitrate was achieved by
dissolving the magnesium scrap in nitric acid. The aqueous solution
of aluminum nitrate was achieved by dissolving the aluminum
scrap in aqua regia of 20% HCl and 20% HNO acids. The stock so-
3
lution used for spinel synthesis was made by dissolving magnesium
and aluminum nitrates in distilled water with stirring.
At present, most techniques used to produce MA spinel,
whether they are solid state reactions or chemical synthesis, are
ꢀ
conducted at high temperature (>800 C). However, it is known
that high temperature processing is detrimental to microstructure
and in turn the properties due to the non-uniform grain growth.
Therefore, low temperature synthesis of nanocrystalline powders
of MA spinel should be investigated.
In a typical synthesis process, the aqueous solution of the
aluminum nitrate was added to a stirring aqueous solution of
magnesium nitrate to give MgO/Al
The aqueous suspended solutions were formed by the addition of
NH OH drop by drop with constant stirring 500 rpm for 15 min to
2 3
O molar ratios of 1:1 powder.
On the other hand, industrial wastes released from different
manufacturing processes and treatment materials are produced in
large quantities and have harmful effects on the environment. Ac-
cording to research conducted by the US Environmental Protection
Agency, recycling scrap metal wastes can be quite beneficial to the
environment. For example, using recycled scrap metal in place of
virgin iron ore can yield 75% savings in energy, 90% savings in raw
materials used, 86% reduction in air pollution, 40% reduction in
water use, 76% reduction in water pollution and 97% reduction in
mining wastes [43].
4
achieve good homogeneity. pH of the solution was maintained at 10
during the precipitation process. The co-precipitates were filtered
off, washed with water and dried in an oven at 110 C overnight. In
order to produce MA spinel, the dry precursor was calcined in a
ꢀ
muffle furnace up to different temperatures (650, 750, 850, 950,
ꢀ ꢀ
300, 1400 and 1500 C) with a rate of heating of 10 C/min for 2 h.
1
2
.2. Characterization
In 1990 total aluminum production was around 28 million
tonnes (with over 8 million tonnes recycled from scrap) and today
the total is close to 56 million tonnes (with close to 18 million
tonnes recycled from scrap). By 2020 metal demand is projected to
have increased to around 97 million tonnes (~31 million tonnes
recycled from scrap).
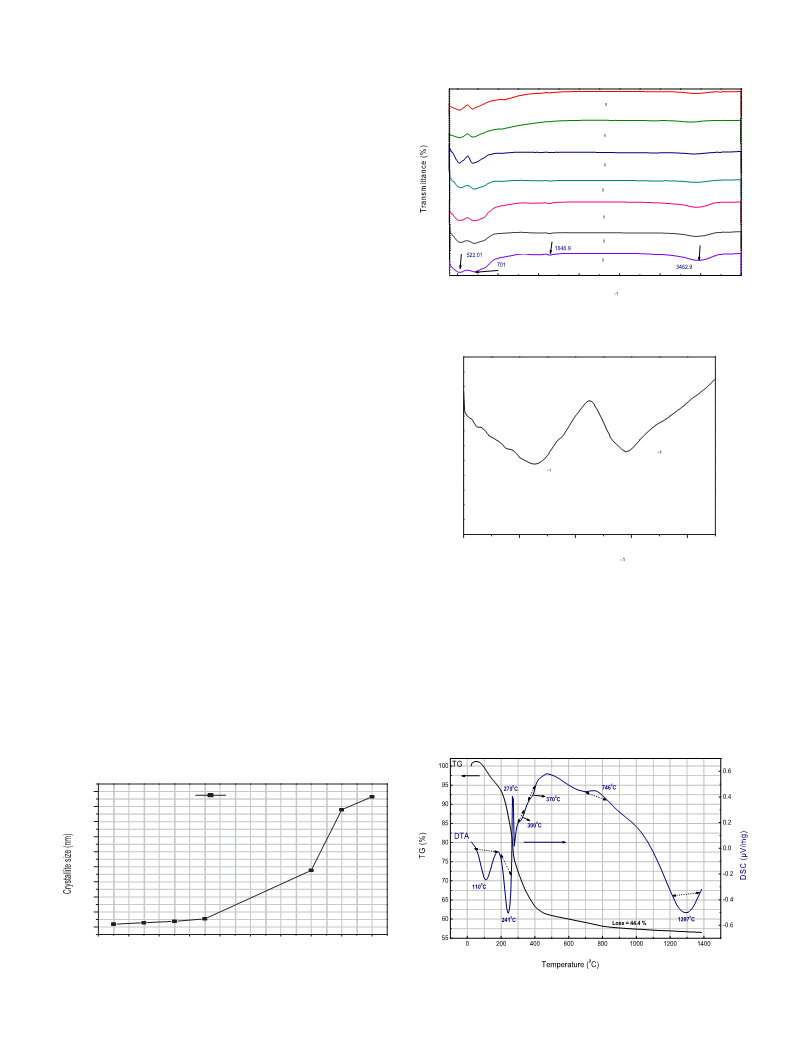
XRD patterns of the resulting products were characterized by a
Brucker D8-advance X-ray powder diffractometer with Cu Ka ra-
diation (k ¼ 1.5406 Å). The crystallite sizes of the produced mag-
nesium aluminate spinels were calculated from the most intense
peak (311) using the DebyeeScherrer formula:
During manufacturing of magnesium products, large amount of
wastes in the form of chips and discards is produced in the
machining process of castings and sheets. Approx. 1/3 of magne-
sium used to fabricate structural products ends as scrap, so it is
really inevitable to find ways for its efficient recycling in order to
keep the use of Mg and thus price at a reasonable level.
