GModel
CATTOD-8786; No. of Pages6
ARTICLE IN PRESS
2
J. Zhang et al. / Catalysis Today xxx (2013) xxx–xxx
a selective oxidation catalyst for biomass conversion. In their sys-
tem, the highest yield of FA was 35% when cellulose was used as the
substrate [13]. By comparison, catalytic conversion of biomass
to AA has rarely been reported because of the low selectivity.
Although the production of AA from biomass can in principle
be realized through selective oxidation of bio-ethanol, a one-
step direct conversion process with a fair yield would be more
attractive.
80
70
60
50
40
30
20
10
0
Formic acid
Acetic acid
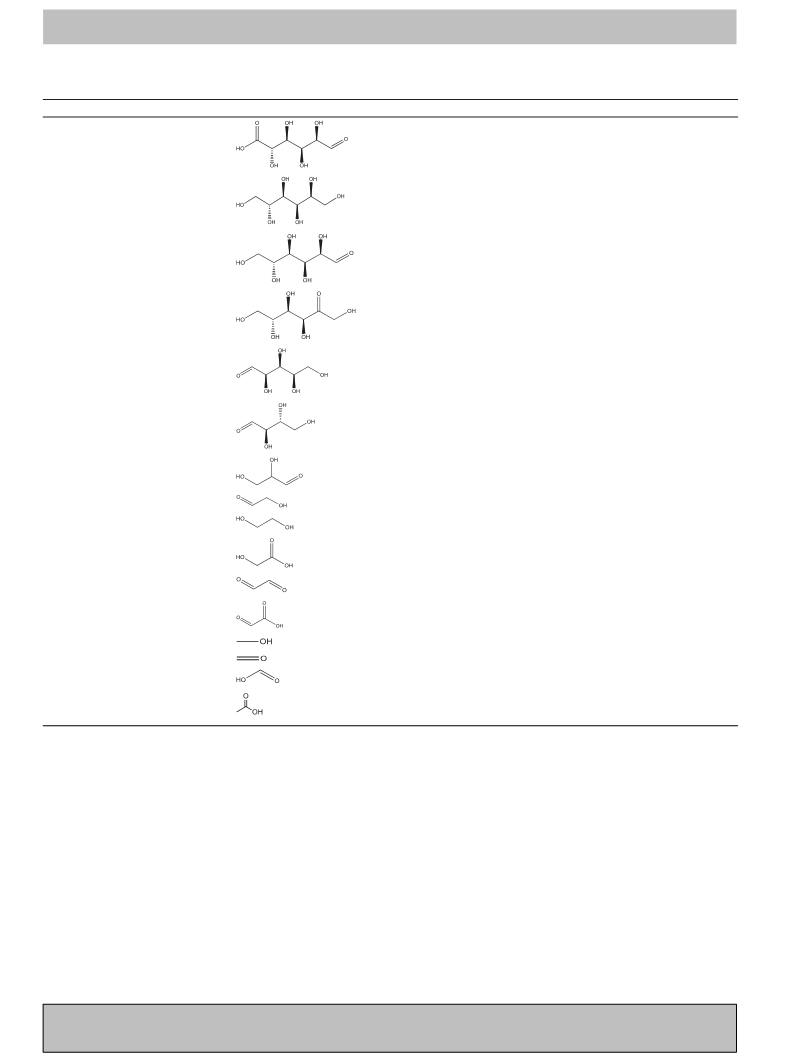
We recently reported the direct conversion of cellulose to gly-
colic acid with remarkably high yields (∼50%) using molecular
oxygen in a water medium, in which a phosphomolybdic acid
(H3PMo12O40) acts as a bi-functional catalyst to catalyze both
the hydrolysis of cellulose and the subsequent oxidation reac-
tions [27]. We have demonstrated that many heteropoly acids
(HPAs) can effectively convert cellulose under oxidative conditions
due to their strong Brönsted acidity, while the reaction path-
way is largely determined by the type of addenda atom in the
HPA catalyst. Here, we report the selective oxidation of cellulose
to FA and AA catalyzed by vanadium-substituted phosphomolyb-
dic acids (H3+nPVnMo12−nO40) under mild reaction conditions in
water using molecular oxygen (O2) as an oxidant. Our work is
different from the above-mentioned studies [10,13] of biomass
conversion by H5PV2Mo10O40 in three respects: (i) we found that
the concentration of vanadium (V) has an important influence
on the selectivity, i.e., H4PVMo11O40 could yield much more FA
than H5PV2Mo10O40; (ii) under optimized reaction conditions, a
FA yield of 67.8% was obtained from cellulose, which is almost
double the results achieved in previous studies; (iii) in addition
ously, giving a total yield as high as 81.2% (FA: 66.0% and AA:
15.2%). These results demonstrate that the incorporation of vana-
dium in a phosphomolybdic acid catalyst can significantly change
the reaction pathway by its special ability to cleave C–C bonds
of vicinal diols to carboxylic acids [30,31]. More importantly,
H4PVMo11O40 is also capable of converting crude cellulosic mate-
rials, such as bagasse and hay, to FA and AA with remarkable
yields.
