
Canadian Journal of Chemistry p. 414 - 425 (1998)
Update date:2022-08-11
Topics:
 Hoveyda
Hoveyda
 Karunaratne, Veranja
Karunaratne, Veranja
 Nichols, Christopher J.
Nichols, Christopher J.
 Rettig, Steven J.
Rettig, Steven J.
 Stephens, Ashley K. W.
Stephens, Ashley K. W.
 Orvig, Chris
Orvig, Chris
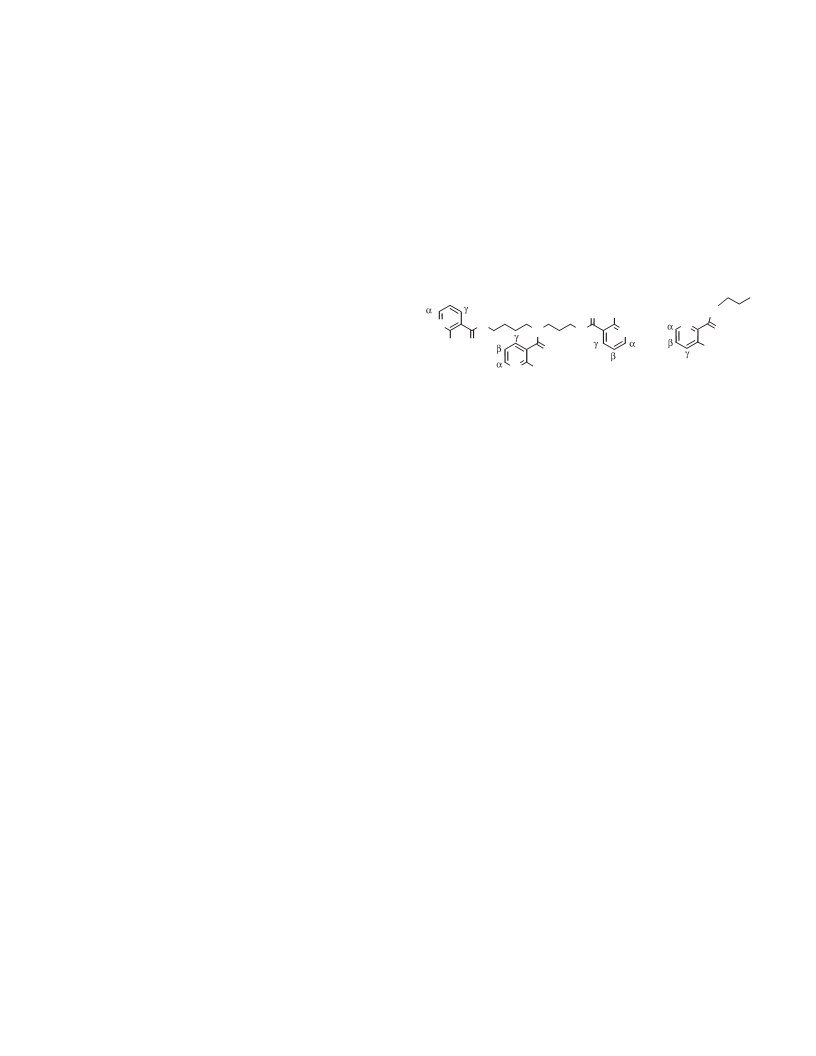
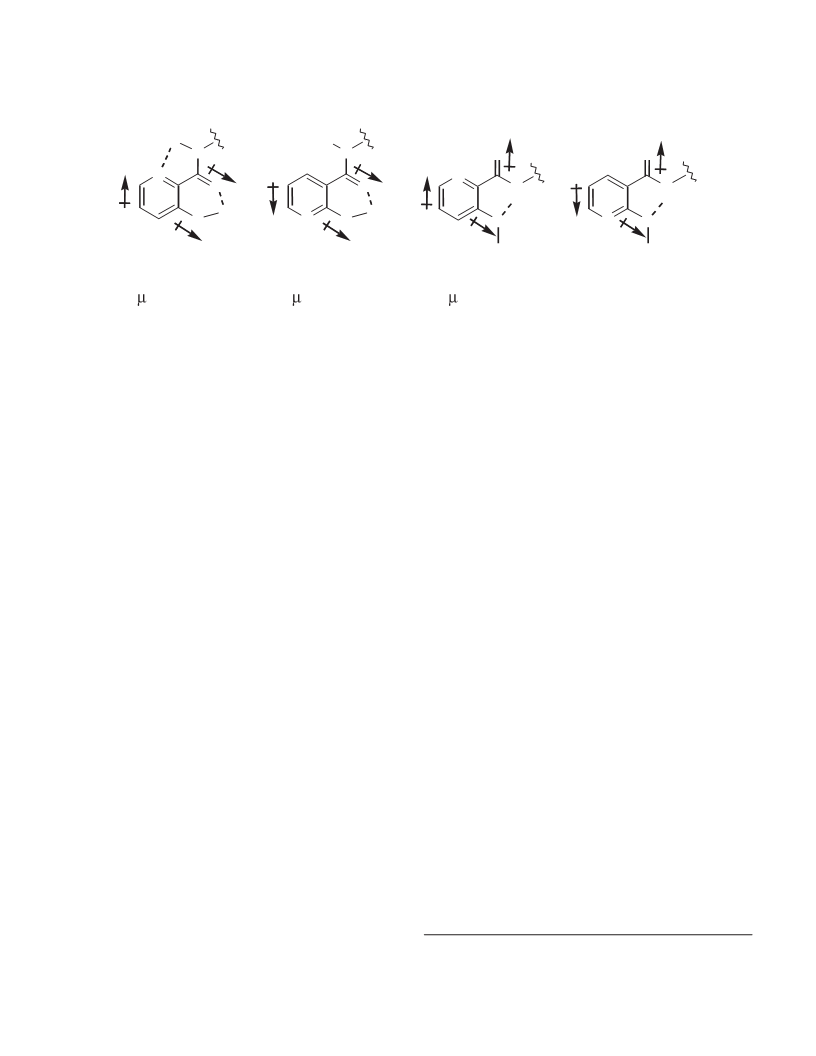
A number of polydentate arylamide ligands have been prepared by coupling various acyclic tripodal or linear polyamines with derivatives of nicotinic and picolinic acids. Two synthetic procedures were utilized; tris{[(2-hydroxynicotinyl)carbonyl]-2-aminoethyl}amine (H3NICTREN) was prepared by Method A, the HOSu/DCC method, and the other arylamides in this study were prepared by Method B, the CDI method. Method A involved the reaction of N-hydroxysuccinimide with 2-hydroxynicotinic acid (in the presence of dicyclohexylcarbodiimide (DCC) as a dehydrative coupling reagent) to form the succinimide ester, followed by reaction with TREN to yield H3NICTREN. Method B involved reaction of a carboxylic acid (2-hydroxynicotinic, 3-hydroxypicolinic, nicotinic, or picolinic acids) with carbonyldiimidazole (CDI) to form the N-acylimidazolide, followed by reaction with the amine (TREN, TAME, spermidine, or TRPN) to yield the desired arylamide. The X-ray structure of 1,1,1-tris([(3-hydroxypicolinyl)carbonyl]-2-aminomethyl)ethane (H3PICTAME) was determined; crystals of H3PICTAME are monoclinic, a = 10.257(2), b = 15.572(3), c = 15.208(2) A, β = 96.124(15)°, Z = 4, space group P21/a. The structure was solved by