R. Shavi et al. / Molecular Catalysis 445 (2018) 232–239
233
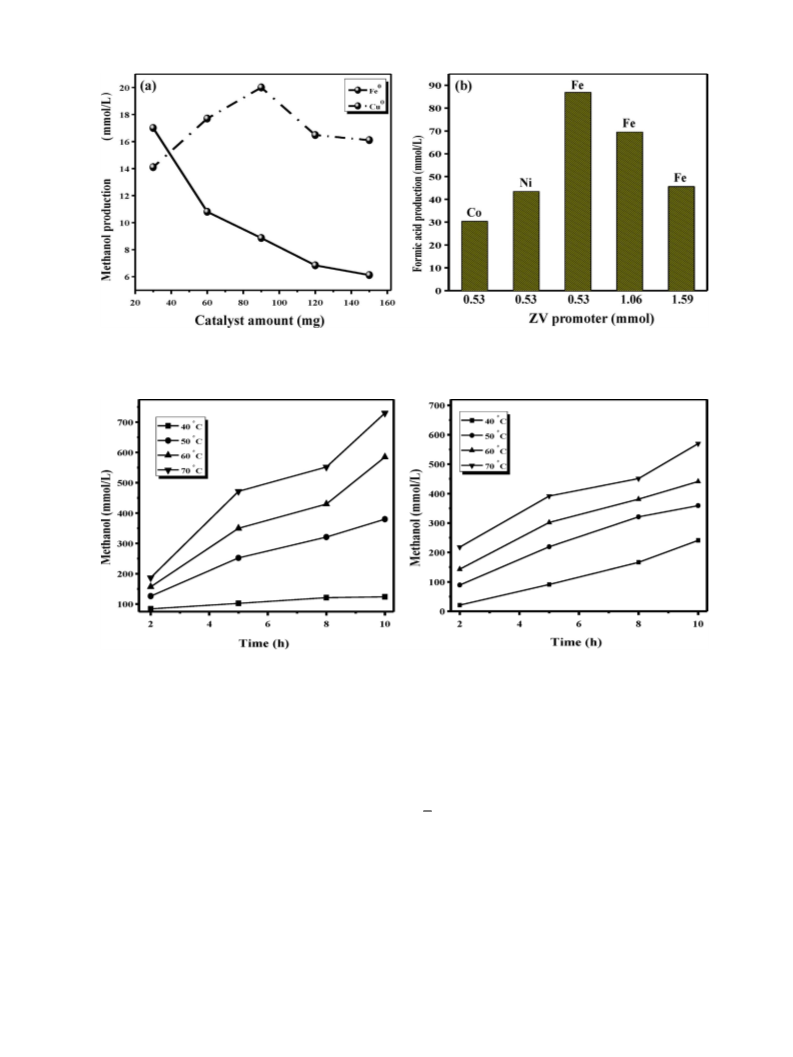
Table 1
Methane oxidation results obtained over various catalysts under different reaction conditions.
ZV catalyst (mmol)
Missed reaction
component
Yield produced (mmol/L)
Selectivity (%)a
Methanol
Methanol
Formic acid
Formic acid
CH4 conversion (%)b
Cu0 (0.53)
K2S2O8
IL
0.15
0.25
2.9
8.25
17
9.6
2.65
–
–
–
–
–
–
–
–
–
0
0
42
48
32
59
80
100
100
100
100
100
negligible
negligible
0.16
0.44
0.70
0.60
0.30
2.80
2.20
Cu0 (0.53)
–
Cu0
2.1
5.5
5.6
9
7.3
87
70
46
43
30
58
52
68
41
20
0
0
0
0
0
Cu0 (0.53)
none
none
none
none
none
none
none
none
none
Fe0 (0.53)
Ni0 (0.53)
Co0 (0.53)
Cu0: Fe:1.4:0.53
Cu0: Fe:1.4:1.06
Cu0: Fe:1.4:1.59
Cu0: Ni:1.4:0.53
Cu0: Co:1.4:0.53
1.50
1.40
0.80
Reaction conditions: Temperature–60 ◦C, Pressure–20 bar, K2S2O8–0.5 mmol, IL–0.5 mmol.
a
Selectivity = Moles of target oxygenate/moles of other oxygenates × 100.
b
CH4 conversion = Total moles of oxygenates produced/initial moles of CH4 × 100.
