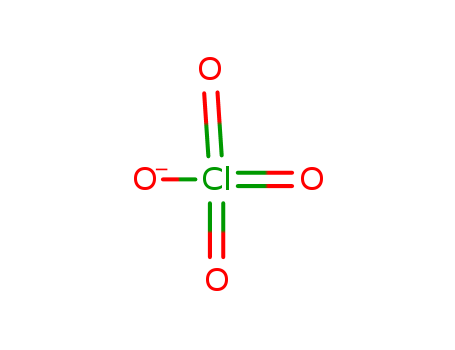
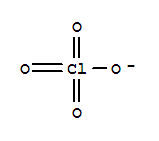Chemical Property of Perchlorate
Edit
Chemical Property:
- Melting Point:262-263 °C
- Boiling Point:°Cat760mmHg
- Flash Point:°C
- PSA:71.44000
- Density:g/cm3
- LogP:0.15630
- XLogP3:2.2
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:4
- Rotatable Bond Count:0
- Exact Mass:98.9485112
- Heavy Atom Count:5
- Complexity:95.8
- Transport DOT Label:Oxidizer
- Purity/Quality:
-
99% *data from raw suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:[O-]Cl(=O)(=O)=O
-
Description
Concern about environmental perchlorate exposure centers on
its inhibition of iodide uptake into the thyroid. Decreased
iodine intake may decrease thyroid hormone production. In
the last few years, perchlorate has become a major inorganic
contaminant in drinking water and has been detected in
a number of public drinking water systems throughout the
Unites States. In 1998, the US EPA added perchlorate to the
Drinking Water Candidate Contaminant List.
-
Uses
Perchlorate is a soluble oxychloro anion most commonly used as a solid salt in the form of ammonium perchlorate, potassium perchlorate, lithium perchlorate, or sodium per- chlorate, all of which are highly soluble. Ammonium perchlorate is the most widely used perchlorate compound. In their pure forms, these salts are white or colorless crystals or powders. Perchlorate salts dissolve in water and readily move from surface to groundwater. Perchlorate is known to originate from both natural and man-made sources.
The most common uses for ammonium perchlorate are in explosives, military munitions, and rocket propellants. In addition, perchlorate salts are used in a wide range of nonmilitary applications, including pyrotechnics and fireworks, blasting agents, solid rocket fuel, matches, lubricating oils, nuclear reactors, air bags, and certain types of fertilizers. Improper storage and disposal related to these uses is the most typical route for perchlorate to enter into the environment. Monitoring data show that more than 4% of public water systems, serving between 5 million and 17 million people, have detected perchlorate in their finished water. The primary source of perchlorate is the ammonium salt.
Ammonium perchlorate is the oxidizer ingredient in solid
propellant mixtures for rockets, missiles, and munitions. Other
uses of perchlorate salts include medicine, matches, metal
cation chemistry, and pyrotechnics (illuminating and signaling
flares, colored and white smoke generators, tracers, incendiary
delays, fuses, photo-flash compounds, and fireworks).
Perchlorate is also found in lubricating oils, finished leather,
fabric fixer, dyes, electroplating, aluminum refining, manufacture
of rubber, paint, and enamel production, as an additive in
cattle feed and magnesium batteries, and as a component of
automobile airbag inflators.