Fig. 1. Yields of FA and AA produced from cellulose using different HPA catalysts:
H3PW12O40 (HPW), H3PMo12O40 (HPMo), H5PV2W12O40 (HPWV2), H4PVMo11O40
(HPMoV1), H5PV2Mo10O40 (HPMoV2), and H6PV3Mo9O40 (HPMoV3). Reaction con-
ditions: 0.2 g of ␣-Cellulose powder, 0.1 mmol of catalyst, at 453 K for 3 h, under
2 MPa O2. The catalyst amount was varied for HPMoV1 and HPMoV3 (the last two
columns): a 0.2 mmol of HPMoV1; b 0.066 mmol of HPMoV3.
3. Results and discussions
Previous studies have demonstrated that V atoms in the HPA
catalyst account for the selective oxidative cleavage of C–C bonds
in the conversion of carbohydrates to FA. However, it remains
unknown how the V concentration in the catalyst influence the
reaction selectivity, because only one catalyst (i.e. H5PV2Mo10O40
)
has thus far been investigated. To provide more insight into this,
we prepared three phosphovanadomolybdic acids with differ-
ent contents of vanadium, namely H4PVMo11O40, H5PV2Mo10O40
and H6PMo9V3O40, and evaluated their catalytic performance for
prepared another three kegging-type HPA catalysts including
two V-free HPAs (H3PW12O40 and H3PMo12O40) and one phos-
phovanadotungstic acid (H5PV2W12O40). Although relatively low
temperatures (∼373 K) were used to convert soluble carbohydrates
[10,13], a higher reaction temperature (>423 K) is essential to an
effective conversion of water-insoluble biomass (e.g. cellulose)
atmosphere of 2 MPa. In each reaction, 0.2 g of ␣-cellulose powder
containing 1.23 mmol glucose units was used as the substrate and
the catalyst load was 0.1 mmol. The yields of FA and AA for each cat-
alyst are shown in Fig. 1. As cellulose was fully converted by these
selected HPA catalysts, the yields of FA and AA are equal to their
selectivities. Our recent studies indicated that under similar aque-
ous phase aerobic oxidation conditions, H3PW12O40 can efficiently
catalyze the hydrolysis of cellulose to glucose but is nearly inac-
tive for the catalytic oxidation of monosaccharide [32,33], while
H3PMo12O40 selectively converts cellulose to glycolic acid with FA
as a secondary product [27]. These results were well reproduced in
this study, i.e., H3PW12O40 and H3PMo12O40 gave low yields (<10%)
for FA and AA (Fig. 1). In contrast, the four V-substituted HPAs all
showed remarkable yields of FA (>35%) along with small amounts
of AA (∼5%), indicating the important role of V on the selectivity
2. Experimental
The oxidative conversions of cellulose by various HPA catalysts
were carried out in a 75 mL Teflon-lined stainless autoclave at 453 K
for 3 h under 0.4–2 MPa O2 with a stirring rate of 600 rpm. Typically,
the reaction mixture comprised 20 mL of H2O, 0.2 g of ␣-cellulose
powder (containing 1.23 mmol glucose units), and 0.1 mmol of HPA
catalyst. In the reactions with other substrates, a fixed amount of
reactant (200 mg) and the typical reaction conditions were used
unless otherwise specified. In the reactions where the solid sub-
strates were not completely converted, the residual substrates
were separated from the solution after the reactions and the super-
natants were used for analysis. After each reaction, the liquid phase
was analysed by HPLC (Agilent, 1260 Infinity Quaternary System)
equipped with refractive index (RI) and ultraviolet (UV) detectors,
and a Waters Shodex SUGAR SH-1011 column (8 × 300 mm) with
0.05 M H2SO4 aqueous solution as the mobile phase at 313 K. The
gas phase was analysed by a GC with a FID detector. Quantifica-
tion of products was carried out for HPLC and GC analysis using an
external standard method. Calibration was done by using a series
of standards covering the concentration range of interest. The peak
for each component was integrated and the peak area was plotted
against concentration to give a calibration curve. The concentra-
tions of the major products were determined from the calibration
curves. Yields of the products were calculated based on the contents
of carbon in the substrates, which were determined via C elemental
analysis.
Please cite this article in press as: J. Zhang, et al., Catalytic oxidative conversion of cellulosic biomass to formic acid and acetic acid with

 Zhang, Jizhe
Zhang, Jizhe
 Sun, Miao
Sun, Miao
 Liu, Xin
Liu, Xin
 Han, Yu
Han, Yu