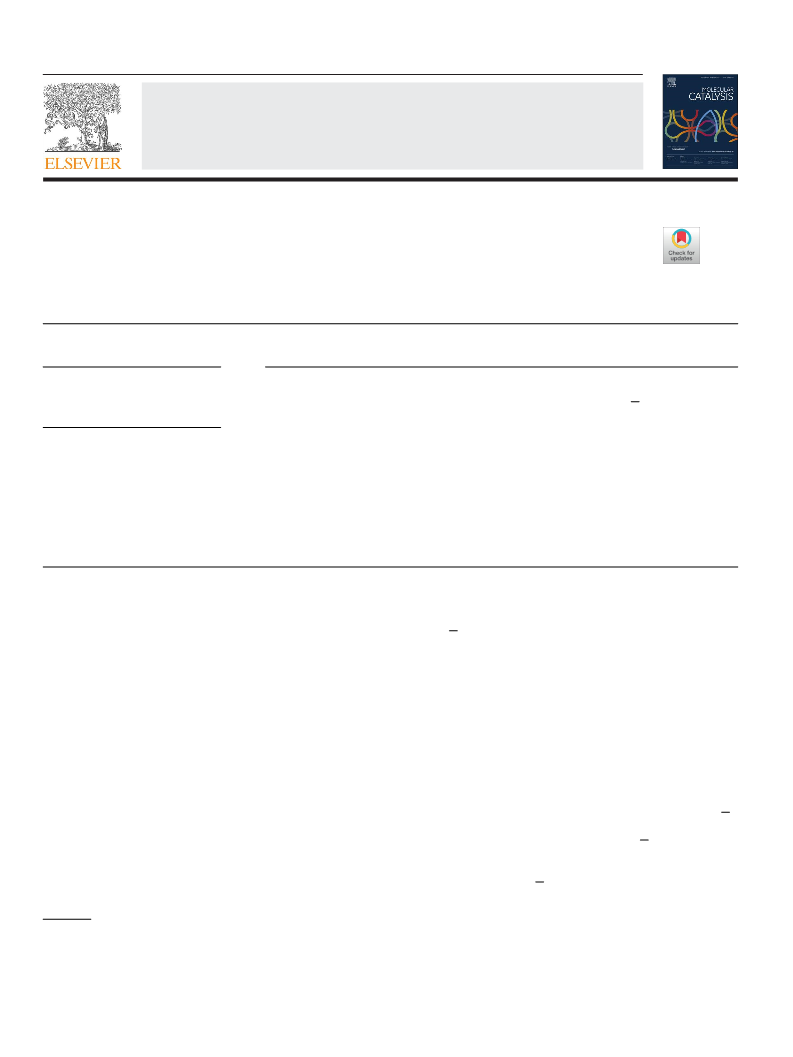
a very high oxidation potential (3.1 V). Previously, K2S2O8 was
reported to oxidize methane at temperatures as low as 80 ◦C [21].
Hence, establishing a radical-based mechanism for the methane
oxidation reaction would be quite worthwhile.
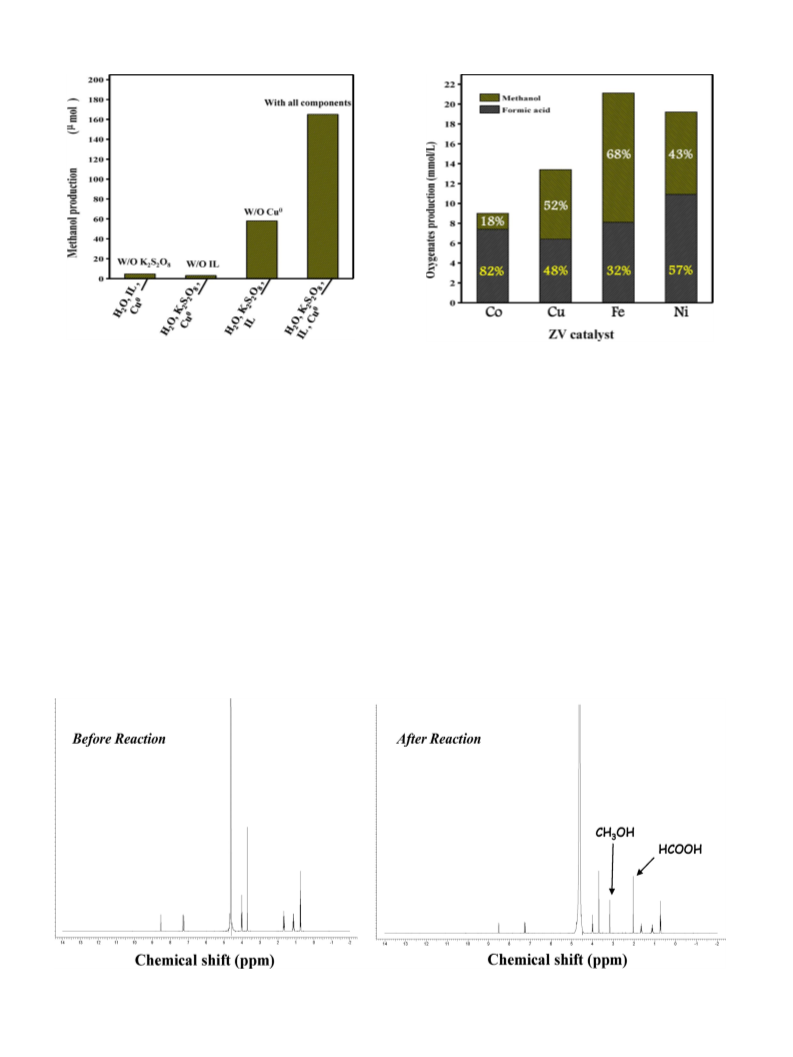
2.2. Characterization
Fourier transform infrared (FTIR) spectroscopy was obtained
using a Varian 2000 Fourier transform infrared spectrophotome-
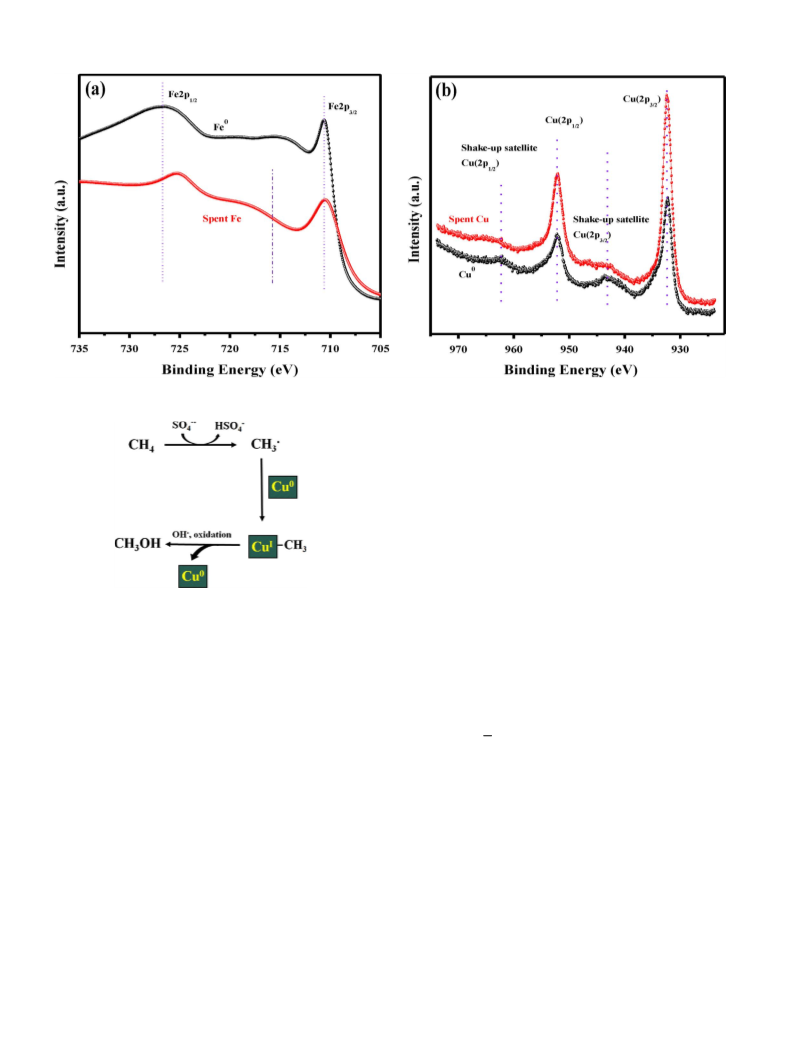
Metallic catalysts or zero-valent (ZV) metals are highly reactive
ingredients in radical-based chemical reactions. They are known to
activate persulfate and enhance reactivity when added to a reac-
tion that takes the radicalpathwayto form products[22]. Hence, the
addition of ZV metals as catalysts for methane oxidation by reaction
ter by accumulating 32 scans between 2000 and 4000 cm−1 1H
.
nuclear magnetic resonance (NMR) spectra were recorded on a
Bruker Advance II+ 400 MHz spectrometer. X-ray diffraction (XRD)
spectra were obtained at 2 between 10 and 90◦. A Cu anode was
used and continuous scans were taken for each sample. The instru-
ment was operated at a rate of 400 MHz with a spin rate of 13 kHz.
