- Propoxyphene hydrochloride
- Propoxyphene, dl-
- 1639-66-3Butanedioic acid,2-sulfo-, 1,4-dioctyl ester, sodium salt (1:1)
- 163972-12-1Dodecanamide,N-(2,5-dioxo-7-oxabicyclo[4.1.0]hept-3-en-3-yl)-
- 16399-77-2iron(II) chloride dihydrate
- 16400-13-81H-Inden-4-ol,2,3-dihydro-7-methyl-
- 16400-50-31,1'-Biphenyl,3,3',5,5'-tetrabromo-
- 16401-41-5Acetic acid,2-[(3-thienylmethyl)thio]-
- 164014-95-3Boronic acid,B-(2,3-dihydro-1,4-benzodioxin-6-yl)-
- 164029-51-01,3,4-Oxadiazole-2-carboxylic acid, 5-[[[(1,1-dimethylethoxy)carbonyl]amino]methyl]-, ethyl ester
- 16403-84-22-Naphthalenecarboxamide,4-[2-[5-(aminocarbonyl)-2-methylphenyl]diazenyl]-3-hydroxy-N-phenyl-
- 1640-39-72,3,3-Trimethylindolenine
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

| Name |
Propoxyphene hydrochloride |
EINECS | 216-683-5 |
| CAS No. | 1639-60-7 | Density | N/A |
| PSA | 29.54000 | LogP | 5.07750 |
| Solubility | 3.7mg/L(25 oC) | Melting Point |
165-1670C |
| Formula | C22H30ClNO2 | Boiling Point | 444 °C at 760 mmHg |
| Molecular Weight | 375.939 | Flash Point | 130.6 °C |
| Transport Information | UN 3249 | Appearance | White solid |
| Safety | 16-36/37-45 | Risk Codes | 11-23/24/25-39/23/24/25 |
| Molecular Structure |
|
Hazard Symbols | F,T |
| Synonyms |
2-Butanol,4-(dimethylamino)-3-methyl-1,2-diphenyl-, propinate (ester), hydrochloride (+)-(7CI);2-Butanol, 4-(dimethylamino)-3-methyl-1,2-diphenyl-, propionate (ester),hydrochloride, (2S,3R)-(+)- (8CI);Benzeneethanol, a-[(1R)-2-(dimethylamino)-1-methylethyl]-a-phenyl-, propanoate (ester),hydrochloride, (aS)-(9CI);Benzeneethanol, a-[2-(dimethylamino)-1-methylethyl]-a-phenyl-, propanoate (ester), hydrochloride,[S-(R*,S*)]-;(+)-Propoxyphene hydrochloride;Algaphan;Antalvic;Darvon;Darvon hydrochloride;Deprancol;Develin;Dextropropoxyphene hydrochloride;Dolene;Dolocap;Doloxen;Doloxene;Erantin;Femadol;Harmar;Propoxychel;Propoxyphen hydrochloride;Propoxyphene hydrochloride;Proxagesic;SK 65;d-4-Dimethylamino-3-methyl-1,2-diphenyl-2-butanol propionate hydrochloride;d-Propoxyphene hydrochloride;d-Propoxyphene monohydrochloride;a-(+)-Propoxyphene hydrochloride;a-Propoxyphene hydrochloride;a-d-Propoxyphene hydrochloride; |
Propoxyphene hydrochloride Chemical Properties
The MW of d-PROPOXYPHENE HYDROCHLORIDE(1639-60-7): 375.93
The Structure:
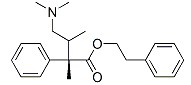
EINECS: 216-683-5
Synonyms: DARVON;DEXTROPROPOXYPHENE HYDROCHLORIDE;PROPOXYPHENE HYDROCHLORIDE;(+)-hlorid;algaphan;alpha-(+)-propoxyphenehydrochloride;(+)-1,2-diphenyl-2-propionoxy-3-methyl-4-dimethylaminobutanehydrochloride;alpha-[2-(dimethylamino)-1-methylethyl]-alpha-phenyl-benzeneethanopropano
Propoxyphene hydrochloride Uses
d-PROPOXYPHENE HYDROCHLORIDE(1639-60-7) is a synthetic, nonantipyritic, orally effective analgesic, with similar pharmacological activity and effects to codeine.
d-PROPOXYPHENE HYDROCHLORIDE(1639-60-7) is not a narcotic but can be substituted for codeine, and is useful in any condition associated with pain. Chemically, this analgesic is not analogous to codeine or to morphine.
d-PROPOXYPHENE HYDROCHLORIDE(1639-60-7) is a stronger analgesic than aspirin but has no antipyretic effects. It is sometimes taken in combination with aspirin and acetaminophen. It has widespread use for dental pain since aspirin is relatively ineffective, but it is not useful for deep pant.
Propoxyphene hydrochloride Production
The manufacture of d-PROPOXYPHENE HYDROCHLORIDE(1639-60-7) starts with relatively simplechemicals,but consists of many steps∶
1.Formation of a ketone from propiophenone and paraformaldehyde
2.Coupling the ketone with benzyl chloride, using the Grignard reaction (and decomposition)
3.Resolution of the optical isomers by the use of d-camphorsulfonic acid in acetone
4.Splitting off the d-camphorsulfontc acid by using ammonium hydroxide and conversion of the desired α-dextro isomer to the hydrochloride; only the dextro isomer is active as an analgesic
5.Esterification of the α-dextro isomer with proptoric anhydride
6.Isolation
7.Filtration
8.Drylng
Propoxyphene hydrochloride Toxicity Data With Reference
| 1. | orl-wmn TDLo:650 µg/kg:GIT,LIV,SYS | GUTTAK Gut. 27 (1986),444. | ||
| 2. | orl-man TDLo:11 mg/kg:CNS,PUL | AIMDAP Archives of Internal Medicine. 129 (1972),62. | ||
| 3. | orl-man LDLo:15 mg/kg:PUL | JFSCAS Journal of Forensic Sciences. 19 (1974),72. | ||
| 4. | orl-wmn LDLo:15 mg/kg:CNS,PUL | JFSCAS Journal of Forensic Sciences. 19 (1974),72. | ||
| 5. | orl-wmn LDLo:39 mg/kg:CNS,PUL | AIMDAP Archives of Internal Medicine. 129 (1972),62. | ||
| 6. | orl-chd TDLo:27 mg/kg:PUL,CNS | JOPDAB Journal of Pediatrics. 63 (1963),158. | ||
| 7. | orl-chd LDLo:70 mg/kg | JOPDAB Journal of Pediatrics. 63 (1963),158. | ||
| 8. | orl-rat LD50:84 mg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 134 (1961),332. | ||
| 9. | ipr-rat LD50:58 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 19 (1971),445. | ||
| 10. | scu-rat LD50:134 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 19 (1971),445. | ||
| 11. | <p class="carditemp |

