Products Categories
| CAS No.: | 482-89-3 |
|---|---|
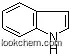
| Name: | Indigo |
| Article Data: | 244 |
| Molecular Structure: | |
|
|
|
| Formula: | C16H10N2O2 |
| Molecular Weight: | 262.268 |
| Synonyms: | Indigo PureBASF (6CI);[D2,2'-Biindoline]-3,3'-dione(8CI);Blue No. 201;C Blue 22;C.I. Pigment Blue 66;C.I. Vat Blue 1;Cystoceva;Diindogen;Indigo Blue;Indigo Ciba;IndigoCiba SL;Indigo J;Indigo N;Indigo NAC;Indigo Powder W;Indigo Pure BASF Powder K;Indigo Synthetic;Indigo VS;Indigotin;Indigotin (natural);Japan Blue 201;Lithosol Deep BlueB;Mitsui Indigo Paste;MonoliteFast Navy Blue BV;Natural Blue 1;Natural blue indigotin;Pigment Blue 66;Pigment Indigo;Pigment Indigo V;Reduced Dark Blue VB;Synthetic indigo;Vat Blue 1;Vulcafor BlueA;Vulcanosine Dark Blue L;[D2,2'(3H,3'H)-Biindole]-3,3'-dione;D2,2'-Bipseudoindoxyl; |
| EINECS: | 207-586-9 |
| Density: | 1.417 g/cm3 |
| Melting Point: | >300 °C(lit.) |
| Boiling Point: | 400.4 °C at 760 mmHg |
| Flash Point: | 158.2 °C |
| Solubility: | Water: <0.1 g/100 mL |
| Appearance: | dark violet powder |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/38-36/37/38 |
| Safety: | 26-36 |
| Transport Information: | UN 3264 8/PG 3 |
| PSA: | 58.20000 |
| LogP: | 3.09080 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine

| Conditions | Yield |
|---|---|
| In toluene for 1h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With tetraphosphorus decasulfide In pyridine at 85℃; for 0.0833333h; | 90% |
| Multi-step reaction with 2 steps 1: PCl5 / benzene / 4 h / Heating 2: 70 percent / thiophenol / benzene / 4 h / Heating; further thiophenols used: 1) p-CH3C6H4SH, 2) p-ClC6H4SH, 3) m-CH3C6H4SH, 4) o-CH3C6H4SH View Scheme | |
| Multi-step reaction with 2 steps 1: benzene; phosphorus pentachloride 2: zinc dust; glacial acetic acid View Scheme |
- 16800-68-3
1-acetyl-2,3-dihydro-1H-indol-3-one

- 16800-67-2
N,O-diacetylindoxyl

A
- 110912-08-8
1,2-diacetyl-3-indolinone

B
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic anhydride at 20℃; for 20h; | A 88.8% B n/a |

- 110912-09-9
1-acetyl-2-propionyl-3-indolinone

A
- 110912-10-2
1-(3-Hydroxy-1H-indol-2-yl)-propan-1-one

B
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 2h; | A 86.4% B n/a |
- 110912-08-8
1,2-diacetyl-3-indolinone

A
- 22079-15-8
1-(3-Hydroxy-1H-indol-2-yl)ethanon

B
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 2h; | A 85.7% B n/a |
- 16800-67-2
N,O-diacetylindoxyl

- 123-62-6
propionic acid anhydride

A
- 110912-09-9
1-acetyl-2-propionyl-3-indolinone

B
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic anhydride | A 72% B n/a |


| Conditions | Yield |
|---|---|
| With thiophenol In benzene for 4h; Heating; further thiophenols used: 1) p-CH3C6H4SH, 2) p-ClC6H4SH, 3) m-CH3C6H4SH, 4) o-CH3C6H4SH; | 70% |
| With hydrogen iodide; acetic acid | |
| With acetic acid; zinc | |
| With ammonium sulfide | |
| With phosphorous |
- 110912-11-3
1-acetyl-2-benzoyl-3-indolinone

A
- 22078-82-6
2-benzoyl-3-hydroxyindole

B
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 20℃; for 20h; | A 67.8% B n/a |
- 144646-98-0
1-(phenylsulfonyl)-2-trimethylsilylindol-3-yl trifluoromethanesulfonate

- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran | 51% |
- 78551-19-6
2,3-dioxo-2,3-dihydro-1H-indole-5-sulfonate

A
- 482-89-3
1H,1H'-2,2'-Biindolylidene-3,3'-dione

| Conditions | Yield |
|---|---|
| With N,N'-bis(6A-deoxy-β-cyclodextrin-6A-yl)NHCOCONH In water at 20℃; pH=10.0; Product distribution; Further Variations:; Reagents; | A 1.8% B 36% |

