Products Categories
| CAS No.: | 62-73-7 |
|---|---|
| Name: | Dichlorvos |
| Molecular Structure: | |
|
|
|
| Formula: | C4H7Cl2O4P |
| Molecular Weight: | 220.977 |
| Synonyms: | o- (2, 2-Dichlorvinyl)-O,O-dimethylphosphat;Equigel;DDVP;Task;Bibesol;Dichlorman;Dimethyl dichlorovinyl phosphate;(2, 2-Dichloro-vinil)dimetil-fosfato;Phosphoric acid 2,2-dichlorovinyl dimethyl ester;Dichlorfos;Dichlorovas;Divipan;Chlorvinphos;Nerkol;Atgard V;Estrosel;Nuvan;Dimethyl O, O-dichlorovinyl-2,2-phosphate;Phosphoric acid,esters,2,2-dichloroethenyl dimethyl ester;TAP 9VP;Tenac;Panaplate;Phosphoric acid, 2,2-dichlorovinyl dimethyl ester;DDVF;o-(2, 2-Dichlorvinyl)-O,O-dimethylphosphat;Vinyl alcohol, 2,2-dichloro-, dimethyl phosphate;Winylophos;(2, 2-Dichlor-vinyl)-dimethyl-phosphat;Lindanmafu;Novotox;Cekusan;Unifos;Brevinyl E-50;Atgard;2,2-Dichlorovinyl dimethyl phosphate;Herkol;Dimethyldichlorovinyl phosphate;Dimethyl 2,2-dichloroethenyl phosphate;Insectigas D;Atgard C;OKO;Herkal;(2,2-Dichloor-vinyl)-dimethyl-fosfaat (Dutch);Vinylofos;Nogos G;Brevinyl E 50;Dichlorvos(DDVP); |
| EINECS: | 200-547-7 |
| Density: | 1.435 g/cm3 |
| Melting Point: | -60 °C |
| Boiling Point: | 176.8 °C at 760 mmHg |
| Flash Point: | 14.7 °C |
| Solubility: | slightly soluble in water |
| Appearance: | an aromatic colorless to amber liquid |
| Hazard Symbols: |
 T, T,  T+, T+,  N N
|
| Risk Codes: | 24/25-26-43-50 |
| Safety: | 1/2-28-36/37-45-61 |
| Transport Information: | UN 2786 |
| PSA: | 54.57000 |
| LogP: | 2.68040 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- Total:2 Page 1 of 1 1
Chemistry
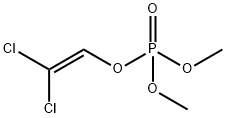
IUPAC Name: 2,2-dichloroethenyl dimethyl phosphate
Empirical Formula: C4H7Cl2O4P
Molecular Weight: 220.9757g/mol
EINECS: 200-547-7
Structure of Dichlorvos (CAS NO.62-73-7):
Index of Refraction: 1.461
Molar Refractivity: 42.28 cm3
Molar Volume: 153.9 cm3
Polarizability: 16.76×10-24cm3
Surface Tension: 38.1 dyne/cm
Density: 1.435 g/cm3
Flash Point: 14.7 °C
Enthalpy of Vaporization: 39.61 kJ/mol
Melting Point: -60°C
Boiling Point: 176.8 °C at 760 mmHg
Vapour Pressure: 1.45 mmHg at 25°C
Water Solubility: Slightly soluble. 1 g/100 mL
Product Categories: Herbicide;INSECTICIDE;AcaricidesPesticides&Metabolites;Alpha sort;D;DAlphabetic;DIA - DICMethod Specific;Endocrine Disruptors (Draft)Pesticides;EPA;Insecticides;Organophorous;Pesticides;Pesticides&Metabolites;AcaricidesAlphabetic
Canonical SMILES: COP(=O)(OC)OC=C(Cl)Cl
InChI: InChI=1S/C4H7Cl2O4P/c1-8-11(7,9-2)10-3-4(5)6/h3H,1-2H3
InChIKey: OEBRKCOSUFCWJD-UHFFFAOYSA-N
Synonyms of Dichlorvos (CAS NO.62-73-7): MAFU(R) ; Atgard;astrobot ; BAYER 19149 ; canogard ; HERKOL(R) ;
DICHLORVOS ; DICHLORVOS (DIMETHYL-D6)
Uses
Dichlorvos (CAS NO.62-73-7): is widely used as a fumigant to control household pests, in public health, and protecting stored product from insects. It is effective against mushroom flies, aphids, spider mites, caterpillars, thrips, and whiteflies in greenhouse, outdoor fruit, and vegetable crops. It is also used in the milling and grain handling industries. Dichlorvos is used to treat a variety of parasitic worm infections in dogs, livestock, and humans. It is fed to livestock to control bot fly larvae in the manure. It is also used in pet collars and "no-pest strips" as pesticide-impregnated plastic.
