Journal of Inorganic and General Chemistry
ARTICLE
Zeitschrift für anorganische und allgemeine Chemie
(0.31 g, 2.77 g) in 10 mL THF were added. After stirring overnight,
the direct synthesis route is quite challenging although it was
reported before.[38]
the solution was evaporated to dryness under reduced pressure
(10À 3 mbar) to obtain a dark yellow solid. 29SiÀ NMR (benzene-d6,
298 K, 59,6 MHz) δ/ppm: À 21.8.
Reaction of KOtBu, (NH4)2Ce(NO3)6 and (Me2SiO)n in acid solution.
To a solution of 0.64 g (1.2 mmol) Cerium ammonium nitrate
((NH4)2Ce(NO3)6) in 15 mL THF a solution of 0.75 g (6.72 mmol)
KOtBu in THF was added and the resulting yellow solution was
separated from the white precipitate by decantation. An excess of
(Me2SiO)n (0.32 g), 5 mL ethanol and 5 mL hydrochloric acid were
added. After stirring overnight, the solution was evaporated to
dryness under reduced pressure (10À 3 mbar) to obtain a dark yellow
solid. 29SiÀ NMR (benzene-d6, 298 K, 59,6 MHz) δ/ppm: À 21.8.
Materials and Methods
All reactions were performed under nitrogen atmosphere using a
modified Stock vacuum line taking utmost precautions to prevent
adventitious hydrolysis. All glass equipment were stored in a drying
°
oven at 140 C before use and were heated in vacuum additionally
to remove finally all impurities of adherent moisture. Solvents were
purified by reported methods.[67] Poly(dimethylsiloxane) was used
without further purification ((Me2OSi)n, Aldrich); Cerium(IV)
ammonium nitrate (CAN, (NH4)2Ce(NO3)6 Aldrich, 98,5%) was dried
in dynamic vacuum for 7 hours; KOtBu and [Sr(OiPr)2] were
synthesized by dissolving the metal in a mixture of toluene and the
corresponding alcohol (tert-butyl alcohol or iso-propyl alcohol);
KOtBu was purified by vacuum sublimation. The amides [In
{N(SiMe3)2}3] and [Sn{N(SiMe3)2}2] were synthesized following pub-
lished procedures.[68,69]
Reaction of KOtBu, (NH4)2Ce(NO3)6 and (Me2SiO)n for 60h at room
temperature. To a solution of 0.48 g (0.8 mmol) Cerium ammonium
nitrate ((NH4)2Ce(NO3)6) in 15 mL THF
a solution of 0.59 g
(5.2 mmol) KOtBu was added and the resulting yellow solution was
separated from the white precipitate by decantation. An excess of
(Me2SiO)n (0.27 g) was added as well as and after stirring for 60 h
the solution was evaporated to dryness under reduced pressure
(10À 3 mbar) to obtain a dark yellow solid. 29SiÀ NMR (benzene-d6,
298 K, 59,6 MHz) δ/ppm: À 21.8.
1H (300.1 MHz) and 29Si (59.6 MHz) chemical shifts are reported in
parts per million (ppm) relative to external tetramethylsilane
references. Microanalysis for C, H, N and S were carried out using a
Hekatech CHNS EuroEA 3000 Analyzer with Helium as carrier gas.
Data collection for X-ray crystal structure determination were
performed on a STOE IPDS I/II diffractometer using graphite-
monochromated Mo-Kα radiation (λ=0.71073 Å). The data were
Reaction of KOtBu, (NH4)2Ce(NO3)6 and (Me2SiO)n under reflux. To
a
solution of 0.41 g (0.7 mmol) Cerium ammonium nitrate
((NH4)2Ce(NO3)6) in 15 mL THF a solution of 0.50 g (4.4 mmol) KOtBu
was added and the resulting yellow solution was separated from
the white precipitate by decantation. An excess of (Me2SiO)n
corrected for Lorentz and polarization effects.