- 483-04-5Raubasine
- 4834-61-14-Benzoyl Acetanilide
- 484-16-29(10H)-Phenanthrenone
- 486-25-99-Fluorenone
- 486-89-5ANAGYRINE
- 487-51-44-CARBETHOXY-3-METHYL-2-CYCLOHEXEN-1-ONE
- 4877-84-3methyl 2,4-dichloro-3-oxobutyrate
- 488-30-2D-ARABINONIC ACID
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Consensus Reports
Reported in EPA TSCA Inventory.
Specification
The Indigo with the cas number 482-89-3, is also called (2E)-2-(3-oxo-1H-indol-2-ylidene)-1H-indol-3-one named by IUPAC. And it's systemic name are(1)(delta(sup 2,2')-Biindoline)-3,3'-dione; (2)(delta2,2'-Biindoline)-3,3'-dione (8CI) ; (3)2-(1,3-Dihydro-3-oxo-2H-indazol-2-ylidene)-1,2-dihydro-3H-indol-3-one ; (4)2-(1,3-Dihydro-3-oxo-2H-indol-2-ylidene)-1,2-dihydro-3H-indol-3-one ; (5)3H-Indol-3-one, 2-(1,3-dihydro-3-oxo-2H-indol-2-ylidene)-1,2-dihydro-. It belongs to rganics product categories. Besides, it seems like dark blue powder with coppery luster. Indigo occurs in isomeric forms (cis and trans). It is Stable but incompatible with strong oxidizing agents. Water solubility is lower than other edible pigment around 25 ° c. 0.05% solution appears blue. It is soluble in glycerin, propylene glycol, slightly soluble in ethanol, insoluble in grease. It shows darkblue when encounter sulfuric acid, and appear to be blue when diluted. with sodium hydroxide in solution, it turns green and yellow green. And it is sensitive to heat and light. Indigo is used as a dye for cotton yarn, which is mainly for the production of denim cloth for blue jeans. Small amounts are used for dying wool and silk.
Physical properties about this chemical are: (1)ACD/LogP: 3.72 ; (2)# of Rule of 5 Violations: 0 ; (3)ACD/LogD (pH 5.5): 3.72 ; (4)ACD/LogD (pH 7.4): 3.72 ; (5)ACD/BCF (pH 5.5): 395.55 ; (6)ACD/BCF (pH 7.4): 395.55 ; (7)ACD/KOC (pH 5.5): 2515.82 ; (8)ACD/KOC (pH 7.4): 2515.82 ; (9)#H bond acceptors: 4 ; (10)#H bond donors: 2 ; (11)#Freely Rotating Bonds: 0 ; (12)Polar Surface Area: 40.62Å2 ; (13)Index of Refraction: 1.709 ; (14)Molar Refractivity: 72.22 cm3 ; (15)Molar Volume: 185 cm3 ; (16)Polarizability: 28.63 ×10-24cm3 ; (17)Surface Tension: 63.4 dyne/cm ; (18)Density: 1.417 g/cm3 ; (19)Flash Point: 158.2 °C ; (20)Enthalpy of Vaporization: 65.13 kJ/mol ; (21)Boiling Point: 400.4 °C at 760 mmHg ; (22)Vapour Pressure: 1.27E-06 mmHg at 25°C
Prepration of Indigo: Indigo powder sulfonation by sulfuric acid, diluted by water, neutralization with sodium carbonate, refined by salting-out. Indigo powder (100%) 210kg can be produced at the cost of every tons product. It is an synthesis edible pigment and applied to food, medicine and cosmetics.
1. N-phenylglycine is treated with a molten mixture of sodium hydroxide, potassium hydroxide, and sodamide. This highly sensitive melt produces indoxyl, which is subsequently oxidised in air to form indigo.

2. An alternative and also viable route to indigo involves heating N-(2-carboxyphenyl)glycine to 200 °C in an inert atmosphere with sodium hydroxide. Indoxyl-2-carboxylic acid is generated. This material readily decarboxylates to give indoxyl, which oxidises in air to form indigo.

When you are using this chemical, please be cautious about it as the following: It is quite irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Before you are using it, please wear suitable protective clothing. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure :
(1).SMILES: O=C/4c1ccccc1NC\4=C3\C(=O)c2ccccc2N3
(2).InChI:InChI=1/C16H10N2O2/c19-15-9-5-1-3-7-11(9)17-13(15)14-16(20)10-6-2-4-8-12(10)18-14/h1-8,17-18H/b14-13+
Toxic information of Indigo can be showed as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 2200mg/kg (2200mg/kg) | "Zhongliu Yanjiu" Cancer Review, Yu, R., et al., eds., Shanghai Science/Technology Publisher,Peop. Rep. China, 1994Vol. -, Pg. 173, 1994. | |
| mouse | LD50 | oral | > 32gm/kg (32000mg/kg) | "Zhongliu Yanjiu" Cancer Review, Yu, R., et al., eds., Shanghai Science/Technology Publisher,Peop. Rep. China, 1994Vol. -, Pg. 173, 1994 |