direct methods and refined by full-matrix least-squares procedures to R = 0.041 and Rw = 0.038 for 2506 reflections with I ≥gt; 3σ(I). In the solid state, H3PICTAME contains an extensive hydrogen-bonding network, with eight intra- and one intermolecular H-bonds per molecule; the ligand is partially preorganized for metal ion chelation. The acid dissociation constants of H3NICTREN and those of 1,1,1-tris{[(2-hydroxynicotinyl)carbonyl]-2-aminomethyl}ethane (H3NICTAME) have been determined; pKa1 = 11.2 (10.68), pKa2 = 10.7 (10.58), pKa3 = 10.0 (9.71), and pKa4 = 6.25 for H3NICTREN (H3NICTAME); the high phenolic pKa's are consistent with the hydrogen bonding observed in the solid state.
View More









Zhejiang Tianyu Pharmaceutical Co., Ltd.
Contact:+86-576-84177669, 89189665,89189688,84168770
Address:Jiangkou Development Zone, Huangyan, Taizhou City, Zhejiang
SHANDONG ZHANHUA YONGHAO PHARMACEUTICAL TECH.CO.,LTD
Contact:+86-576-88685096
Address:GENGJU VILLAGE NORTH ONE KILOMETER,ZHANHUA DISTRICT,BINZHOU CITY,SHANDONG PROVINCE,CHINA.
Changzhou Welton Chemical Co., Ltd,
Contact:0086-519-85910828,85920537,85912897
Address:No.8 Jinlong Road, Binjiang Park, Changzhou, Jiangsu, China.
JIAXING SUNS INTERNATIONAL TRADE CO LTD
Contact:18367306858
Address:No.123,Huixin Avenue,Huimin Dist
Contact:+86-710-3516804
Address:Number 83,Panggong road,Xiangcheng District,Xiangyang ,Hubei
Doi:10.3390/molecules24010047
(2019)Doi:10.1016/j.ejmech.2019.111902
(2020)Doi:10.1089/152308603322110832
(1936)Doi:10.1002/jhet.3731
(2019)Doi:10.1002/hlca.19490320519
(1949)Doi:10.1039/b308042k
(2003)