A numerical
°
(0.23 g) was added as well as and after stirring at 60 C ovnernight,
absorption correction based on crystal-shape optimization was
applied for all data.[51] The programs used in this work are Stoe’s X-
Area,[52] including X-RED and X-Shape for data reduction and
absorption correction[53] and the WinGX suite of programs[56]
including SIR-92[55] and SHELXL-97[57] for structure solution and
refinement. The hydrogen atoms were placed in idealized positions
and constrained to ride on their parent atom. The last cycles of
refinement included atomic positions for all atoms, anisotropic
thermal parameters for all non-hydrogen atoms and isotropic
thermal parameters for all hydrogen atoms. Graphics for publica-
tion were prepared using DIAMOND.[70]
the solution was evaporated to dryness under reduced pressure
(10À 3 mbar) to obtain a dark yellow solid. 29SiÀ NMR (benzene-d6,
298 K, 59,6 MHz) δ/ppm: À 21.8.
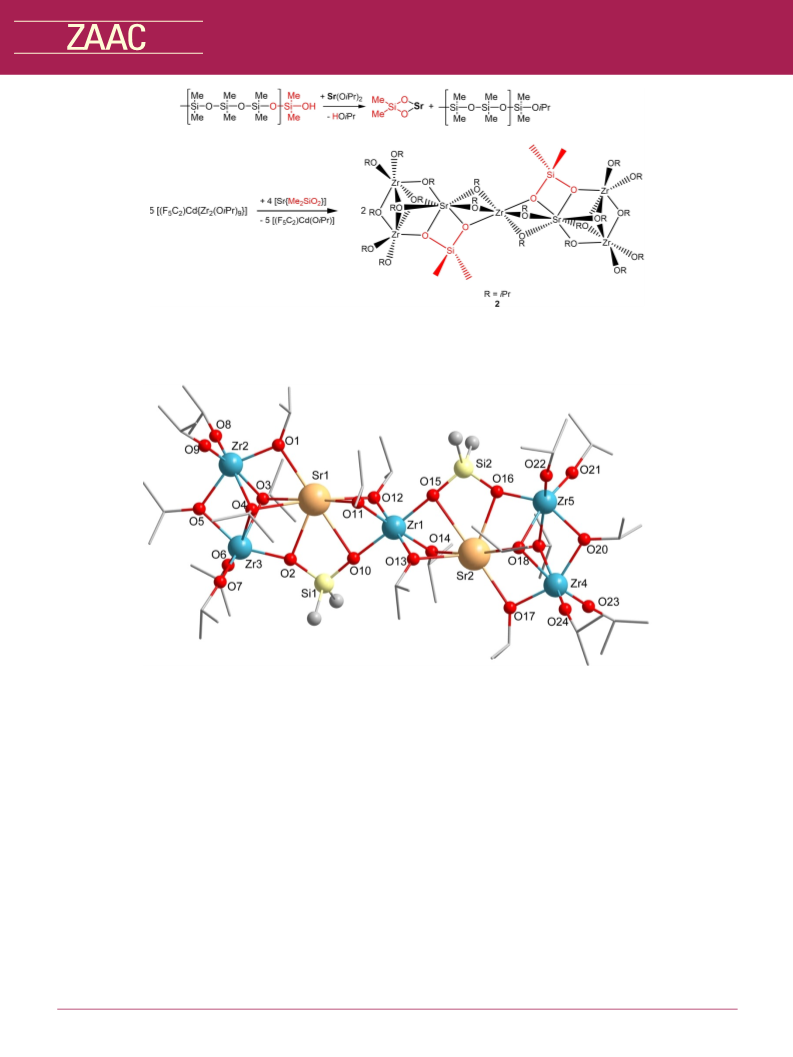
Reaction of [(F5C2)Cd{Zr2(OiPr)9}] and [Sr(OiPr)2]. 0.50 g
(0.53 mmol) [(F5C2)Cd{Zr2(OiPr)9}] were dissolved in 10 mL toluene
and 0.16 g (0.78 mmol) [Sr(OiPr)2] in toluene (4 mL) were added.
After stirring over night at room temperature all volatiles were
removed under reduced pressure (10À 3 mbar) and the product (2)
was obtained as a light yellow solid.
