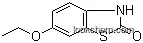
72680-01-4Relevant articles and documents
Hydroxylamine as an oxygen nucleophile: Substitution of sulfonamide by a hydroxyl group in benzothiazole-2-sulfonamides
Kamps, Jos J. A. G.,Belle, Roman,Mecinovi?, Jasmin
, p. 1103 - 1108 (2013/03/28)
Benzothiazole-2-sulfonamides react with an excess of hydroxylamine in aqueous solutions to form 2-hydroxybenzothiazole, sulfur dioxide, and the corresponding amine. Mechanistic studies that employ a combination of structure-reactivity relationships, oxygen labeling experiments, and (in)direct detection of intermediates and products reveal that the reaction proceeds via oxygen attack, and that oxygen incorporated in the 2-hydroxybenzothiazole product derives from hydroxylamine. The reaction, which is performed under mild conditions, can be used as a deprotection method for cleavage of benzothiazole-2-sulfonyl-protected amino acids.
Suppressing pain with benzothiazol-2(3H)-ones
-
, (2008/06/13)
There are described pharmaceutical compositions containing as active substances benzothiazol-2(3H)-ones of the general formula STR1 wherein R represents a hydrogen atom or a methyl, methoxy, or ethoxy group. The pharmaceutical compositions serve as analgesics and antipyretics and possess little or no methemoglobin-forming activity.