Electrospray-ionization mass spectra (ESI–MS) were measured in
the range 50–30 m/z with a SYNAPT G2 (Waters, U.K.) instrument.
A Perkin-Elmer Lambda 25 ultraviolet-visible (UV–vis) spectrom-
eter was used to analyze samples by absorption spectroscopy in
the wavelength range 800–200 nm. High-performance liquid chro-
matography (HPLC) on an Ultimate 3000 (Dionex, USA) was used to
analyze the products in solution. An Aminex 87H (Bio-Rad) column
with oven temperature 40 ◦C and refractive index (RI) (ERC refrac-
toMAX520) detector was used. The eluent was H2SO4 (0.01 N) at
flow rate 0.5 mL/min. Gas chromatography technique was used to
detect gaseous products. The instrument used was Agilent 7890A
TCD/FID.
−
with K2S2O8 would maximize the concentration of SO4 radicals,
which act as a fuel for the reaction to achieve higher conversion of
methane. Although radicals are extremely reactive, which is vital to
degrading methane, their low stability is always an issue. Radicals
need to be stable or possess great longevity that avoids their mutual
coupling leading to form unwanted, less oxidation potential rad-
icals. 1-Butyl-3-methylimidazolium chloride ([BMIM]+Cl−), a big
and dense IL molecule, is known to form carbine radicals at its acidic
proton site [23]. Hence, utilizing denser ionic liquids to increase the
longevity of radicals by forming them on a bulkier molecule could
allow the reaction to consume the radical fuel efficiently.
In this work, methane oxidation was carried out in a water-
diluted IL medium using K2S2O8 and ZV metal as oxidizer and
catalyst, respectively. The reaction proceeds at a temperature as
low as 60 ◦C to produce concentrated methanol. The methanol for-
mation mechanism was found to be radical-based, confirmed by
radical trapping experiments carried out in the presence of CBrCl3
(a radical trapper). ZV metals, when used as a catalyst, were capable
of abruptly increasing methane conversion, possibly by maximizing
radical fuels. Hence, applying this strategy of using radical fuels to
degrade methane to form methanol could further inspire research
into methane oxidation.
2.3. Catalytic activity tests
A 75 mL reactor fitted with a quartz vessel was used for all the
reactions. The quartz vessel was washed with diluted HNO3 before
each experiment to avoid any effects of metal impurities on the
reaction. In a typical reaction, 20 mL of doubly distilled water was
added into the vessel followed by addition of [BMIM]+Cl− ionic liq-
uid. In order to avoid any unnecessary exothermic side reactions,
mixture was cooled in a refrigerator, maintained at 4–5 ◦C, for 5 min
before the addition of K2S2O8 (0.5 mmol). The required amount of
metallic catalyst was added into the vessel just before fixing it to the
reactor. Flushing with nitrogen for 3 min for removal of air from the
reaction system. Later, the reactor outlet was closed and methane
was purged until 20 bar pressure was obtained inside the reactor.
The reactor was covered with a heating jacket. The reaction was
carried out for 10 h, unless mentioned, at 60 ◦C. After the reaction,
the heating jacket was removed and the system was allowed to cool
naturally; finally, it was ice cooled to condense gaseous products.
The gas was released slowly, and a sample was collected in a gas
sampler for gas chromatography (GC) analysis. The liquid products
were analyzed by HPLC.
2. Material and methods
2.1. Materials
Iron powder (99%, <212 m size), copper powder (99.5%,
<425 m size), cobalt powder (99.9%, <150 m size), and nickel
powder (99.9%, <150 nm) were purchased from Sigma-Aldrich and
ground finely prior to use. 1-Butyl-3-methylimidazolium chlo-
ride ([BMIM]+Cl−, >98%) and potassium persulfate (K2S2O8, 99.0%)
were used as obtained from Sigma-Aldrich. High purity methane
(99.95%) and carbon dioxide (99.999%) were used for the reactions,
and high purity hydrogen (99.99%) was used as standard for gas
chromatography (GC) analysis. Double-distilled water was used for
all reactions.

 Shavi, Raghavendra
Shavi, Raghavendra
 Hiremath, Vishwanath
Hiremath, Vishwanath
 Seo, Jeong Gil
Seo, Jeong Gil