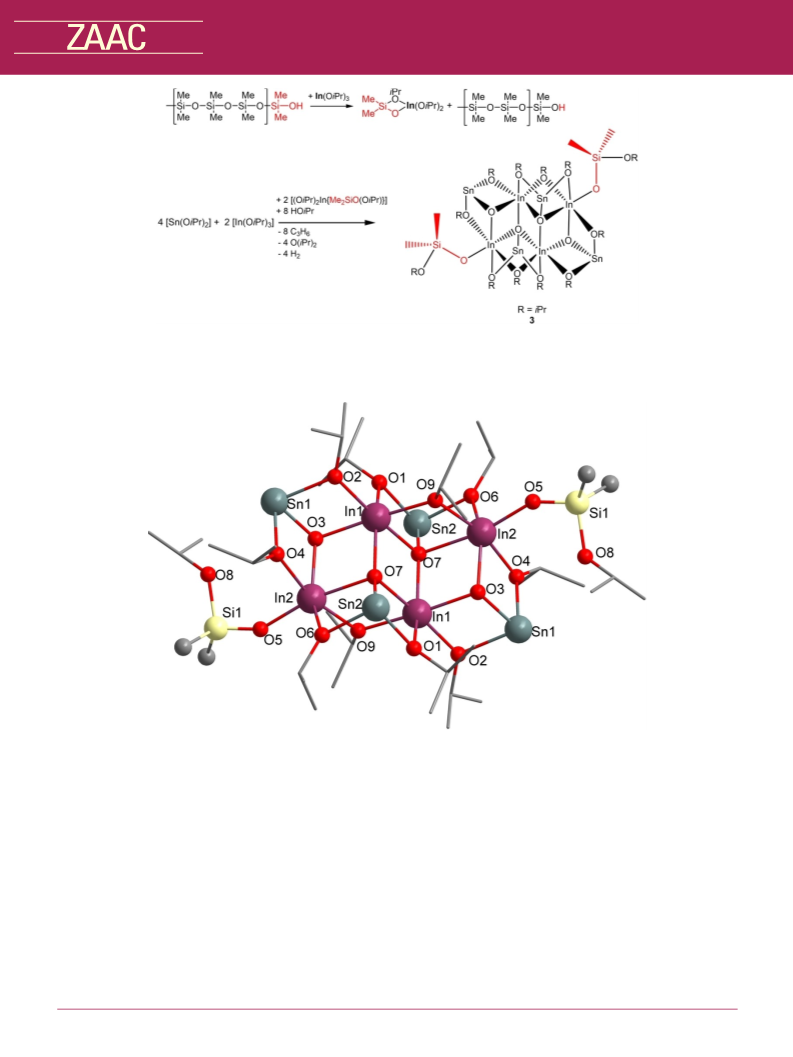
Reaction of [In{N(SiMe3)2}3] and [Sn{N(SiMe3)2}2]. 0.13 g
(0.23 mmol) [In{N(SiMe3)2}3] in 10 mL toluene was added to a
solution of 0.10 g (0.23 mmol) [Sn{N(SiMe3)2}2] in 10 mL toluene.
After the addition of 5 mL of dry iso-propyl alcohol, the colorless
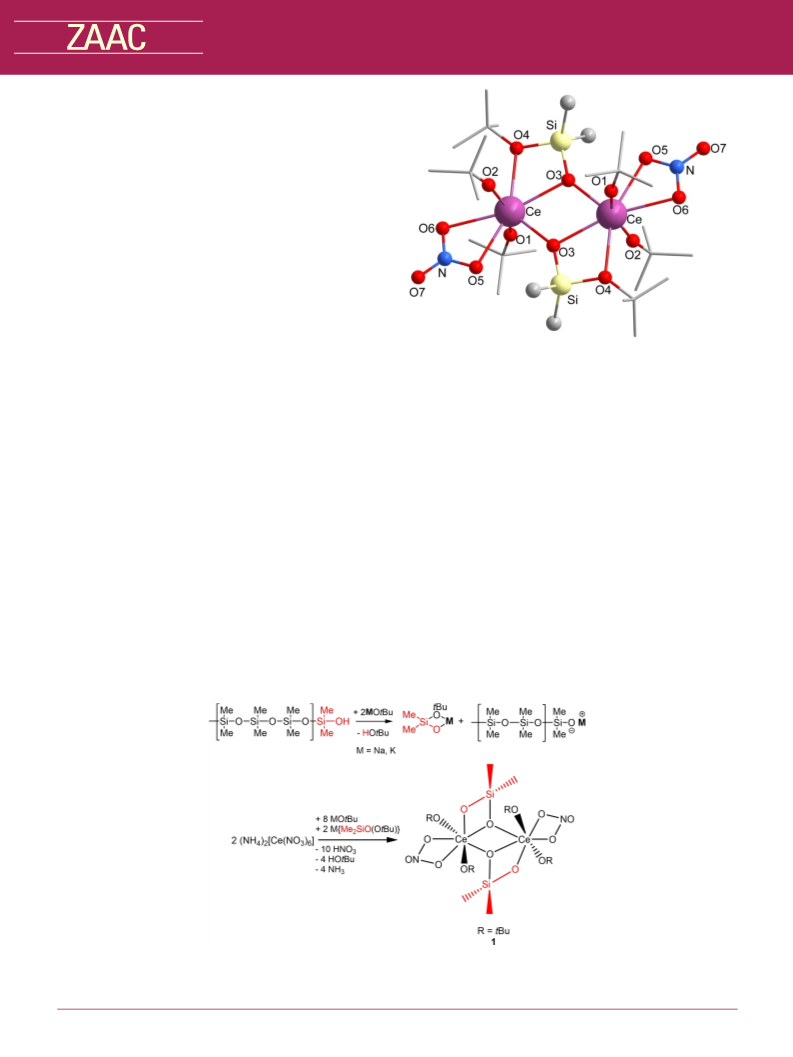
Reaction of KOtBu and (NH4)2Ce(NO3)6. To a solution of 0.62 g
(1.1 mmol) Cerium ammonium nitrate ((NH4)2Ce(NO3)6) in 20 mL
THF a solution of 0.76 g (6.8 mmol) KOtBu was added. The resulting
yellow solution was separated from the white precipitate by
decantation and was evaporated to dryness under reduced
pressure. 0.11 g (5%, 0.1 mmol) of the cerium siloxide
[Ce2(OtBu)4{Me2Si(OtBu)O}2(NO3)2] (1) were obtained as yellow
°
solution was stirred overnight at 70 C. All volatiles were removed
under reduced pressure (10À 3 mbar) and the product (3) was
obtained as colorless crystals from a solution in toluene.
crystals from
a concentrated solution in THF. Anal. Calcd
Ce2O14C28H66N2Si2: C 33.93, H 6.71, N 2.83. Found: C 34.52, H 7.05, N
1.98.
Conclusions
Reaction of KOtBu, (NH4)2Ce(NO3)6 and silicon grease. To a
solution of 0.35 g (0.6 mmol) Cerium ammonium nitrate ((NH4)2Ce-
(NO3)6) in 15 mL THF a solution of 0.43 g (3.8 mmol) KOtBu was
added and the resulting yellow solution was separated from the
white precipitate by decantation. An excess of silicon grease
This paper reports the structural investigations of alkoxide-
siloxide complexes of tetravalent cerium, zirconium and
