127045-41-4 Usage
Description
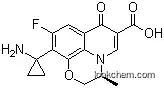
Pazufloxacin is a fluoroquinolone antibiotic with a unique 1-aminocyclopropyl substituent at the C10 position, which confers potent broad-spectrum activity against both gram-positive and gram-negative bacteria. It is marketed in Japan for the treatment of bacterial infections and is synthesized from commercially available 2,3,4,5-tetrafluorobenzoic acid. Pazufloxacin works by inhibiting bacterial DNA gyrase, essential for bacterial DNA replication, and has demonstrated effectiveness against various resistant strains of bacteria.
Uses
Used in Pharmaceutical Industry:
Pazufloxacin is used as an injectable antibiotic for the treatment of bacterial infections, particularly against cephalosporin-resistant, carbapenem-resistant, and aminoglycoside-resistant strains of bacteria. Its broad-spectrum activity and potent inhibition of bacterial DNA gyrase make it a valuable addition to the arsenal of antibiotics for combating drug-resistant infections.
Additionally, pazufloxacin has shown potential as an antimicrobial and/or antiviral agent, further expanding its applications in the pharmaceutical industry.
Used in Clinical Trials:
Pazufloxacin has been tested in clinical trials for patients with urinary tract infections and, to a lesser extent, respiratory tract infections. Good clinical responses have been observed, indicating its effectiveness in treating these types of infections.
Brand Names:
Pazufloxacin is marketed under the brand names Pasil and Pazucross.
References
[1] A. Vora, Pazufloxacin, tracked from www.japi.org on 16.07.2017
[2] Jeffrey K. Aronson, Meyler’s Side Effects of Antimicrobial Drugs, 2009
[3] Satoshi Watabe, Yoshiaki Yokoyama, Kazuyuki Nakazawa, Kimikazu Shinozaki, Rika Hiraoka, Kei Takeshita and Yukio Suzuki, Simultaneous measurement of pazufloxacin, ciprofloxacin, and levofloxacin in human serum by high-performance liquid chromatography with fluorescence detection, Journal of Chromatography B, 2010, vol. 878, 1555-1561
Originator
Toyama (Japan)
Pharmaceutical Applications
A tricyclic fluoroquinolone, formulated as mesylate and hydrochloride salts for oral or parenteral use or as a methane sulfonate (eye ointment).It displays good activity in vitro against methicillin
susceptible Staph. aureus (MIC 0.2 mg/L), but is inactive against Str. pyogenes, Str. pneumoniae (MIC ≥4 mg/L) and enterococci. L. pneumophila is inhibited by 0.03 mg/L. Activity against Enterobacteriaceae, fastidious Gram-negative bacilli, Ps. aeruginosa and Acinetobacter spp. is similar to that of ofloxacin. It is weakly active against Sten. maltophilia and Burkholderia cepacia (MIC c. 2 mg/L). Against M. tuberculosis, MICs range from 0.8 to 4 mg/L. It is inactive against anaerobes.After oral doses of 100 or 400 mg, peak plasma concentrations range from 0.94 mg/L (100 mg) to 4.5 mg/L (400 mg) after <1 h. The apparent elimination half-life is around 2 h. Most of the administered dose is eliminated in urine, about 70% within 24 h. Four metabolites have been reported. In elderly patients, according to the renal function, the peak plasma concentration may be elevated (up to 5.6 mg/L) and significantly delayed (2–6 h). The plasma protein binding ranges from 17% to 28%.
Check Digit Verification of cas no
The CAS Registry Mumber 127045-41-4 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,2,7,0,4 and 5 respectively; the second part has 2 digits, 4 and 1 respectively.
Calculate Digit Verification of CAS Registry Number 127045-41:
(8*1)+(7*2)+(6*7)+(5*0)+(4*4)+(3*5)+(2*4)+(1*1)=104
104 % 10 = 4
So 127045-41-4 is a valid CAS Registry Number.
InChI:InChI=1/C16H15FN2O4/c1-7-6-23-14-11(16(18)2-3-16)10(17)4-8-12(14)19(7)5-9(13(8)20)15(21)22/h4-5,7H,2-3,6,18H2,1H3,(H,21,22)
127045-41-4Relevant articles and documents
Pyridonecarboxylic acids as antibacterial agents. VIII. Synthesis and structure-activity relationship of 7-(1-aminocyclopropyl)-4-oxo-1,8-naphthyridine-3-carboxylic acids and 7-(1-aminocyclopropyl)-4-oxoquinoline-3-carboxylic acids
Todo,Nitta,Miyajima,Fukuoka,Yamashiro,Nishida,Saikawa,Narita
, p. 2063 - 2070 (2007/10/02)
4-Oxo-1,8-naphthyridine- and 4-oxoquinoline-3-carboxylic acids (2a, b and 3a-l) possessing a 1-aminocyclopropyl group at the 7-position have been synthesized and evaluated for in vitro antibacterial activities. The three quinolones (3d, h, i) exhibited potent antibacterial activities against both gram-positive and gram-negative bacteria, which are comparable to those of ciprofloxacin (CPFX) and ofloxacin (OFLX). Among the three compounds, the best pharmacological and pharmacokinetic profile was obtained with 3i, an OFLX analogue, which was considerably less toxic than three reference quinolones (1, CPFX, and OFLX).