Nowadays, industrial waste materials are getting a wide exposure
to the future generation of the material science world. The novelty
and the idea of this work is how to convert unwanted waste mate-
rials into something useful or value-added products via an envi-
ronmental friendly method for advanced applications. In this study,
dRX ¼ Kl=b cosq
where dRX is the crystallite size, k ¼ 0.9 is a correction factor to
account for particle shapes,
b is the full width at half maximum
(FWHM) of the most intense diffraction peak (311) plane of MA
spinel phase,
the Bragg angle.
l
is the wavelength of Cu target ¼ 1.5406 Å, and
q is
Fourier transform infrared spectra (FTIR) spectroscopy (Model,
Jasco-6300 type A, Japan spectrometer) was used at room tem-
ꢁ1
perature in the range of 400e4000 cm
with a resolution of
ꢁ1
4
cm to identify functional groups.
2 4
our group reported for the first time, the synthesis of MgAl O spinel
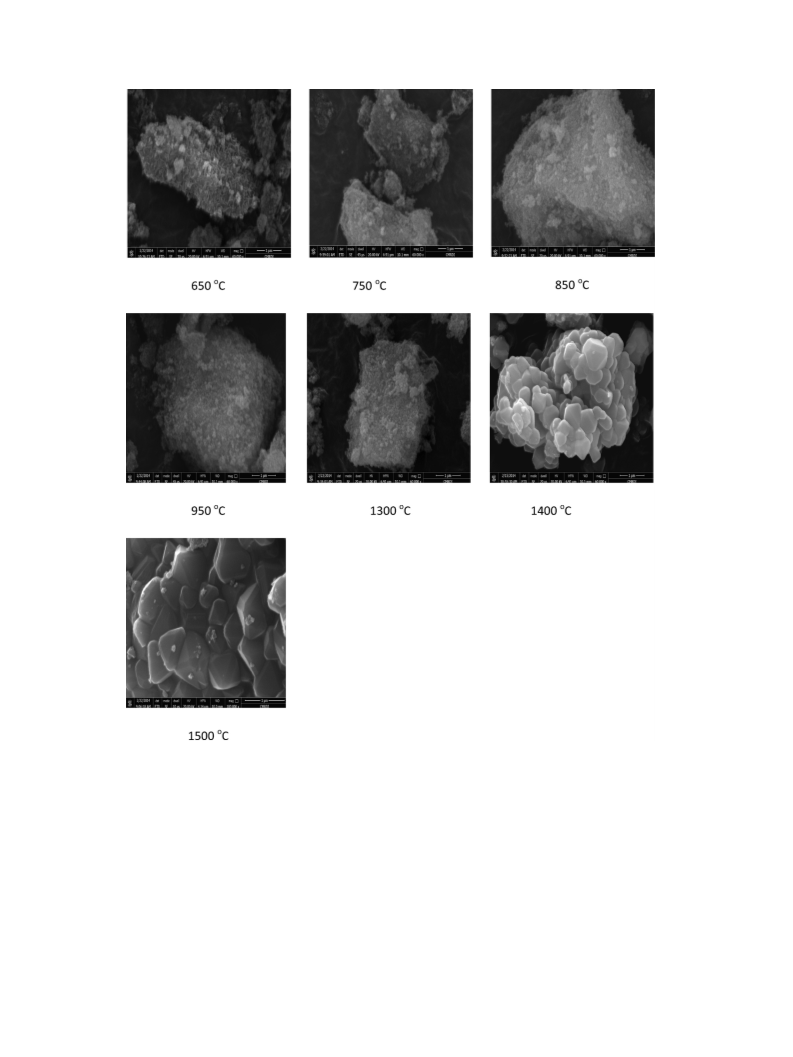
Powder morphology and microstructure of the produced mag-
nesium aluminate spinel were examined by using backscattered
electron (BSE) in the field emission scanning electron microscopy
nanoparticles from industrial wastes of aluminum and magnesium
scraps released from foundries by co-precipitation method at low
temperature. Magnesium aluminate spinel was produced at
ꢀ
different temperatures 650e1500 C. The phase composition, ther-
mal analysis, and microstructure of these compositions were
detected using X-ray diffraction (XRD), differential thermal analysis
MA Spinel
(
DTA), infrared spectrum (IR), scanning electron microscope (FE-
1500 C
SEM) as well as transmission electron microscope (TEM). The optical
properties of the produced MA were also studied by UV-VIS-NIR
spectrophotometer and photoluminescence analysis.
1400 C
1300 C
2
. Materials and experimental procedure
950 C
2
.1. Materials and processing
8
7
50 C
50 C
Aluminum and magnesium scrap released from foundries
(
supplied by Central Metallurgical Research & Development Insti-
tute [CMRDI], Helwan, Egypt) were used as starting materials.
Ammonia solution [NH OH] was used during the co-precipitation
650 C
10
20
30
40
50
60
70
4
2
Theta (degree)
to maintain the pH at 10. The chemical composition of aluminum
scrap is illustrated in Table 1.
Fig. 1. XRD patterns of MA spinel powders annealed at different temperatures.

 Ewais, Emad M.M.
Ewais, Emad M.M.
 Besisa, Dina H.A.
Besisa, Dina H.A.
 El-Amir, Ahmed A.M.
El-Amir, Ahmed A.M.
 El-Sheikh, Said M.
El-Sheikh, Said M.
 Rayan, Diaa E.
Rayan, Diaa E.