trivalent indium serendipitously formed in the adventitious
reaction of the starting materials with silicon grease used for
lubricating the glass joints. The unexpected incorporation of
the dimethylsiloxane units {Me2Si(OR)O}À (R=iPr, tBu) and
{Me2SiO2} in the molecular structures of mono- and hetero-
bimetallic alkoxides opened up new insights in the broad
spectrum of possible heteroleptic polymetallic coordination
compounds that can act as model intermediates in co-
hydrolysis of metal alkoxides and siloxides involved in the sol-
gel processing of MOxÀ SiOx nanocomposites. The targeted
synthesis of such complexes out of defined molecular educts
°
(0.22 g) was added, and the solution was stirred at 60 C for 3 h,
followed by the evaporation to dryness under reduced pressure
(10À 3 mbar) to obtain a dark yellow solid. 29SiÀ NMR (benzene-d6,
298 K, 59,6 MHz) δ/ppm: À 21.8.
Reaction of KOtBu, (NH4)2Ce(NO3)6 and (Me2SiO)n in alkaline
solution. To a solution of 0.39 g (0.7 mmol) Cerium ammonium
nitrate ((NH4)2Ce(NO3)6) in 15 mL THF
a solution of 0.48 g
(4.2 mmol) KOtBu in THF was added and the resulting yellow
solution was separated from the white precipitate by decantation.
An excess of (Me2SiO)n (0.14 g), 5 mL ethanol and additional KOtBu
Z. Anorg. Allg. Chem. 2021, 1102–1109
published by Wiley-VCH GmbH.

 Nahrstedt, Vanessa
Nahrstedt, Vanessa
 Raauf, Aida
Raauf, Aida
 Hegemann, Corinna
Hegemann, Corinna
 Brune, Veronika
Brune, Veronika
 Schl?fer, Johannes
Schl?fer, Johannes
 Mathur, Sanjay
Mathur, Sanjay