Toxicity Data With Reference
| 1. | dns-hmn:oth 65 mmol/L | PSSCBG Pesticide Science. 15 (1984),439. | ||
| 2. | dni-hmn:lym 62 mg/L | TUMOAB Tumori. 66 (1980),425. | ||
| 3. | cyt-ham-ipr 3 mg/kg | ARTODN Archives of Toxicology. 58 (1985),152. | ||
| 4. | orl-rat TDLo:4120 mg/kg/2Y-C:NEO | NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-342 ,1989. | ||
| 5. | orl-rat TDLo:2060 mg/kg/2Y-C:CAR | JJCREP Japanese Journal of Cancer Research (Gann). 82 (1991),157. | ||
| 6. | orl-mus TDLo:20,600 mg/kg/2Y-C:CAR | NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-342 ,1989. | ||
| 7. | orl-rat LD50:17 mg/kg | JPIFAN Japan Pesticide Information.(13),(1972),36. | ||
| 8. | ihl-rat LC50:15 mg/m3/4H | GISAAA Gigiena i Sanitariya. 33 (12)(1968),35. | ||
| 9. | skn-rat LD50:70,400 µg/kg | APYPAY Acta Physiologica Polonica. 32 (1981),507. | ||
| 10. | ipr-rat LD50:23,300 µg/kg | IJPPAZ Indian Journal of Physiology and Pharmacology. 31 (1987),19. |
Consensus Reports
IARC Cancer Review: Group 2B IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 53 (1991),p. 267.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Sufficient Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 53 (1991),p. 267.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 20 (1979),p. 97.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Human No Adequate Data IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 20 (1979),p. 97.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Human Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 53 (1991),p. 267.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . NCI Carcinogenesis Bioassay (feed); No Evidence: mouse, rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-10 ,1977. . EPA Genetic Toxicology Program. Community Right-To-Know List. EPA Extremely Hazardous Substances List.
Safety Profile
Confirmed carcinogen with carcinogenic and tumorigenic data. Poison by ingestion, inhalation, skin contact, subcutaneous, intravenous, and intraperitoneal routes. Experimental teratogenic and reproductive effects. Human mutation data reported. A cholinesterase inhibitor, it is used in flea (pest) collars for pets. No neurotoxicity has been observed. It is very rapidly metabolized and excreted. When heated to decomposition it emits very toxic fumes of Cl− and POx. See also PARATHION.
Hazard Codes:  T;
T; N,
N, T+
T+
Risk Statements: 24/25-26-43-50
R24:Toxic in contact with skin.
R25 :Toxic if swallowed.
R26:Very toxic by inhalation.
R43:May cause sensitization by skin contact.
R50:Very toxic to aquatic organisms.
Safety Statements: 1/2-28-36/37-45-61
S1/2:Keep locked up and out of the reach of children.
S28:After contact with skin, wash immediately with plenty of soap-suds.
S36/37:Wear suitable protective clothing and gloves.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
RIDADR: 2786
RTECS: TC0350000
HazardClass: 6.1(a)
PackingGroup: II
Hazardous: Substances Data 62-73-7(Hazardous Substances Data)
Standards and Recommendations
OSHA PEL: TWA 1 mg/m3 (skin)
ACGIH TLV: TWA 0.1 ppm (skin, sensitizer); Not Classifiable as a Human Carcinogen
DFG MAK: 0.1 ppm (1 mg/m3)
Analytical Methods
For occupational chemical analysis use OSHA: #ID-62.

